Humans live their entire lives within a very small, fiercely protected range of internal body temperatures. The maximal tolerance limits for living cells range from about 0ºC (ice crystal formation) to about 45ºC (thermal coagulation of intracellular proteins); however, humans can tolerate internal temperatures below 35ºC or above 41ºC for only very brief periods of time. To maintain internal temperature within these limits, people have developed very effective and in some instances specialized physiological responses to acute thermal stresses. These responses—designed to facilitate the conservation, production or elimination of body heat—involve the finely controlled coordination of several body systems.
Human Thermal Balance
By far, the largest source of heat imparted to the body results from metabolic heat production (M). Even at peak mechanical efficiency, 75 to 80% of the energy involved in muscular work is liberated as heat. At rest, a metabolic rate of 300 ml O2 per minute creates a heat load of approximately 100 Watts. During steady-state work at an oxygen consumption of 1 l/min, approximately 350 W of heat are generated—less any energy associated with external work (W). Even at such a mild to moderate work intensity, body core temperature would rise approximately one degree centigrade every 15 min were it not for an efficient means of heat dissipation. In fact, very fit individuals can produce heat in excess of 1,200 W for 1 to 3 hours without heat injury (Gisolfi and Wenger 1984).
Heat can also be gained from the environment via radiation (R) and convection (C) if the globe temperature (a measure of radiant heat) and air (dry-bulb) temperature, respectively, exceed skin temperature. These avenues of heat gain are typically small relative to M, and actually become avenues of heat loss when the skin-to-air thermal gradient is reversed. The final avenue for heat loss—evaporation (E)—is also typically the most important, since the latent heat of vaporization of sweat is high—approximately 680 W-h/l of sweat evaporated. These relations are discussed elsewhere in this chapter.
Under cool to thermoneutral conditions, heat gain is balanced by heat loss, no heat is stored, and body temperature equilibrates; that is:
M–W ± R ± C–E = 0
However, in more severe exposure to heat:
M–W ± R ± C >E
and heat is stored. In particular, heavy work (high energy expenditure which increases M–W), excessively high air temperatures (which increase R+C), high humidity (which limits E) and the wearing of thick or relatively impermeable clothing (which creates a barrier to effective evaporation of sweat) create such a scenario. Finally, if exercise is prolonged or hydration inadequate, E may be outstripped by the limited ability of the body to secrete sweat (1 to 2 l/h for short periods).
Body Temperature and Its Control
For purposes of describing physiological responses to heat and cold, the body is divided into two components—the “core” and the “shell”. Core temperature (Tc) represents internal or deep body temperature, and can be measured orally, rectally or, in laboratory settings, in the oesophagus or on the tympanic membrane (eardrum). The temperature of the shell is represented by mean skin temperature (Tsk). The average temperature of the body (Tb) at any time is a weighted balance between these temperatures, that is
Tb = k Tc + (1– k) Tsk
where the weighting factor k varies from about 0.67 to 0.90.
When confronted with challenges to thermal neutrality (heat or cold stresses), the body strives to control Tc through physiological adjustments, and Tc provides the major feedback to the brain to coordinate this control. While the local and mean skin temperature are important for providing sensory input, Tsk varies greatly with ambient temperature, averaging about 33 ºC at thermoneutrality and reaching 36 to 37 ºC under conditions of heavy work in the heat. It can drop considerably during whole-body and local exposure to cold; tactile sensitivity occurs between 15 and 20 ºC, whereas the critical temperature for manual dexterity is between 12 and 16 ºC. The upper and lower pain threshold values for Tsk are approximately 43 ºC and 10 ºC, respectively.
Precise mapping studies have localized the site of greatest thermoregulatory control in an area of the brain known as the pre- optic/anterior hypothalamus (POAH). In this region are nerve cells which respond to both heating (warm-sensitive neurons) and cooling (cold-sensitive neurons). This area dominates control of body temperature by receiving afferent sensory information about body temperature and sending efferent signals to the skin, the muscles and other organs involved in temperature regulation, via the autonomic nervous system. Other areas of the central nervous system (posterior hypothalamus, reticular formation, pons, medulla and spinal cord) form ascending and descending connections with the POAH, and serve a variety of facilitory functions.
The body’s control system is analogous to thermostatic control of temperature in a house with both heating and cooling capabilities. When body temperature rises above some theoretical “set point” temperature, effector responses associated with cooling (sweating, increasing skin blood flow) are turned on. When body temperature falls below the set point, heat gain responses (decreasing skin blood flow, shivering) are initiated. Unlike home heating/cooling systems however, the human thermoregulatory control system does not operate as a simple on-off system, but also has proportional control and rate-of-change control characteristics. It should be appreciated that a “set point temperature” exists in theory only, and thus is useful in visualizing these concepts. Much work is yet to be done toward a full understanding of the mechanisms associated with the thermoregulatory set point.
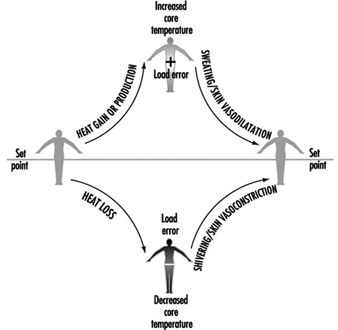
Whatever its basis, the set point is relatively stable and is unaffected by work or ambient temperature. In fact, the only acute perturbation known to shift the set point is the group of endogenous pyrogens involved in the febrile response. The effector responses employed by the body to maintain thermal balance are initiated and controlled in response to a “load error”, that is, a body temperature which is transiently above or below the set point (figure 1). A core temperature below the set point creates a negative load error, resulting in heat gain (shivering, vasoconstriction of the skin) being initiated. A core temperature above the set point creates a positive load error, leading to heat loss effectors (skin vasodilatation, sweating) being turned on. In each case, the resultant heat transfer decreases the load error and helps return the body temperature to a steady state.
Figure 1. A model of thermoregulation in the human body.
Temperature Regulation in the Heat
As mentioned above, humans lose heat to the environment primarily through a combination of dry (radiation and convection) and evaporative means. To facilitate this exchange, two primary effector systems are turned on and regulated—skin vasodilatation and sweating. While skin vasodilatation often results in small increases in dry (radiative and convective) heat loss, it functions primarily to transfer heat from the core to the skin (internal heat transfer), while evaporation of sweat provides an extremely effective means of cooling the blood prior to its return to deep body tissues (external heat transfer).
Skin vasodilatation
The amount of heat transferred from the core to the skin is a function of the skin blood flow (SkBF), the temperature gradient between core and skin, and the specific heat of blood (a little less than 4 kJ/°C per litre of blood). At rest in a thermoneutral environment, the skin gets approximately 200 to 500 ml/min of blood flow, representing only 5 to 10% of the total blood pumped by the heart (cardiac output). Because of the 4ºC gradient between Tc (about 37ºC) and Tsk (about 33ºC under such conditions), the metabolic heat produced by the body to sustain life is constantly convected to the skin for dissipation. By contrast, under conditions of severe hyperthermia such as high-intensity work in hot conditions, the core-to-skin thermal gradient is smaller, and the necessary heat transfer is accomplished by large increases in SkBF. Under maximal heat stress, SkBF can reach 7 to 8 l/min, about one-third of cardiac output (Rowell 1983). This high blood flow is achieved through a poorly understood mechanism unique to humans which has been called the “active vasodilator system”. Active vasodilatation involves sympathetic nerve signals from the hypothalamus to the skin arterioles, but the neurotransmitter has not been determined.
As mentioned above, SkBF is primarily responsive to increases in Tc and, to a lesser extent, Tsk. Tc rises as muscular work is initiated and metabolic heat production begins, and once some threshold Tc is reached, SkBF also begins to increase dramatically. This basic thermoregulatory relationship is also acted upon by non-thermal factors. This second level of control is critical in that it modifies SkBF when overall cardiovascular stability is threatened. The veins in the skin are very compliant, and a significant portion of the circulating volume pools in these vessels. This aids in heat exchange by slowing the capillary circulation to increase transit time; however, this pooling, coupled with fluid losses from sweating, may also decrease the rate of blood return to the heart. Among the non-thermal factors which have been shown to influence SkBF during work are upright posture, dehydration and positive-pressure breathing (respirator use). These act through reflexes which are turned on when cardiac filling pressure is decreased and stretch receptors located in the large veins and right atrium are unloaded, and are therefore most evident during prolonged aerobic work in an upright posture. These reflexes function to maintain arterial pressure and, in the case of work, to maintain adequate blood flow to active muscles. Thus, the level of SkBF at any given point in time represents the aggregate effects of thermoregulatory and non-thermoregulatory reflex responses.
The need to increase blood flow to the skin to aid in temperature regulation greatly impacts on the ability of the cardiovascular system to regulate blood pressure. For this reason, a coordinated response of the entire cardiovascular system to heat stress is necessary. What cardiovascular adjustments occur that allow for this increase in cutaneous flow and volume? During work in cool or thermoneutral conditions, the needed increase in cardiac output is well supported by increasing heart rate (HR), since further increases in stroke volume (SV) are minimal beyond exercise intensities of 40% of maximum. In the heat, HR is higher at any given work intensity as compensation for the reduced central blood volume (CBV) and SV. At higher levels of work, maximal heart rate is reached, and this tachycardia is therefore incapable of sustaining the necessary cardiac output. The second way in which the body supplies a high SkBF is by distributing blood flow away from such areas as the liver, kidneys and intestines (Rowell 1983). This redirection of flow can provide an additional 800 to 1,000 ml of blood flow to the skin, and helps offset the detrimental effects of peripheral pooling of blood.
Sweating
Thermoregulatory sweat in humans is secreted from 2 to 4 million eccrine sweat glands scattered non-uniformly over the body surface. Unlike apocrine sweat glands, which tend to be clustered (on the face and hands and in the axial and genital regions) and which secrete sweat into hair follicles, eccrine glands secrete sweat directly onto the skin surface. This sweat is odourless, colourless and relatively dilute, since it is an ultrafiltrate of plasma. Thus it has a high latent heat of vaporization and is ideally suited for its cooling purpose.
As an example of the effectiveness of this cooling system, a man working at an oxygen cost of 2.3 l/min produces a net metabolic heat (M–W) of about 640 W. Without sweating, body temperature would increase at a rate of about 1°C every 6 to 7 min. With efficient evaporation of about 16 g of sweat per minute (a reasonable rate), the rate of heat loss can match the rate of heat production, and body core temperature can be maintained at a steady state; that is,
M–W±R±C–E = 0
Eccrine glands are simple in structure, consisting of a coiled secretory portion, a duct and a skin pore. The volume of sweat produced by each gland is dependent upon both the structure and the function of the gland, and total sweating rate in turn depends on both the recruitment of glands (active sweat gland density) and sweat gland output. The fact that some people sweat more heavily than others is attributable mainly to differences in sweat gland size (Sato and Sato 1983). Heat acclimation is another major determinant of sweat production. With ageing, lower sweating rates are attributable not to fewer activated eccrine glands, but to a decreased sweat output per gland (Kenney and Fowler 1988). This decline probably relates to a combination of structural and functional alterations which accompany the ageing process.
Like vasomotor signals, nerve impulses to the sweat glands originate in the POAH and descend through the brainstem. The fibres which innervate the glands are sympathetic cholinergic fibres, a rare combination in the human body. While acetylcholine is the primary neurotransmitter, adrenergic transmitters (catecholamines) also stimulate eccrine glands.
In many ways, control of sweating is analogous to control of skin blood flow. Both have similar onset characteristics (threshold) and linear relationships to increasing Tc. The back and chest tend to have earlier onsets of sweating, and the slopes for the relationship of local sweat rate to Tc are steepest for these sites. Like SkBF, sweating is modified by non-thermal factors such as hypohydration and hyperosmolality. Also worth noting is a phenomenon called “hidromeiosis”, which occurs in very humid environments or on skin areas constantly covered with wet clothing. Such areas of skin, due to their continuously wet state, decrease sweat output. This serves as a protective mechanism against continued dehydration, since sweat which stays on the skin rather than evaporating serves no cooling function.
If sweating rate is adequate, evaporative cooling is determined ultimately by the water vapour pressure gradient between the wet skin and the air surrounding it. Thus, high humidity and heavy or impermeable clothing limit evaporative cooling, while dry air, air movement about the body and minimal, porous clothing facilitate evaporation. On the other hand, if work is heavy and sweating profuse, evaporative cooling can likewise be limited by the body’s ability to produce sweat (maximally about 1 to 2 l/h).
Temperature Regulation in the Cold
One important difference in the way humans respond to cold compared to heat is that behaviour plays a much greater role in thermoregulatory response to cold. For example, wearing appropriate clothing and assuming postures which minimize surface area available for heat loss (“huddling”) are far more important in cold ambient conditions than in the heat. A second difference is the greater role played by hormones during cold stress, including the increased secretion of catecholamines (norepinephrine and epinephrine) and thyroid hormones.
Skin vasoconstriction
An effective strategy against heat loss from the body through radiation and convection is to increase the effective insulation provided by the shell. In humans this is accomplished by decreasing blood flow to the skin—that is, by skin vasoconstriction. Constriction of the cutaneous vessels is more pronounced in the extremities than on the trunk. Like active vasodilatation, skin vasoconstriction is also controlled by the sympathetic nervous system, and is influenced by Tc, Tsk and local temperatures.
The effect of skin cooling on the heart rate and blood pressure response varies with the area of the body which is cooled, and whether the cold is severe enough to cause pain. For example, when the hands are immersed in cold water, HR, systolic blood pressure (SBP) and diastolic blood pressure (DBP) all increase. When the face is cooled, SBP and DBP increase due to the generalized sympathetic response; however, HR goes down due to a parasympathetic reflex (LeBlanc 1975). To further confound the complexity of the overall response to cold, there is a wide range of variability in responses from one person to another. If the cold stress is of sufficient magnitude to decrease body core temperature, HR may either increase (due to sympathetic activation) or decrease (due to the increased central blood volume).
A specific case of interest is termed cold-induced vasodilatation (CIVD). When the hands are placed in cold water, SkBF initially decreases to conserve heat. As tissue temperatures drop, SkBF paradoxically increases, decreases again, and repeats this cyclical pattern. It has been suggested that CIVD is beneficial in preventing tissue damage from freezing, but this is unproven. Mechanistically, the transient dilation probably occurs when the direct effects of the cold are severe enough to decrease nerve transmission, which transiently overrides the effect of the cold on the blood vessel sympathetic receptors (mediating the constrictor effect).
Shivering
As body cooling progresses, the second line of defence is shivering. Shivering is the random involuntary contraction of superficial muscle fibres, which does not limit heat loss but rather increases heat production. Since such contractions do not produce any work, heat is generated. A resting person can increase his or her metabolic heat production about three- to fourfold during intense shivering, and can increase Tc by 0.5ºC. The signals to initiate shivering arise principally from the skin, and, in addition to the POAH region of the brain, the posterior hypothalamus is also involved to a large extent.
Although many individual factors contribute to shivering (and cold tolerance in general), one important factor is body fatness. A man with very little subcutaneous fat (2 to 3 mm thickness) starts shivering after 40 min at 15ºC and 20 min at 10ºC, while a man who has more insulating fat (11 mm) may not shiver at all at 15ºC and after 60 min at 10ºC (LeBlanc 1975).