Smith, N.A.
Environmental and Public Health Issues
The main environmental problems associated with electrical appliance and equipment manufacture involve pollution and treatment of materials discarded during the manufacturing processes, together with the recycling, where possible, of the complete product when it has reached the end of its life.
Batteries
The exhaust of air contaminated with acid, alkali, lead, cadmium and other potentially harmful materials into the atmosphere and the pollution of water from the manufacturing of batteries should be prevented as far as possible, and where this is not possible it should be monitored to ensure compliance with relevant legislation.
The use of batteries can generate public health concerns. Leaking lead-acid or alkaline batteries can result in burns from the electrolyte. Recharging large lead-acid batteries can produce hydrogen gas, a fire and explosion hazard in enclosed areas. Release of thionyl chloride or sulphur dioxide from large lithium batteries can involve exposure to sulphur dioxide, hydrochloric acid mist, burning lithium and so on, and has caused at least one fatality (Ducatman, Ducatman and Barnes 1988). This could also be a hazard during manufacture of these batteries.
Battery manufacturers have become aware of increasing environmental concern from the disposal of batteries containing toxic heavy metals by putting them in landfills or incinerating them with other garbage. Leakage of toxic metals from waste dumps or alternatively escaping from the chimneys of waste incinerators can result in water and air contamination. The manufacturers therefore recognized the need to reduce the mercury content of batteries, in particular, within the limits allowable by modern technology. The campaign for mercury elimination commenced in advance of the legislation introduced in the European Union, the EC Battery Directive.
Recycling is another way to deal with environmental pollution. Nickel-cadmium batteries can be recycled relatively easily. The recovery of cadmium is very efficient and it is re-used in the construction of nickel-cadmium batteries. The nickel will subsequently be used in the steel industry. The initial economics suggested that the recycling of nickel-cadmium batteries was not cost effective, but advances in technology are expected to improve the situation. Mercuric oxide cells, which are covered by the EC Battery Directive, have been used primarily in hearing aids, and are being replaced typically with lithium or zinc-air batteries. Silver oxide cells are recycled, especially by the jewellery industry, due to the value of the silver content.
When recycling harmful materials, care has to be taken similar to that exercised during the manufacturing processes. During the recycling of silver batteries, for example, workers may be exposed to mercury vapour and silver oxide.
The repair and recycling of lead-acid batteries can result not only in lead poisoning among the workers, and sometimes their families, but also in extensive lead contamination of the environment (Matte et al. 1989). In many countries, particularly in the Caribbean and Latin America, lead car battery plates are burned to produce lead oxide for pottery glazes.
Electric Cable Manufacture
Electric cable manufacture has three major sources of pollution: solvent vapours, potential release of toluene di-isocyanate from enamelled wire manufacture and environmental emissions during the manufacture of materials used in cables. All of these require appropriate environmental controls.
Electric Lamp and Tube Manufacture
The major environmental concerns here are the waste disposal and/or recycling of mercury-containing lamps and the disposal of PCBs from the ballasts of fluorescent lamps. Glass manufacturing can also be a significant source of emission of nitrogen oxides into the atmosphere.
Domestic Electric Appliances
Since the electric appliance industry is to a large extent an assembly industry, environmental issues are minimal, with the major exception being paints and solvents used as surface coatings. Standard pollution control measures should be instituted in accordance with environmental regulations.
The recycling of electrical appliances involves separation of the recovered equipment into different materials such as copper and mild steel which can be reused, which is discussed elsewhere in this Encyclopaedia.
Domestic Electrical Appliance Manufacture
Adapted from 3rd edition, Encyclopaedia of Occupational Health and Safety.
The domestic electrical appliance industry is responsible for the manufacture of a wide-ranging variety of equipment including appliances designed for audio-visual, cooking, heating, food preparation and storage (refrigeration) uses. The production and manufacture of such appliances involve many highly-automated processes which can have associated health hazards and disease patterns.
Manufacturing Processes
Materials used in the manufacture of domestic electrical appliances can be categorized into:
- metals which are used typically for electric conductors in cables and appliance structure and/or framework
- dielectrics or insulating materials used for prevention of accidental contact with live electrical equipment
- paints and finishes
- chemicals.
Examples of the materials included in the four categories referred to are shown in table 1.
Table 1. Examples of materials used in the manufacture of domestic electrical appliances
|
Metals |
Dielectrics |
Paints/finishes |
Chemicals |
|
Steel |
Inorganic materials (e.g., mica) |
Paints |
Acids |
|
Aluminium |
Plastics (e.g., PVC) |
Lacquers |
Alkalis |
|
Lead |
Rubber |
Varnishes |
Solvents |
|
Cadmium |
Silico-organic materials |
Corrosion-resistant treatments |
|
|
Mercury |
Other polymers (e.g., nylon) |
Note: Lead and mercury are decreasingly common in domestic electrical appliance manufacturing
The materials used in the domestic electrical appliance industry must satisfy exacting requirements, including the ability to withstand the handling likely to be encountered in normal operation, the ability to withstand metal fatigue and the ability to be unaffected by any other processes or treatment which could render the appliance dangerous to use either immediately or after a prolonged period of time.
The materials used in the industry will often be received at the appliance assembly stage having already undergone several manufacturing processes, each of which is likely to have its own hazards and health problems. Details of these hazards and problems are considered under the appropriate chapters elsewhere in this Encyclopaedia.
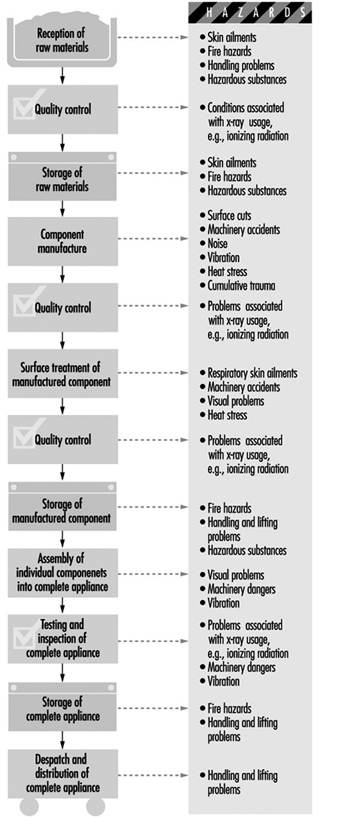
The manufacturing processes will vary from product to product, but in general will follow the production flow shown in figure 1. This chart also shows the hazards associated with the different processes.
Figure 1. Manufacturing process sequence & hazards
Health and Safety Issues
Fire and explosion
Many of the solvents, paints and insulating oils used in the industry are flammable substances. These materials should be stored in suitable cool, dry premises, preferably in a fireproof building separate from the production facility. Containers should be clearly labelled and different substances well separated or stored apart as required by their flashpoints and their class of risk. In the case of insulating materials and plastics, it is important to obtain information on the combustibility or fire characteristics of each new substance used. Powdered zirconium, which is now used in significant quantities in the industry, is also a fire hazard.
The quantities of flammable substances issued from storerooms should be kept to the minimum required for production. When flammable liquids are being decanted, charges of static electricity may form, and consequently all containers should be grounded. Fire-extinguishing appliances must be provided and the personnel of the storeplace instructed in their use.
Painting of components is usually carried out in specially built paint booths, which must have adequate exhaust and ventilation equipment that, when used with personal protective equipment (PPE), will create a safe working environment.
During welding, special fire precautions should be taken.
Accidents
Reception, storage and dispatch of raw materials, components and finished products can give rise to accidents involving trips and falls, falling objects, fork trucks and so forth. Manual materials handling can also create ergonomic problems which can be alleviated by automation whenever possible.
Since numerous different processes are employed in the industry, the accident hazards will vary from shop to shop in the plant. During component production there will be machine hazards in the use of machine tools, power presses, plastics injection-moulding machines and so on, and efficient machinery guarding is essential. During electroplating, precautions must be taken against splashes of corrosive chemicals. During component assembly, the constant movement of components from one process to another means that the danger of accidents due to in-plant transport and mechanical handling equipment is high.
Quality testing does not give rise to any special safety problems. However, performance testing requires special precautions since the tests are often carried out on semi-finished or uninsulated appliances. During electrical testing, all live components, conductors, terminals and measuring instruments should be protected to prevent accidental contact. The workplace should be screened off, entrance of unauthorized persons prohibited and warning notices posted. In electrical testing areas, the provision of emergency switches is particularly advisable, and the switches should be in a prominent position so that in an emergency all equipment can be immediately de-energized.
For testing appliances that emit x rays or contain radioactive substances, there are radiation protection regulations. A competent supervisor should be made responsible for observance of the regulations.
There are special risks in the use of compressed gases, welding equipment, lasers, impregnation plant, spray-painting equipment, annealing and tempering ovens and high-voltage electrical installations.
During all repair and maintenance activities, adequate lockout/tagout programmes are essential.
Health Hazards
Occupational diseases associated with the manufacture of domestic electrical equipment are relatively low in number and not normally considered to be severe. Such problems that do exist are typified by:
- the development of skin conditions due to the use of solvents, cutting oils, hardeners used with epoxy resin and polychlorinated biphenyls (PCBs)
- the onset of silicosis due to the inhalation of silica in sandblasting (although sand is being increasingly replaced by less toxic blasting agents such as corundum, steel grit or shot)
- health problems due to inhalation of solvent vapours in painting and degreasing, and lead poisoning from use of lead pigments, enamels, etc.
- varying levels of noise produced during the processes.
Wherever possible, highly toxic solvents and chlorinated compounds should be replaced by less dangerous substances; under no circumstances should benzene or carbon tetrachloride be employed as solvents. Lead poisoning may be overcome by substitution of safer materials or techniques and the strict application of safe working procedures, personal hygiene and medical supervision. Where there is a danger of exposure to hazardous concentrations of atmospheric contaminants, the workplace air should be regularly monitored, and appropriate measures such as the installation of an exhaust system taken where necessary. The noise hazard may be reduced by enclosure of noise sources, the use of sound-absorbent materials in workrooms or the use of personal hearing protection.
Safety engineers and industrial physicians should be called upon at the design and planning stage of new plants or operations, and the hazards of processes or machines should be eliminated before processes are started up. This should be followed up by regular inspection of machines, tools, plant, transport equipment, firefighting appliances, workshops and test areas and so on.
Worker participation in the safety effort is essential, and supervisors should ensure that personal protective equipment is available and worn where necessary. Particular attention should be paid to the safety training of new workers, since these account for a relatively high proportion of accidents.
Workers should receive a pre-placement medical examination and, where there is the possibility of hazardous exposure, periodic examination as necessary.
Many processes in the production of individual components will involve the rejection of waste material (e.g., “swarf” from sheet or bar metal), and the disposal of such materials must be in accordance with safety requirements. Furthermore, if such process waste cannot be returned to the producer or manufacturer for recycling, then its subsequent disposal must be by approved processes in order to avoid environmental pollution.
Batteries
The term battery refers to a collection of individual cells, which can generate electricity though chemical reactions. Cells are categorized as either primary or secondary. In primary cells, the chemical reactions that produce the electron flow are not reversible, and therefore the cells are not easily recharged. Conversely, secondary cells must be charged prior to their use, which is achieved by passing an electrical current through the cell. Secondary cells have the advantage that they can often be repeatedly recharged and discharged through use.
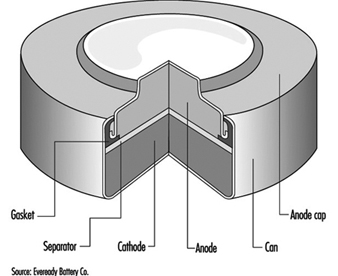
The classic primary battery in everyday use is the Leclanché dry cell, so called because the electrolyte is a paste, not a liquid. The Leclanché cell is typified by the cylindrical batteries used in flashlights, portable radios, calculators, electric toys and the like. In recent years, alkaline batteries, such as the zinc-manganese dioxide cell, have become more prevalent for this type of use. Miniature or “button” batteries have found use in hearing aids, computers, watches, cameras and other electronic equipment. The silver oxide-zinc cell, mercury cell, the zinc-air cell, and the lithium-manganese dioxide cell are some examples. See figure 1 for a cutaway view of a typical alkaline miniature battery.
Figure 1. Cutaway view of alkaline miniature battery
The classic secondary or storage battery is the lead-acid battery, widely used in the transportation industry. Secondary batteries are also used in power plants and industry. Rechargeable, battery-operated tools, toothbrushes, flashlights and the like are a new market for secondary cells. Nickel-cadmium secondary cells are becoming more popular, especially in pocket cells for emergency lighting, diesel starting and stationary and traction applications, where the reliability, long life, frequent rechargeability and low-temperature performance outweigh their extra cost.
Rechargeable batteries under development for use in electric vehicles utilize lithium-ferrous sulphide, zinc-chlorine and sodium-sulphur.
Table 1 gives the composition of some common batteries.
Table 1. Composition of common batteries
|
Type of battery |
Negative electrode |
Positive electrode |
Electrolyte |
|
Primary cells |
|||
|
Leclanché dry cell |
Zinc |
Manganese dioxide |
Water, zinc chloride, ammonium chloride |
|
Alkaline |
Zinc |
Manganese dioxide |
Potassium hydroxide |
|
Mercury (Ruben’s cell) |
Zinc |
Mercuric oxide |
Potassium hydroxide, zinc oxide, water |
|
Silver |
Zinc |
Silver oxide |
Potassium hydroxide, zinc oxide, water |
|
Lithium |
Lithium |
Manganese dioxide |
Lithium chlorate, LiCF3SO3 |
|
Lithium |
Lithium |
Sulphur dioxide |
Sulphur dioxide, acetonitrile, lithium bromide |
|
Thionyl chloride |
Lithium aluminium chloride |
||
|
Zinc in air |
Zinc |
Oxygen |
Zinc oxide, potassium hydroxide |
|
Secondary cells |
|||
|
Lead-acid |
Lead |
Lead dioxide |
Dilute sulphuric acid |
|
Nickel-iron (Edison battery) |
Iron |
Nickel oxide |
Potassium hydroxide |
|
Nickel-cadmium |
Cadmium hydroxide |
Nickel hydroxide |
Potassium hydroxide, possibly lithium hydroxide |
|
Silver-zinc |
Zinc powder |
Silver oxide |
Potassium hydroxide |
Manufacturing Processes
While there are clear differences in the manufacture of the different types of batteries, there are several processes which are common: weighing, grinding, mixing, compressing and drying of constituent ingredients. In modern battery plants many of these processes are enclosed and highly automated, using sealed equipment. Therefore, exposure to the various ingredients can occur during weighing and loading and during cleaning of the equipment.
In older battery plants, many of the grinding, mixing and other operations are done manually, or the transfer of ingredients from one step of the process to another is done manually. In these instances, the risk of inhalation of dusts or skin contact with corrosive substances is high. Precautions for dust-producing operations include total enclosure and mechanized handling and weighing of powders, local exhaust ventilation, daily wet mopping and/or vacuuming and wearing of respirators and other personal protective equipment during maintenance operations.
Noise is also a hazard, since compressing machines and wrapping machines are noisy. Noise control methods and hearing conservation programmes are essential.
The electrolytes in many batteries contain corrosive potassium hydroxide. Enclosure and skin and eye protection are indicated precautions. Exposures can also occur to the particulates of toxic metals such as cadmium oxide, mercury, mercuric oxide, nickel and nickel compounds, and lithium and lithium compounds, which are used as anodes or cathodes in particular types of batteries. The lead-acid storage battery, sometimes referred to as the accumulator, can involve considerable lead exposure hazards and is discussed separately in the article “Lead-acid battery manufacture”.
Lithium metal is highly reactive, thus lithium batteries must be assembled in a dry atmosphere in order to avoid the lithium reacting with water vapour. Sulphur dioxide and thionyl chloride, used in some lithium batteries, are respiratory hazards. Hydrogen gas, used in nickel-hydrogen batteries, is a fire and explosion hazard. These, as well as materials in newly developed batteries, will require special precautions.
Leclanché Cells
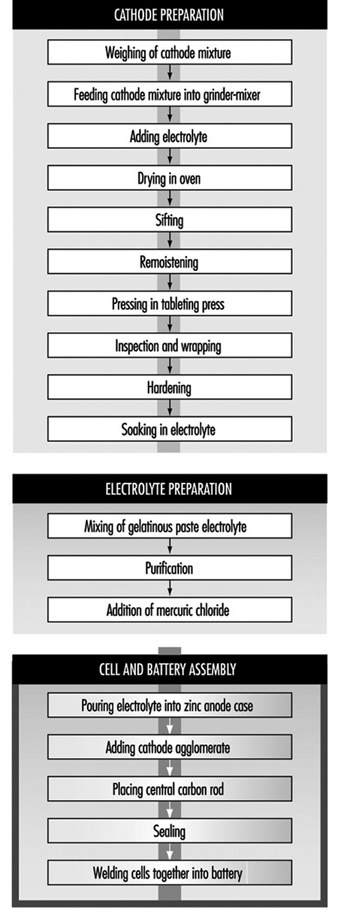
Leclanché dry-cell batteries are produced as shown in figure 2. The positive electrode or cathode mixture comprises 60 to 70% manganese dioxide, the remainder being made up of graphite, acetylene black, ammonium salts, zinc chloride and water. Dry, finely ground manganese dioxide, graphite and acetylene black are weighed and fed into a grinder-mixer; electrolyte containing water, zinc chloride and ammonium chloride is added, and the prepared mixture is pressed on a hand-fed tableting or agglomerating press. In certain cases, the mixture is dried in an oven, sifted and remoistened before tableting. The tablets are inspected and wrapped on hand-fed machines after being allowed to harden for a few days. The agglomerates are then placed in trays and soaked in electrolyte, and are now ready for assembly.
Figure 2. Leclanché cell battery production
The anode is the zinc case, which is prepared from zinc blanks on a hot press (or zinc sheets are folded and welded to the case). An organic gelatinous paste consisting of maize and flour starches soaked in electrolyte is mixed in large vats. The ingredients are usually poured in from sacks without weighing. The mixture is then purified with zinc chips and manganese dioxide. Mercuric chloride is added to the electrolyte to form an amalgam with the interior of the zinc container. This paste will form the conducting medium or electrolyte.
Cells are assembled by automatic pouring of the required amount of gelatinous paste into the zinc cases to form an inner sleeve lining on the zinc container. In some cases, the cases receive a chromate finish by the pouring in and emptying of a mixture of chromic and hydrochloric acid before adding the gelatinous paste. The cathode agglomerate is then placed in position in the centre of the case. A carbon rod is placed centrally in the cathode to act as the current collector.
The zinc cell is then sealed with molten wax or paraffin and heated with a flame to give a better seal. The cells are then welded together to form the battery. The reaction of the battery is:
2 MnO2 + 2 NH4Cl + Zn → ZnCl2 + H2O2 + Mn2O3
Workers may be exposed to manganese dioxide during weighing, mixer loading, grinding, cleaning the oven, sifting, hand pressing and wrapping, depending on the degree of automation, sealed enclosure and local exhaust ventilation. In manual pressing and wet wrapping, there may be exposure to the wet mixture, which can dry to produce inhalable dust; dermatitis may occur from exposure to the slightly corrosive electrolyte. Personal hygiene measures, gloves and respiratory protection for cleaning and maintenance operations, showering facilities and separate lockers for work and street clothes can reduce these risks. As mentioned above, noise hazards can result from the wrapping and tableting press.
Mixing is automatic during manufacture of the gelatinous paste, and the only exposure is during addition of the materials. During addition of mercuric chloride to the gelatinous paste, there is the risk of inhalation and skin absorption and possible mercury poisoning. LEV or personal protective equipment is necessary.
Exposure to spills of chromic acid and hydrochloric acid during chromating and exposure to welding fumes and fumes from heating the sealing compound are also possible. Mechanization of the chromating process, use of gloves and LEV for heat sealing and welding are suitable precautions.
Nickel-Cadmium Batteries
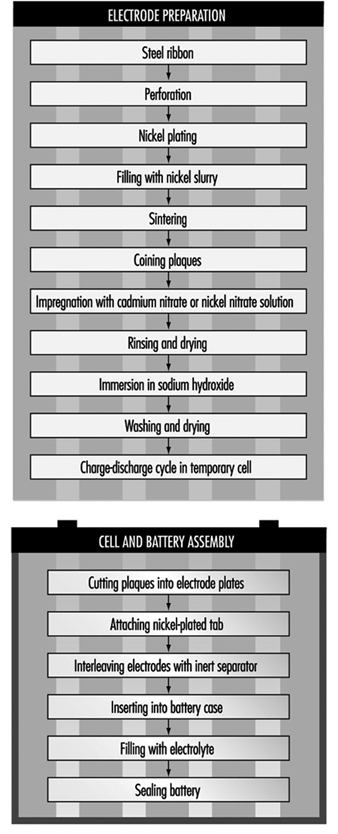
The most common method today of making nickel-cadmium electrodes is by depositing the active electrode material directly into a porous sintered nickel substrate, or plate. (See figure 3.) The plate is prepared by pressing a paste of sintered grade nickel powder (often made by decomposition of nickel carbonyl) into the open grid of nickel-plated perforated sheet steel (or nickel gauze or nickel-plated steel gauze) and then sintering or drying in an oven. These plates may then be cut, weighed and coined (compressed) for particular purposes or rolled into a spiral for household-type cells.
Figure 3. Nickel-cadmium battery production
The sintered plaque is then impregnated with nickel nitrate solution for the positive electrode or cadmium nitrate for the negative electrode. These plaques are rinsed and dried, immersed in sodium hydroxide to form nickel hydroxide or cadmium hydroxide and washed and dried again. Usually the next step is to immerse the positive and negative electrodes in a large temporary cell containing 20 to 30% sodium hydroxide. Charge-discharge cycles are run to remove impurities and the electrodes are removed, washed and dried.
An alternative way of making cadmium electrodes is to prepare a paste of cadmium oxide mixed with graphite, iron oxide and paraffin, which is milled and finally compacted between rollers to form the active material. This is then pressed into a moving perforated steel strip that is dried, sometimes compressed, and cut into plates. Lugs may be attached at this stage.
The next steps involve cell and battery assembly. For large batteries, the individual electrodes are then assembled into electrode groups with plates of opposite polarity interleaved with plastic separators. These electrode groups may be bolted or welded together and placed in a nickel-plated steel casing. More recently, plastic battery casings have been introduced. The cells are filled with an electrolyte solution of potassium hydroxide, which may also contain lithium hydroxide. The cells are then assembled into batteries and bolted together. Plastic cells may be cemented or taped together. Each cell is connected with a lead connector to the adjacent cell, leaving a positive and negative terminal at the ends of the battery.
For cylindrical batteries, the impregnated plates are assembled into electrode groups by winding the positive and negative electrodes, separated by an inert material, into a tight cylinder. The electrode cylinder is then placed in a nickel-plated metal case, potassium hydroxide electrolyte is added and the cell is sealed by welding.
The chemical reaction involved in the charging and discharging of nickel-cadmium batteries is:
![]()
The major potential exposure to cadmium occurs from handling of cadmium nitrate and its solution while making paste from cadmium oxide powder and handling the dried active powders. Exposure can also occur during reclamation of cadmium from scrap plates. Enclosure and automated weighing and mixing can reduce these hazards during the early steps.
Similar measures can control exposures to nickel compounds. The production of sintered nickel from nickel carbonyl, although done in sealed machinery, involves potential exposure to extremely toxic nickel carbonyl and carbon monoxide. The process requires continuous monitoring for gas leaks.
The handling of caustic potassium or lithium hydroxide requires suitable ventilation and personal protection. Welding generates fumes and requires LEV.
Health Effects and Disease Patterns
The most serious health hazards in traditional battery making are lead, cadmium, mercury and manganese dioxide exposures. Lead hazards are discussed elsewhere in this chapter and Encyclopaedia. Cadmium can cause kidney disease and is carcinogenic. Cadmium exposure was found to be widespread in US nickel-cadmium battery plants, and many workers have had to be medically removed under the Occupational Safety and Health Administration’s Cadmium Standard provisions due to high cadmium levels in blood and urine (McDiarmid et al. 1996). Mercury affects the kidneys and nervous system. Excessive exposure to mercury vapour has been shown in studies of several mercury battery plants (Telesca 1983). Manganese dioxide exposures have been shown to be high in powder mixing and handling in alkaline dry cell manufacturing (Wallis, Menke and Chelton 1993). This can result in neurofunctional deficits in battery workers (Roels et al. 1992). Manganese dusts can, if absorbed in excessive quantities, lead to disorders of the central nervous system similar to Parkinson’s syndrome. Other metals of concern include nickel, lithium, silver and cobalt.
Skin burns can result from exposure to zinc chloride, potassium hydroxide, sodium hydroxide and lithium hydroxide solutions used in the electrolytes of batteries.
General Profile
Overview of the Sector
Electrical equipment includes a wide-ranging field of devices. It would be impossible to include information on all items of equipment, and this chapter will therefore be limited to coverage of products of some of the major industries. Numerous processes are involved in the manufacture of such equipment. This chapter discusses the hazards likely to be encountered by persons working in the manufacture of batteries, electric cables, electric lamps and general domestic electrical equipment. It concentrates upon electrical equipment; electronic equipment is discussed in detail in the chapter Microelectronics and semiconductors.
Evolution of the Industry
The pioneering discovery of electromagnetic induction was instrumental in the development of today’s vast electrical industry. The discovery of the electrochemical effect led to the development of batteries as a means of supplying electrical equipment from portable power sources using direct current systems. As devices which relied upon power from mains were invented, a system of transmission and distribution of electricity was required, which led to the introduction of flexible electrical conductors (cables).
The early forms of artificial lighting (i.e., carbon arc and gas lighting) were superseded by the filament lamp (originally with a carbon filament, exhibited by Joseph Swan in England in January 1879). The filament lamp was to enjoy an unprecedented monopoly in domestic, commercial and industrial applications prior to the outbreak of the Second World War, at which stage the fluorescent lamp was introduced. Other forms of discharge lighting, all of which depend upon the passage of an electric current through a gas or vapour, have subsequently been developed and have a variety of applications in commerce and industry.
Other electrical appliances in many fields (e.g., audio-visual, heating, cooking and refrigeration) are constantly being developed, and the range of such devices is increasing. This is typified by the introduction of satellite television and the microwave cooker.
While the availability and accessibility of raw materials had a significant effect upon the development of the industries, the locations of the industries were not necessarily determined by the locations of the raw material sources. The raw materials are often processed by a third party before being used in the assembly of electrical appliances and equipment.
Characteristics of the Workforce
The skills and expertise possessed by those who work in the industry now are different from those possessed by the workforce in earlier years. Equipment used in the production and manufacture of batteries, cables, lamps and domestic electrical appliances is highly automated.
In many instances those who are currently involved in the industry require specialized training in order to carry out their work. Teamwork is a significant factor in the industry, since many processes involve production line systems, where the work of individuals depends upon the work of others.
An ever-increasing number of manufacturing processes involved in the production of electrical appliances rely on some form of computerization. It is necessary, therefore, for the workforce to be familiar with computer techniques. This may not present any problems to the younger workforce, but older workers may not have had any previous computer experience, and it is likely that they will need to be re-trained.
Economic Importance of the Industry
Some countries benefit more than others from the electrical appliances and equipment industry. The industry has economic importance for those countries from which raw materials are obtained and those in which the end products are assembled and/or constructed. Assembly and construction take place in many different countries.
Raw materials do not have infinite availability. Discarded equipment should be re-used wherever possible. However, the costs involved in recovering those parts of discarded equipment which may be re-used may ultimately be prohibitive.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."