Kraus, Richard S.
Address: Petroleum Safety Consultants, 8712 Chippendale Court, Annandale, VA 22003-3807
Country: United States
Phone: 1 (703) 978-0006
Fax: 1 (703) 978-2392
Past position(s): Mobil Oil Corporation, US Marketing and Refining Safety Executive Management
Education: BS, 1956, Purdue University, West Lafayette; PE, Professional Fire Protection Engineer (Wisconsin); Board Certified Forensic Examiner (American Board of Forensic Examiners); Emergency Programs Manager (Federal Emergency Management Agency)
Storage and Transportation of Crude Oil, Natural Gases, Liquid Petroleum Products and Other Chemicals
Pipelines, marine vessels, tank trucks, rail tank cars and so forth are used to transport crude oils, compressed and liquefied hydrocarbon gases, liquid petroleum products and other chemicals from their point of origin to pipeline terminals, refineries, distributors and consumers.
Crude oils and liquid petroleum products are transported, handled and stored in their natural liquid state. Hydrocarbon gases are transported, handled and stored in both the gaseous and liquid states and must be completely confined in pipelines, tanks, cylinders or other containers prior to use. The most important characteristic of liquefied hydrocarbon gases (LHGs) is that they are stored, handled and shipped as liquids, taking up a relatively small amount of space and then expanding into a gas when used. For example, liquefied natural gas (LNG) is stored at –162°C, and when it is released the difference in storage and atmospheric temperatures causes the liquid to expand and gasify. One gallon (3.8 l) of LNG converts to approximately 2.5 m3 of natural gas at normal temperature and pressure. Because liquefied gas is much more “concentrated” than compressed gas, more useable gas can be transported and provided in the same size container.
Pipelines
It is generally the case that all crude oils, natural gas, liquefied natural gas, liquefied petroleum gas (LPG) and petroleum products flow through pipelines at some time in their migration from the well to a refinery or gas plant, then to a terminal and eventually to the consumer. Aboveground, underwater and underground pipelines, varying in size from several centimetres to a metre or more in diameter, move vast amounts of crude oil, natural gas, LHGs and liquid petroleum products. Pipelines run throughout the world, from the frozen tundra of Alaska and Siberia to the hot deserts of the Middle East, across rivers, lakes, seas, swamps and forests, over and through mountains and under cities and towns. Although the initial construction of pipelines is difficult and expensive, once they are built, properly maintained and operated, they provide one of the safest and most economical means of transporting these products.
The first successful crude-oil pipeline, a 5-cm-diameter wrought iron pipe 9 km long with a capacity of about 800 barrels a day, was opened in Pennsylvania (US) in 1865. Today, crude oil, compressed natural gas and liquid petroleum products are moved long distances through pipelines at speeds from 5.5 to 9 km per hour by large pumps or compressors located along the route of the pipeline at intervals ranging from 90 km to over 270 km. The distance between pumping or compressor stations is determined by the pump capacity, viscosity of the product, size of the pipeline and the type of terrain crossed. Regardless of these factors, pipeline pumping pressures and flow rates are controlled throughout the system to maintain a constant movement of product within the pipeline.
Types of pipelines
The four basic types of pipelines in the oil and gas industry are flow lines, gathering lines, crude trunk pipelines and petroleum product trunk pipelines.
- Flow lines. Flow lines move crude oil or natural gas from producing wells to producing field storage tanks and reservoirs. Flow lines may vary in size from 5 cm in diameter in older, lower-pressure fields with only a few wells, to much larger lines in multi-well, high-pressure fields. Offshore platforms use flow lines to move crude and gas from wells to the platform storage and loading facility. A lease line is a type of flow line which carries all of the oil produced on a single lease to a storage tank.
- Gathering and feeder lines. Gathering lines collect oil and gas from several locations for delivery to central accumulating points, such as from field crude oil tanks and gas plants to marine docks. Feeder lines collect oil and gas from several locations for delivery direct into trunk lines, such as moving crude oil from offshore platforms to onshore crude trunk pipelines. Gathering lines and feeder lines are typically larger in diameter than flow lines.
- Crude trunk pipelines. Natural gas and crude oil are moved long distances from producing areas or marine docks to refineries and from refineries to storage and distribution facilities by 1- to 3-m- or larger-diameter trunk pipelines.
- Petroleum product trunk pipelines. These pipelines move liquid petroleum products such as gasoline and fuel oil from refineries to terminals, and from marine and pipeline terminals to distribution terminals. Product pipelines may also distribute products from terminals to bulk plants and consumer storage facilities, and occasionally from refineries direct to consumers. Product pipelines are used to move LPG from refineries to distributor storage facilities or large industrial users.
Regulations and standards
Pipelines are constructed and operated to meet safety and environmental standards established by regulatory agencies and industry associations. Within the United States, the Department of Transportation (DOT) regulates the operation of pipelines, the Environmental Protection Agency (EPA) regulates spills and releases, the Occupational Safety and Health Administration (OSHA) promulgates standards covering worker health and safety, and the Interstate Commerce Commission (ICC) regulates common carrier pipelines. A number of industry organizations, such as the American Petroleum Institute and the American Gas Association, also publish recommended practices covering pipeline operations.
Pipeline construction
Pipeline routes are planned using topographic maps developed from aerial photogrammetric surveys, followed by actual ground surveying. After planning the route, obtaining right-of-way and permission to proceed, base camps are established and a means of access for construction equipment is required. Pipelines can be constructed working from one end to another or simultaneously in sections which are then connected.
The first step in laying pipeline is to construct a 15- to 30-m-wide service road along the planned route to provide a stable base for the pipe-laying and pipe-joining equipment and for underground pipeline excavation and backfill equipment. The pipe sections are laid on the ground alongside the service road. The ends of the pipe are cleaned, the pipe is bent horizontally or vertically, as necessary, and the sections are held in position by chocks above the ground and joined by multi-pass electrical arc-welding. The welds are checked visually and then with gamma radiation to assure that no defects are present. Each connected section is then coated with liquid soap and air-pressure tested to detect leaks.
The pipeline is cleaned, primed and coated with a hot, tar-like material to prevent corrosion and wrapped in an outer layer of heavy paper, mineral wool or plastic. If the pipe is to be buried, the bottom of the trench is prepared with a sand or gravel bed. The pipe may be weighed down by short, concrete sleeves to prevent its lifting out of the trench from groundwater pressure. After the underground pipeline is placed in the trench, the trench is backfilled and the surface of the ground returned to normal appearance. After coating and wrapping, aboveground piping is lifted up onto prepared stanchions or casements, which may have various design features such as anti-earthquake shock absorption. Pipelines may be insulated or have heat trace capabilities to keep products at desired temperatures throughout transport. All pipeline sections are hydrostatically tested prior to entering gas or liquid hydrocarbon service.
Pipeline operations
Pipelines may be either privately owned and operated, carrying only the owner’s products, or they may be common carriers, required to carry any company’s products provided that the pipeline’s product requirements and tariffs are met. The three major pipeline operations are pipeline control, pumping or compressor stations and delivery terminals. Storage, cleaning, communication and shipment are also important functions.
- Pipeline control. Regardless of the product being transported, the size and length of the pipeline or the terrain, pipeline pumping stations, pressures and flow rates are completely controlled in order to ensure appropriate flow rates and continuous operations. Typically an operator and computer control the pumps, valves, regulators and compressors throughout the pipeline system from a central location.
- Oil pumping and gas compressor stations. Crude oil and petroleum products pumping stations and gas compressor stations are located at wellheads and along the pipeline route as needed to maintain pressure and volume. Pumps are driven by electric motors or diesel engines, and turbines may be powered by fuel oil, gas or steam. Many of these stations are automatically controlled and not staffed at most times. Pumps, with and without vapour return lines or pressure equalizing lines, are commonly used in smaller pipelines for transport of LNG, LPG and compressed natural gas (CNG). Pressure drop detectors are installed to signal any leaks in pipelines, and excess flow valves or other flow limiting devices are used to minimize the rate of flow in case of a pipeline leak. Storage vessels and reservoirs may be isolated from main pipelines by manually operated or remote control valves or fusible link valves.
- Pipeline product storage. Crude and petroleum product pipeline terminals have breakout storage tanks to which shipments may be diverted, where they are held until required by a refinery, terminal or user (see figure 1). Other tanks at pipeline pumping stations contain fuel for operating diesel-driven pump motors or for running electrical generators. Because gas fields produce continuously and gas pipelines operate continuously, during times of reduced demand, such as summertime, liquefied natural and petroleum gases are stored underground in natural caverns or salt domes until needed.
- Pipeline cleaning. Pipelines are cleaned on a scheduled basis or as necessary in order to continue flow by reducing friction and maintaining as large a diameter interior as possible. A special cleaning device, called a pig or go-devil, is placed into the pipeline and pushed along by the flow of oil from one pumping station to the next. As the pig passes through the pipeline it scrapes off any dirt, wax or other deposits which have built up inside the pipeline walls. When it reaches a pumping station, the pig is removed, cleaned and reinserted into the pipeline to travel to the next station.
- Communications. It is important that there be communication and agreement concerning schedules, pumping rates and pressures and emergency procedures between pipeline stations and operators and those shipping and receiving crude oil, gas and petroleum products. Some pipeline companies have private telephone systems which transmit the signal along the pipeline, while others use radios or public telephones. Many pipelines use ultra-high-frequency microwave transmitter systems for computer communications between control centres and pumping stations.
- Petroleum product shipment. Petroleum products may be shipped a number of different ways on pipelines. A company operating a refinery may blend a specific grade of its own gasoline with appropriate additives (additize) and ship a batch through a pipeline directly to its own terminal for distribution to its customers. Another method is for a refinery to produce a batch of gasoline, called a frangible or specification product, which is blended to meet a common carrier pipeline company’s product specifications. The gasoline is placed into the pipeline for delivery to any company’s terminals which are connected to the pipeline system. In a third method, products are shipped by companies to each other’s terminals and exchanged in order to avoid extra transportation and handling. Frangible and exchange products are usually blended and additized at the terminal which receives the product from the pipeline, to meet the specific requirements of each company operating from the terminal. Finally, some products are delivered by pipeline from terminals and refineries direct to large commercial consumers—jet fuel to airports, gas to gas distribution companies and fuel oil to electric generating plants.
- Product receipt and delivery. Pipeline operators and terminal operators should jointly establish programmes to provide for the safe receipt and transfer of products and to coordinate actions in case an emergency occurs on the pipeline or at the terminal during shipment which requires shutdown or diversion of product.
Figure 1. A terminal operator transfers product the Pasagoula Refinery into holding tanks in the Deraville Terminal near Atlanta, Georgia, US.
American Petroleum Institute
Instructions for receiving pipeline deliveries should include verification of the availability of the storage tanks to hold the shipment, opening and aligning tank and terminal valves in anticipation of delivery, checking to assure that the proper tank is receiving product immediately after the start of delivery, conducting required sampling and testing of batches at the start of delivery, performing batch changes and tank switches as required, monitoring receipts to assure that overfills do not occur and maintaining communications between the pipeline and the terminal. The use of written communications between terminal workers, especially when shift changes occur during product transfer, should be considered.
Batch shipments and interface
Although pipelines originally were used to move only crude oil, they evolved into carrying all types and different grades of liquid petroleum products. Because petroleum products are transported in pipelines by batches, in succession, there is commingling or mixing of the products at the interfaces. The product intermix is controlled by one of three methods: downgrading (derating), using liquid and solid spacers for separation or reprocessing the intermix. Radioactive tracers, colour dyes and spacers may be placed into the pipeline to identify where the interfaces occur. Radioactive sensors, visual observation or gravity tests are conducted at the receiving facility to identify different pipeline batches.
Petroleum products are normally transported through pipelines in batch sequences with compatible crude oils or products adjoining one another. One method of maintaining product quality and integrity, downgrading or derating, is accomplished by lowering the interface between the two batches to the level of the least affected product. For example, a batch of high-octane premium gasoline is typically shipped immediately before or after a batch of lower-octane regular gasoline. The small quantity of the two products which has intermixed will be downgraded to the lower octane rating regular gasoline. When shipping gasoline before or after diesel fuel, a small amount of diesel interface is allowed to blend into the gasoline, rather than blending gasoline into the diesel fuel, which could lower its flashpoint. Batch interfaces are typically detected by visual observation, gravitometers or sampling.
Liquid and solid spacers or cleaning pigs may be used to physically separate and identify different batches of products. The solid spacers are detected by a radioactive signal and diverted from the pipeline into a special receiver at the terminal when the batch changes from one product to another. Liquid separators may be water or another product that does not commingle with either of the batches it is separating and is later removed and reprocessed. Kerosene, which is downgraded (derated) to another product in storage or is recycled, can also be used to separate batches.
A third method of controlling the interface, often used at the refinery ends of pipelines, is to return the interface to be reprocessed. Products and interfaces which have been contaminated with water may also be returned for reprocessing.
Environmental protection
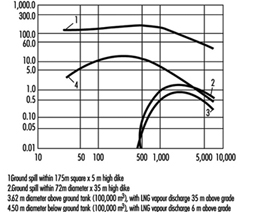
Because of the large volumes of products which are transported by pipelines on a continuous basis, there is opportunity for environmental damage from releases. Depending on company and regulatory safety requirements and the pipeline’s construction, location, weather, accessibility and operation, a considerable amount of product may be released should a break in the line or leak occur. Pipeline operators should have emergency response and spill contingency plans prepared and have containment and clean-up materials, personnel and equipment available or on call. Simple field solutions such as building earth dykes and drainage ditches can be quickly implemented by trained operators to contain and divert spilled product.
Maintaining pipelines and worker health and safety
The first pipelines were made of cast iron. Modern trunk pipelines are constructed of welded, high-strength steel, which can withstand high pressures. Pipe walls are periodically tested for thickness to determine whether internal corrosion or deposits have occurred. Welds are checked visually and with gamma radiation to assure that no defects are present.
Plastic pipe may be used for low-pressure, small-diameter flow lines and gathering lines in gas and crude-oil-producing fields, since plastic is light in weight and easy to handle, assemble and move.
When a pipeline is separated by cutting, spreading flanges, removing a valve or opening the line, an electrostatic arc may be created by impressed cathodic protection voltage, corrosion, sacrificial anodes, nearby high-voltage power lines or stray ground currents. This should be minimized by grounding (earthing) the pipe, de-energizing the cathodic rectifiers closest to both sides of the separation and connecting a bonding cable to each side of the piping prior to starting work. As additional pipeline sections, valves and so on are added to an existing line, or during construction, they should first be bonded to the pipelines in place.
Work on pipelines should cease during electrical storms. Equipment used to lift and place pipe should not be operated within 3 m of high-voltage electric lines. Any vehicles or equipment working in the vicinity of high-voltage lines should have trailing grounding straps attached to the frames. Temporary metal buildings should also be grounded.
Pipelines are specially coated and wrapped to prevent corrosion. Cathodic electrical protection may also be required. After the pipeline sections are coated and insulated, they are joined by special clamps connected to metallic anodes. The pipeline is subjected to a grounded source of direct current of sufficient capacity so that the pipeline acts as a cathode and does not corrode.
All pipeline sections are hydrostatically tested prior to entering gas or liquid hydrocarbon service and, depending on regulatory and company requirements, at regular intervals during the life of the pipeline. Air must be eliminated from pipelines prior to hydrostatic testing, and hydrostatic pressure built up and reduced at safe rates. Pipelines are regularly patrolled, usually by aerial surveillance, to visually detect leaks, or monitored from the control centre to detect a drop in flow rate or pressure, which would signify that a break in the pipeline has occurred.
Pipeline systems are provided with warning and signalling systems to alert operators so they may take corrective action in an emergency. Pipelines may have automatic shutdown systems which activate emergency pressure valves upon sensing increased or reduced pipeline pressure. Manually or automatically operated isolation valves are typically located at strategic intervals along pipelines, such as at pumping stations and at both sides of river crossings.
An important consideration when operating pipelines is to provide a means of warning contractors and others who may be working or conducting excavations along the pipeline route, so that the pipeline is not inadvertently ruptured, breached or punctured, resulting in a vapour or gas explosion and fire. This is usually done by regulations which require construction permits or by pipeline companies and associations providing a central number which contractors can call prior to excavation.
Because crude oil and flammable petroleum products are transported in pipelines, the possibility exists for fire or explosion in case of a line break or release of vapour or liquid. Pressure should be reduced to a safe level before working on high-pressure pipelines. Combustible gas testing should be conducted and a permit issued prior to repair or maintenance involving hot work or hot tapping on pipelines. The pipeline should be cleared of flammable liquids and vapours or gas prior to starting work. If a pipeline cannot be cleared and an approved plug is used, safe work procedures should be established and followed by qualified workers. The line should be vented a safe distance from the hot work area to relieve any build-up of pressure behind the plug.
Proper safety procedures should be established and followed by qualified workers when hot tapping pipelines. If welding or hot tapping is conducted in an area where a spill or leak has occurred, the outside of the pipe should be cleaned of liquid, and contaminated soil should be removed or covered to prevent ignition.
It is very important to notify operators at the nearest pumping stations on each side of the operating pipeline where maintenance or repair is to be performed, in case shutdown is required. When crude oil or gas is being pumped into pipelines by producers, the pipeline operators must provide specific instructions to the producers as to actions to take during repair, maintenance or in an emergency. For example, prior to tie-in of production tanks and lines to pipelines, all gate valves and bleeders for the tanks and lines involved in the tie-in should be closed and locked or sealed until the operation is completed.
Normal safety precautions concerning pipe and materials handling, toxic and hazardous exposures, welding and excavation apply during pipeline construction. Workers clearing right-of-way should protect themselves from climatic conditions; poisonous plants, insects and snakes; falling trees and rocks; and so on. Excavations and trenches should be sloped or shored to prevent collapse during underground pipeline construction or repair (see the article “Trenching” in the chapter Construction). Workers should follow safe work practices when opening and de-energizing electrical transformers and switches.
Pipeline operating and maintenance personnel often work alone and are responsible for long stretches of pipeline. Atmospheric testing and the use of personal and respiratory protective equipment is needed to determine oxygen and flammable vapour levels and protect against toxic exposures to hydrogen sulphide and benzene when gauging tanks, opening lines, cleaning spills, sampling and testing, shipping, receiving and performing other pipeline activities. Workers should wear dosimeters or film badges and avoid exposure when working with density gauges, source holders or other radioactive materials. The use of personal and respiratory protective equipment should be considered for exposure to burns from the hot protective tar used in pipe-coating operations and from toxic vapours which contain polynuclear aromatic hydrocarbons.
Marine Tankers and Barges
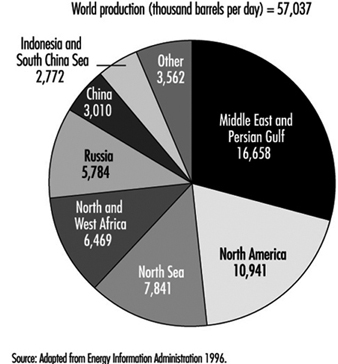
The majority of the world’s crude oil is transported by tankers from producing areas such as the Middle East and Africa to refineries in consumer areas such as Europe, Japan and the United States. Oil products were originally transported in large barrels on cargo ships. The first tanker ship, which was built in 1886, carried about 2,300 SDWT (2,240 pounds per ton) of oil. Today’s supertankers can be over 300 m long and carry almost 200 times as much oil (see figure 2). Gathering and feeder pipelines often end at marine terminals or offshore platform loading facilities, where the crude oil is loaded into tankers or barges for transport to crude trunk pipelines or refineries. Petroleum products also are transported from refineries to distribution terminals by tanker and barge. After delivering their cargoes, the vessels return in ballast to loading facilities to repeat the sequence.
Figure 2. SS Paul L. Fahrney oil tanker.
American Petroleum Institute
Liquefied natural gas is shipped as a cryogenic gas in specialized marine vessels with heavily insulated compartments or reservoirs (see figure 3). At the delivery port, the LNG is off-loaded to storage facilities or regasification plants. Liquefied petroleum gas may be shipped both as a liquid in uninsulated marine vessels and barges and as a cryogenic in insulated marine vessels. Additionally, LPG in containers (bottled gas) may be shipped as cargo on marine vessels and barges.
Figure 3. LNG Leo tanker loading at Arun, Sumatra, Indonesia.
American Petroleum Institute
LPG and LNG marine vessels
The three types of marine vessels used for transport of LPG and LNG are:
- vessels with reservoirs pressurized up to 2 mPa (LPG only)
- vessels with heat-insulated reservoirs and a reduced pressure of 0.3 to 0.6 mPa (LPG only)
- cryogenic vessels with heat-insulated reservoirs pressurized close to atmospheric pressure (LPG and LNG).
Shipment of LHGs on marine vessels requires constant safety awareness. Transfer hoses must be suitable for the correct temperatures and pressures of the LHGs being handled. To prevent a flammable mixture of gas vapour and air, inert gas (nitrogen) blanketing is provided around reservoirs, and the area is continually monitored to detect leaks. Before loading, storage reservoirs should be inspected to ensure that they are free of contaminants. If reservoirs contain inert gas or air, they should be purged with LHG vapour prior to loading the LHG. Reservoirs should be constantly inspected to ensure integrity, and safety valves should be installed to relieve the LHG vapour generated at maximum heat load. Marine vessels are provided with fire suppression systems and have comprehensive emergency response procedures in place.
Crude oil and petroleum products marine vessels
Oil tankers and barges are vessels designed with the engines and quarters at the rear of the vessel and the remainder of the vessel divided into special compartments (tanks) to carry crude oil and liquid petroleum products in bulk. Cargo pumps are located in pump rooms, and forced ventilation and inerting systems are provided to reduce the risk of fires and explosions in pump rooms and cargo compartments. Modern oil tankers and barges are built with double hulls and other protective and safety features required by the United States Oil Pollution Act of 1990 and the International Maritime Organization (IMO) tanker safety standards. Some new ship designs extend double hulls up the sides of the tankers to provide additional protection. Generally, large tankers carry crude oil and small tankers and barges carry petroleum products.
- Supertankers. Ultra-large and very large crude carriers (ULCCs and VLCCs) are restricted by their size and draft to specific routes of travel. ULCCs are vessels whose capacity is over 300,000 SDWTs, and VLCCs have capacities ranging from 160,000 to 300,000 SDWTs. Most large crude carriers are not owned by oil companies, but are chartered from transportation companies which specialize in operating these super-sized vessels.
- Oil tankers. Oil tankers are smaller than VLCCs, and, in addition to ocean travel, they can navigate restricted passages such as the Suez and Panama Canals, shallow coastal waters and estuaries. Large oil tankers, which range from 25,000 to 160,000 SDWTs, usually carry crude oil or heavy residual products. Smaller oil tankers, under 25,000 SDWT, usually carry gasoline, fuel oils and lubricants.
- Barges. Barges operate mainly in coastal and inland waterways and rivers, alone or in groups of two or more, and are either self-propelled or moved by tugboat. They may carry crude oil to refineries, but more often are used as an inexpensive means of transporting petroleum products from refineries to distribution terminals. Barges are also used to off-load cargo from tankers offshore whose draft or size does not allow them to come to the dock.
Barge and ship loading and unloading
Vessel-to-shore procedures, safety checklists and guidelines should be established and agreed upon by terminal and marine vessel operators. The International Safety Guide for Oil Tankers and Terminals (International Chamber of Shipping 1978) contains information and samples of checklists, guidelines, permits and other procedures covering safe operations when loading or unloading vessels, which may be used by vessel and terminal operators.
Although marine vessels sit in water and are thereby intrinsically grounded, there is a need to provide protection from static electricity which can build up during loading or unloading. This is accomplished by bonding or connecting metal objects on the dock or loading/unloading apparatus to the metal of the vessel. Bonding is also accomplished by use of conductive loading hose or piping. An electrostatic spark of ignitable intensity may also be generated when lowering equipment, thermometers or gauging devices into compartments immediately after loading; enough time must be allowed for the static charge to dissipate.
Ship-to-shore electric currents, which are different from static electricity, may be generated by cathodic protection of the vessel’s hull or dock, or by galvanic potential differences between the vessel and the shore. These currents also build up in metal loading/unloading apparatus. Insulating flanges may be installed within the length of the loading arm and at the point where flexible hoses connect to the shore pipeline system. When the connections are broken, there is no opportunity for a spark to jump from one metal surface to another.
All vessels and terminals need mutually agreed upon emergency response procedures in case of a fire or release of product, vapour or toxic gas. These must cover emergency operations, stopping product flow and emergency removal of a vessel from the dock. The plans should consider communications, fire-fighting, vapour cloud mitigation, mutual aid, rescue, clean-up and remediation measures.
Fire protection portable equipment and fixed systems should be in accord with government and company requirements and appropriate to the size, function, exposure potential and value of the dock and wharf facilities. The International Safety Guide for Oil Tankers and Terminals (International Chamber of Shipping 1978) contains a sample fire notice which may be used as a guide by terminals for dock fire prevention.
Marine vessel health and safety
In addition to the usual maritime working hazards, transporting crude oil and flammable liquids by marine vessel creates a number of special health, safety and fire prevention situations. These include surging and expansion of liquid cargo, flammable vapour hazards during transport and when loading and unloading, possibility of pyrophoric ignition, toxic exposures to materials such as hydrogen sulphide and benzene and safety considerations when venting, flushing and cleaning compart-ments. The economics of operating modern tankers requires them to be at sea for extended periods of time with only short intervals in port to load or unload cargo. This, together with the fact that tankers are highly automated, creates unique mental and physical demands on the few crew members used to operate the vessels.
Fire and explosion protection
Emergency plans and procedures should be developed and implemented that are appropriate for the type of cargo on board and other potential hazards. Fire-fighting equipment must be supplied. Response team members who have shipboard fire-fighting, rescue and spill clean-up responsibilities should be trained, drilled and equipped to handle potential emergencies. Water, foam, dry chemicals, halon, carbon dioxide and steam are used as cooling, inhibiting and smothering fire-fighting agents aboard marine vessels, although halon is being phased out due to environmental concerns. The requirements for vessel fire-fighting equipment and systems are established by the country under whose flag the vessel sails and by company policy, but usually follow the recommendations of the 1974 International Convention for the Safety of Life at Sea (SOLAS).
Strict control of flames or naked lights, lighted smoking materials and other sources of ignition, such as welding or grinding sparks, electrical equipment and unprotected light bulbs, is required on vessels at all times to reduce the risk of fire and explosion. Prior to conducting hot work on board marine vessels, the area should be examined and tested to assure that conditions are safe, and permits should be issued for each specific task allowed.
One method of preventing explosions and fires in the vapour space of cargo compartments is to maintain the level of oxygen below 11% by making the atmosphere inert with a noncombustible gas. Sources for inert gas are exhaust gases from the vessel’s boilers or an independent gas generator or a gas turbine fitted with an afterburner. The 1974 SOLAS Convention implies that vessels carrying cargo with flashpoints below 60°C should have compartments fitted with inert systems. Vessels using inert gas systems should maintain cargo compartments in non-flammable conditions at all times. Inert gas compartments should be constantly monitored to assure safe conditions and should not be allowed to become flammable, because of the danger of ignition from pyrophoric deposits.
Confined spaces
Confined spaces on marine vessels, such as cargo compartments, paint lockers, pump rooms, fuel tanks and spaces between double hulls, must be treated the same as any confined space for entry, hot work and cold work. Tests for oxygen content, flammable vapours and toxic substances, in that order, must be conducted prior to entering confined spaces. A permit system should be established and followed for all confined space entry, safe (cold) work and hot work, which indicates safe exposure levels and required personal and respiratory protective equipment. In waters of the United States, these tests may be conducted by qualified individuals called “marine chemists”.
Compartments on marine vessels such as cargo tanks and pump rooms are confined spaces; when cleaning those which have been made inert or have flammable vapour, toxic or unknown atmospheres, they should be tested, and special safety and respiratory protection procedures should be followed. After crude oil has been unloaded, a small amount of residue, called clingage, remains on the interior surfaces of the compartments, which may then be washed and filled with water for ballast. One method of reducing the amount of residue is to install fixed equipment which removes up to 80% of the clingage by washing down the sides of inerted compartments with crude oil during unloading.
Pumps, valves and equipment
A work permit should be issued and safe work procedures followed, such as bonding, draining and vapour freeing, flammable vapour and toxic exposure testing, and providing stand-by fire protection equipment when operations, maintenance or repair requires opening cargo pumps, lines, valves or equipment on board marine vessels.
Toxic exposures
There is an opportunity for vented gases such as flue gas or hydrogen sulphide to reach the decks of vessels, even from specially designed vent systems. Testing should be continuously conducted to determine inert gas levels on all vessels and hydrogen sulphide levels on vessels which contain or previously carried sour crude oil or residual fuel. Tests should be conducted for benzene exposure on vessels carrying crude oil and gasoline. Inert gas scrubber effluent water and condensate water is acidic and corrosive; PPE should be used when contact is possible.
Environmental protection
Marine vessels and terminals should establish procedures and provide equipment to protect the environment from spills on water and land, and from releases of vapour to the air. The use of large vapour recovery systems at marine terminals is growing. Care must be taken to comply with air pollution requirements when vessels vent compartments and enclosed spaces. Emergency response procedures should be established, and equipment and trained personnel should be available to respond to spills and releases of crude oil and flammable and combustible liquids. A responsible person should be designated to ensure that notifications are made to both the company and the appropriate authorities should a reportable spill or release occur.
In the past, the oil-contaminated ballast water and tank washings were flushed out of the compartments at sea. In 1973, the International Convention for Prevention of Pollution from Ships established requirements that before the water is discharged at sea, the oily residue must be separated and retained on board for eventual onshore processing. Modern tankers have segregated ballast systems, with different lines, pumps and tanks than those used for cargo (in accordance with international recommen-dations), so that there is no possibility of contamination. Older vessels still carry ballast in cargo tanks, so special procedures, such as pumping oily water into designated onshore tanks and processing facilities, must be followed when discharging ballast in order to prevent pollution.
Motor Vehicle and Railroad Transport of Petroleum Products
Crude oil and petroleum products were initially transported by horse-drawn tank wagons, then by railroad tank cars and finally by motor vehicles. Following receipt at terminals from marine vessels or pipelines, bulk liquid petroleum products are delivered by non-pressure tank trucks or rail tank cars directly to service stations and consumers or to smaller terminals, called bulk plants, for redistribution. LPG, gasoline anti-knock compounds, hydrofluoric acid and many other products, chemicals and additives used in the oil and gas industry are transported in pressure tank cars and tank trucks. Crude oil may also be transported by tank truck from small producing wells to gathering tanks, and by tank truck and railroad tank car from storage tanks to refineries or main pipelines. Packaged petroleum products in bulk bins or drums and pallets and cases of smaller containers are carried by package truck or railroad box car.
Government regulations
Transportation of petroleum products by motor vehicle or railroad tank car is regulated by government agencies throughout most of the world. Agencies such as the US DOT and the Canadian Transport Commission (CTC) have established regulations governing the design, construction, safety devices, testing, preventive maintenance, inspection and operation of tank trucks and tank cars. Regulations governing railroad tank car and tank truck operations typically include tank pressure and pressure relief device testing and certification before being placed into initial service and at regular intervals thereafter. The Association of American Railroads and the National Fire Protection Association (NFPA) are typical of organizations which publish specifications and requirements for the safe operation of tank cars and tank trucks. Most governments have regulations or adhere to United Nations Conventions which require the identification of and information concerning hazardous materials and petroleum products which are shipped in bulk or in containers. Railroad tank cars, tank trucks and package trucks are placarded to identify any hazardous products being transported and to provide emergency response information.
Railroad tank cars
Railroad tank cars are constructed of carbon steel or aluminium and may be pressurized or unpressurized. Modern tank cars can hold up to 171,000 l of compressed gas at pressures up to 600 psi (1.6 to 1.8 mPa). Non-pressure tank cars have evolved from small wooden tank cars of the late 1800s to jumbo tank cars which transport as much as 1.31 million litres of product at pressures up to 100 psi (0.6 mPa). Non-pressure tank cars may be individual units with one or multiple compartments or a string of interconnected tank cars, called a tank train. Tank cars are loaded individually, and entire tank trains can be loaded and unloaded from a single point. Both pressure and non-pressure tank cars may be heated, cooled, insulated and thermally protected against fire, depending on their service and the products transported.
All railroad tank cars have top- or bottom-liquid or vapour valves for loading and unloading and hatch entries for cleaning. They are also equipped with devices intended to prevent the increase of internal pressure when exposed to abnormal con-ditions. These devices include safety relief valves held in place by a spring which can open to relieve pressure and then close; safety vents with rupture discs that burst open to relieve pressure but cannot reclose; or a combination of the two devices. A vacuum relief valve is provided for non-pressure tank cars to prevent vacuum formation when unloading from the bottom. Both pressure and non-pressure tank cars have protective housings on top surrounding the loading connections, sample lines, thermometer wells and gauging devices. Platforms for loaders may or may not be provided on top of cars. Older non-pressure tank cars may have one or more expansion domes. Fittings are provided on the bottom of tank cars for unloading or cleaning. Head shields are provided on the ends of tank cars to prevent puncture of the shell by the coupler of another car during derailments.
LNG is shipped as a cryogenic gas in insulated tank truck and rail pressure tank cars. Pressure tank trucks and rail tank cars for LNG transport have a stainless steel inner reservoir suspended in an outer reservoir of carbon steel. The annular space is a vacuum filled with insulation to maintain low temperatures during shipment. To prevent gas from igniting back to the tanks, they are equipped with two independent, remotely controlled fail-safe emergency shut-off valves on the filling and discharge lines and have gauges on both the inside and outside reservoirs.
LPG is transported on land in specially designed rail tank cars (up to 130 m3 capacity) or tank trucks (up to 40 m3 capacity). Tank trucks and rail tank cars for LPG transport are typically uninsulated steel cylinders with spherical bottoms, equipped with gauges, thermometers, two safety relief valves, a gas level meter and maximum fill indicator and baffles.
Rail tank cars transporting LNG or LPG should not be overloaded, since they may sit on a siding for some period of time and be exposed to high ambient temperatures, which could cause overpressure and venting. Bond wires and grounding cables are provided at rail and tank truck loading racks to help neutralize and dissipate static electricity. They should be connected before operations commence and not disconnected until operations are complete and all valves are closed. Truck and rail loading facilities are typically protected by fire water spray or mist systems and fire extinguishers.
Tank trucks
Petroleum products and crude oil tank trucks are typically constructed of carbon steel, aluminium or a plasticized fibreglass material, and vary in size from 1,900-l tank wagons to jumbo 53,200-l tankers. The capacity of tank trucks is governed by regulatory agencies, and usually is dependent upon highway and bridge capacity limitations and the allowable weight per axle or total amount of product allowed.
There are pressurized and non-pressurized tank trucks, which may be non-insulated or insulated depending on their service and the products transported. Pressurized tank trucks are usually single compartment, and non-pressurized tank trucks may have single or multiple compartments. Regardless of the number of compartments on a tank truck, each compartment must be treated individually, with its own loading, unloading and safety-relief devices. Compartments may be separated by single or double walls. Regulations may require that incompatible products and flammable and combustible liquids carried in different compartments on the same vehicle be separated by double walls. When pressure testing compartments, the space between the walls should also be tested for liquid or vapour.
Tank trucks have either hatches which open for top loading, valves for closed top- or bottom-loading and unloading, or both. All compartments have hatch entries for cleaning and are equipped with safety relief devices to mitigate internal pressure when exposed to abnormal conditions. These devices include safety relief valves held in place by a spring which can open to relieve pressure and then close, hatches on non-pressure tanks which pop open if the relief valves fail and rupture discs on pressurized tank trucks. A vacuum relief valve is provided for each non-pressurized tank truck compartment to prevent vacuum when unloading from the bottom. Non-pressurized tank trucks have railings on top to protect the hatches, relief valves and vapour recovery system in case of a rollover. Tank trucks are usually equipped with breakaway, self-closing devices installed on compartment bottom loading and unloading pipes and fittings to prevent spills in case of damage in a rollover or collision.
Rail tank car and tank truck loading and unloading
While railroad tank cars are almost always loaded and unloaded by workers assigned to these specific duties, tank trucks may be loaded and unloaded by either loaders or drivers. Tank cars and tank trucks are loaded at facilities called loading racks, and may be top loaded through open hatches or closed connections, bottom loaded through closed connections, or a combination of both.
Loading
Workers who load and unload crude oil, LPG, petroleum products, and acids and additives used in the oil and gas industry, should have a basic understanding of the characteristics of the products handled, their hazards and exposures and the operating procedures and work practices needed to perform the job safely. Many government agencies and companies require the use and completion of inspection forms upon receipt and shipment and prior to loading and unloading railroad tank cars and tank trucks. Tank trucks and railroad tank cars may be loaded through open hatches on the top or through fittings and valves at the top or bottom of each tank or compartment. Closed connections are required when pressure loading and where vapour recovery systems are provided. If loading systems do not activate for any reason (such as improper operation of the vapour recovery system or a fault in the grounding or bonding system), by-pass should not be attempted without approval. All hatches should be closed and securely latched during transit.
Workers should follow safe work practices to avoid slips and falls when top loading. If loading controls use pre-set meters, loaders must be careful to load the correct products into the assigned tanks and compartments. All compartment hatches should be shut when bottom loading, and when top loading, only the compartment being loaded should be open. When top loading, splash loading should be avoided by placing the loading tube or hose close to the bottom of the compartment and starting to load slowly until the opening is submerged. During manual top loading operations, loaders should remain in attendance, not tie down the loading shut-off (deadman) control and not overfill the compartment. Loaders should avoid exposures to product and vapour by standing upwind and averting the head when top loading through open hatches and by wearing protective equipment when handling additives, obtaining samples and draining hoses. Loaders should be aware of and follow prescribed response actions in case of a hose or line rupture, spill, release, fire or other emergency.
Unloading and delivery
When unloading tank cars and tank trucks, it is important first to assure that each product is unloaded into the proper designated storage tank and that the tank has sufficient capacity to hold all of the product being delivered. Although valves, fill pipes, lines and fill covers should be colour coded or otherwise marked to identify the product contained, the driver should still be responsible for product quality during delivery. Any misdelivery of product, mixing or contamination should be immediately reported to the recipient and to the company to prevent serious consequences. When drivers or operators are required to additize products or obtain samples from storage tanks following delivery to assure product quality or for any other reason, all safety and health provisions specific to the exposure should be followed. Persons engaged in delivery and unloading operations should remain in the vicinity at all times and know what to do in an emergency, including notification, stopping product flow, cleaning spills and when to leave the area.
Pressurized tanks may be unloaded by compressor or pump, and unpressurized tanks by gravity, vehicle pump or recipient pump. Tank trucks and tank cars which carry lubrication or industrial oils, additives and acids are sometimes unloaded by pressurizing the tank with an inert gas such as nitrogen. Tank cars or tank trucks may need to be heated using steam or electric coils in order to unload heavy crude oils, viscous products and waxes. All of these activities have inherent dangers and exposures. Where required by regulation, unloading should not commence until the vapour recovery hoses have been connected between the delivery tank and the storage tank. When delivering petroleum products to residences, farms and commercial accounts, drivers should gauge any tank which is not equipped with a vent alarm in order to prevent an overfill.
Loading-rack fire protection
Fires and explosions at top and bottom tank car and tank truck loading racks may occur from causes such as electrostatic build-up and incendiary spark discharge in a flammable atmosphere, unauthorized hot work, flashback from a vapour recovery unit, smoking or other unsafe practices.
Sources of ignition, such as smoking, running internal combustion engines and hot work activity, should be controlled at the loading rack at all times, and particularly during loading or other operations when a spill or release may occur. Loading racks may be equipped with portable fire extinguishers and manually or automatically operated foam, water or dry chemical fire extinguishing systems. If vapour recovery systems are in use, flame arrestors should be provided to prevent flashback from the recovery unit to the loading rack.
Drainage should be provided at loading racks to divert product spills away from the loader, tank truck or tank car and the loading rack pad. Drains should be provided with fire traps to prevent migration of flames and vapours through sewer systems. Other loading-rack safety considerations include emergency shut-down controls placed at loading spots and other strategic locations in the terminal and automatic pressure-sensing valves which stop product flow to the rack in case of a leak in the product lines. Some companies have installed automatic brake lock systems on their tank truck fill connections, which lock the brakes and will not allow the truck to be moved from the rack until the fill lines have been disconnected.
Electrostatic ignition hazards
Some products such as intermediate distillates and low-vapour-pressure fuels and solvents tend to accumulate electrostatic charges. When loading tank cars and tank trucks, there is always an opportunity for electrostatic charges to be generated by friction as product goes through lines and filters and by splash loading. This can be mitigated by designing loading racks to allow for relaxation time in piping downstream from pumps and filters. Compartments should be checked to assure that they do not contain any unbonded or floating objects which could act as static accumulators. Bottom loaded compartments may be provided with internal cables to help dissipate electrostatic charges. Sample containers, thermometers or other items should not be lowered into compartments until a waiting period of at least 1 minute has elapsed, to allow any electrostatic charge which has accumulated in the product to dissipate.
Bonding and grounding are important considerations in dissipating electrostatic charges which build up during loading operations. By keeping the fill pipe in contact with the metal side of the hatch when top loading, and through the use of metal loading arms or conductive hose when loading through closed connections, the tank truck or tank car is bonded to the loading rack, maintaining the same electrical charge between the objects so that a spark is not created when the loading tube or hose is removed. The tank car or tank truck may also be bonded to the loading rack by use of a bonding cable, which carries any accumulated charge from a terminal on the tank to the rack, where it is then grounded by a grounding cable and rod. Similar bonding precautions are needed when unloading from tank cars and tank trucks. Some loading racks are provided with electronic connectors and sensors which will not allow loading pumps to activate until a positive bond is achieved.
During cleaning, maintenance or repair, pressurized LPG tank cars or tank trucks are usually opened to the atmosphere, allowing air to enter the tank. In order to prevent combustion from electrostatic charges when loading these cars for the first time after such activities, it is necessary to reduce the oxygen level below 9.5% by blanketing the tank with inert gas, such as nitrogen. Precautions are needed to prevent liquid nitrogen from entering the tank if the nitrogen is provided from portable containers.
Switch loading
Switch loading occurs when intermediate- or low-vapour-pressure products such as diesel fuel or fuel oil are loaded into a tank car or tank truck compartment which previously contained a flammable product such as gasoline. The electrostatic charge generated during loading can discharge in an atmosphere which is within the flammable range, with a resultant explosion and fire. This hazard can be controlled when top loading by lowering the fill tube to the bottom of the compartment and loading slowly until the end of the tube is submerged to avoid splash loading or agitation. Metal to metal contact should be maintained during loading in order to provide a positive bond between the loading tube and the tank hatchway. When bottom loading, initial slow fill or splash deflectors are used to reduce static build-up. Prior to switch loading, tanks which cannot be drained dry may be flushed out with a small amount of the product to be loaded, to remove any flammable residue in sumps, lines, valves and onboard pumps.
Shipping products by rail box cars and package vans
Petroleum products are shipped by motor truck package vans and railroad box cars in metal, fibre and plastic containers of various sizes, from 55-gallon (209-l) drums to 5-gallon (19-l) pails and from 2-1/2-gallon (9.5-l) to 1-quart (.95-l) containers, in corrugated boxes, usually on pallets. Many industrial and commercial petroleum products are shipped in large metal, plastic or combination intermediate bulk containers ranging in size from 380 to over 2,660 l capacity. LPG is shipped in large and small pressure containers. In addition, samples of crude oil, finished products and used products are shipped by mail or express freight carrier to laboratories for assay and analysis.
All of these products, containers and packages have to be handled in accordance with government regulations for hazardous chemicals, flammable and combustible liquids and toxic materials. This requires the use of hazardous materials manifests, shipping documents, permits, receipts and other regulatory requirements, such as marking the outsides of packages, containers, motor trucks and box cars with proper identification and a hazard warning label. Proper utilization of tank trucks and tank cars is important to the petroleum industry. Because storage capacity is finite, delivery schedules need to be met, from the delivery of crude oil to keep refineries running to the delivery of gasoline to service stations, and from the delivery of lubricants to commercial and industrial accounts to the delivery of heating oil to homes.
LPG is supplied to consumers by bulk tank trucks which pump directly into smaller onsite storage tanks, both above ground and below ground (e.g., service stations, farms, commercial and industrial consumers). LPG is also delivered to consumers by truck or van in containers (gas cylinders or bottles). LNG is delivered in special cryogenic containers which have an inner fuel tank surrounded by insulation and an outer shell. Similar containers are provided for vehicles and appliances which use LNG as a fuel. Compressed natural gas is normally delivered in conventional compressed gas cylinders, such as those used on industrial lift trucks.
In addition to the normal safety and health precautions required in rail car and package trucking operations, such as moving and handling heavy objects and operating industrial trucks, workers should be familiar with the hazards of the products they are handling and delivering, and know what to do in case of a spill, release or other emergency. For example, intermediate bulk containers and drums should not be dropped out of box cars or from the tailgates of trucks onto the ground. Both companies and government agencies have established special regulations and requirements for drivers and operators who are involved in the transport and delivery of flammable and hazardous petroleum products.
Tank truck and package van drivers often work alone and may have to travel great distances for a number of days to deliver their loads. They work both day and night and in all sorts of weather conditions. Manoeuvring super-sized tank trucks into service stations and customer locations without hitting parked vehicles or fixed objects requires patience, skill and experience. Drivers should have the physical and mental characteristics required for this work.
Driving tank trucks is different from driving package vans in that the liquid product tends to shift forward as the truck stops, backwards as the truck accelerates and from side to side as the truck turns. Tank truck compartments should be fitted with baffles which restrict the movement of product during transport. Considerable skill is required by drivers to overcome the inertia created by this phenomenon, called “mass in motion”. Occasionally, tank truck drivers are required to pump out storage tanks. This activity requires special equipment, including suction hose and transfer pumps, and safety precautions, such as bonding and grounding to dissipate electrostatic build-up and to prevent any release of vapours or liquids.
Motor vehicle and rail car emergency response
Drivers and operators should be familiar with notification requirements and emergency response actions in case of a fire or a release of product, gas or vapour. Product identification and hazard warning placards in compliance with industry, association or national marking standards are posted on trucks and rail cars to allow emergency responders to determine the precautions needed in case of a spill or release of vapour, gas or product. Motor vehicle drivers and train operators may also be required to carry material safety data sheets (MSDSs) or other documentation describing the hazards and precautions for handling the products being transported. Some companies or government agencies require that vehicles transporting flammable liquids or hazardous materials carry first aid kits, fire extinguishers, spill clean-up materials and portable hazard warning devices or signals to alert motorists if the vehicle is stopped alongside a highway.
Special equipment and techniques are required if a tank car or tank truck needs to be emptied of product as the result of an accident or rollover. Removal of product through fixed piping and valves or by using special knock-out plates on tank truck hatches is preferred; however, under certain conditions holes may be drilled in tanks using prescribed safe work procedures. Regardless of the method of removal, tanks should be grounded and a bond connection provided between the tank being emptied and the receiving tank.
Cleaning tank cars and tank trucks
Entering a tank car or tank truck compartment for inspection, cleaning, maintenance or repair is a hazardous activity requiring that all ventilation, testing, gas freeing and other confined-space entry and permit system requirements be followed in order to assure a safe operation. Cleaning tank cars and tank trucks is not any different from cleaning petroleum-product storage tanks, and all the same safety and health exposure precautions and procedures apply. Tank cars and tank trucks may contain residue of flammable, hazardous or toxic materials in sumps and unloading piping, or have been unloaded using an inert gas, such as nitrogen, so that what may appear to be a clean, safe space is not. Tanks which have contained crude oil, residues, asphalt or high-melting-point products may need to be steam or chemically cleaned prior to ventilation and entry, or may have a pyrophoric hazard. Ventilating tanks to free them from vapours and toxic or inert gases may be accomplished by opening the lowest and furthest valve or connection on each tank or compartment and placing an air eductor at the furthest top opening. Monitoring should be performed prior to entry without respiratory protection to assure that all of the corners and low spots in the tank, such as sumps, have been thoroughly vented, and ventilation should continue while working in the tank.
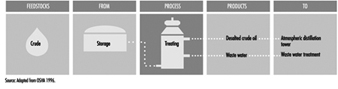
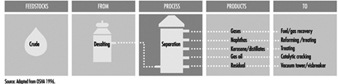
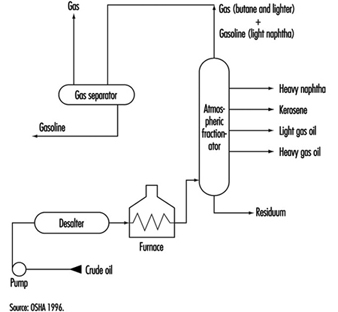
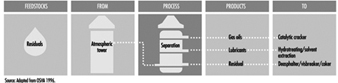
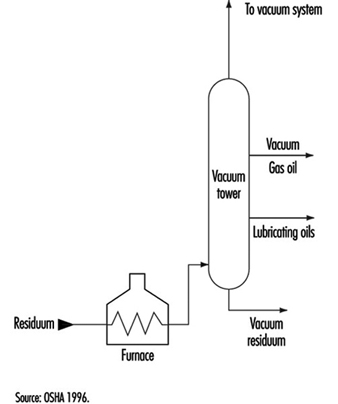
Aboveground Tank Storage of Liquid Petroleum Products
Crude oil, gas, LNG and LPG, processing additives, chemicals and petroleum products are stored in aboveground and underground atmospheric (non-pressure) and pressure storage tanks. Storage tanks are located at the ends of feeder lines and gathering lines, along truck pipelines, at marine loading and unloading facilities and in refineries, terminals and bulk plants. This section covers aboveground atmospheric storage tanks in refinery, terminal and bulk plant tank farms. (Information concerning aboveground pressure tanks is covered below, and information concerning underground tanks and small aboveground tanks is in the article “Motor vehicle fuelling and servicing operations”.)
Terminals and bulk plants
Terminals are storage facilities which generally receive crude oil and petroleum products by trunk pipeline or marine vessel. Terminals store and redistribute crude oil and petroleum products to refineries, other terminals, bulk plants, service stations and consumers by pipelines, marine vessels, railroad tank cars and tank trucks. Terminals may be owned and operated by oil companies, pipeline companies, independent terminal operators, large industrial or commercial consumers or petroleum product distributors.
Bulk plants are usually smaller than terminals and typically receive petroleum products by rail tank car or tank truck, normally from terminals but occasionally direct from refineries. Bulk plants store and redistribute products to service stations and consumers by tank truck or tank wagon (small tank trucks of approximately 9,500 to 1,900 l capacity). Bulk plants may be operated by oil companies, distributors or independent owners.
Tank farms
Tank farms are groupings of storage tanks at producing fields, refineries, marine, pipeline and distribution terminals and bulk plants which store crude oil and petroleum products. Within tank farms, individual tanks or groups of two or more tanks are usually surrounded by enclosures called berms, dykes or fire walls. These tank farm enclosures may vary in construction and height, from 45-cm earth berms around piping and pumps inside dykes to concrete walls that are taller than the tanks they surround. Dykes may be built of earth, clay or other materials; they are covered with gravel, limestone or sea shells to control erosion; they vary in height and are wide enough for vehicles to drive along the top. The primary functions of these enclosures are to contain, direct and divert rain water, physically separate tanks to prevent the spread of fire in one area to another, and to contain a spill, release, leak or overflow from a tank, pump or pipe within the area.
Dyke enclosures may be required by regulation or company policy to be sized and maintained to hold a specific amount of product. For example, a dyke enclosure may need to contain at least 110% of the capacity of the largest tank therein, allowing for the volume displaced by the other tanks and the amount of product remaining in the largest tank after hydrostatic equilibrium is reached. Dyke enclosures may also be required to be constructed with impervious clay or plastic liners to prevent spilled or released product from contaminating soil or groundwater.
Storage tanks
There are a number of different types of vertical and horizontal aboveground atmospheric and pressure storage tanks in tank farms, which contain crude oil, petroleum feedstocks, intermediate stocks or finished petroleum products. Their size, shape, design, configuration, and operation depend on the amount and type of products stored and company or regulatory requirements. Aboveground vertical tanks may be provided with double bottoms to prevent leakage onto the ground and cathodic protection to minimize corrosion. Horizontal tanks may be constructed with double walls or placed in vaults to contain any leakage.
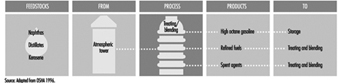
Atmospheric cone roof tanks
Cone roof tanks are aboveground, horizontal or vertical, covered, cylindrical atmospheric vessels. Cone roof tanks have external stairways or ladders and platforms, and weak roof to shell seams, vents, scuppers or overflow outlets; they may have appurtenances such as gauging tubes, foam piping and chambers, overflow sensing and signalling systems, automatic gauging systems and so on.
When volatile crude oil and flammable liquid petroleum products are stored in cone roof tanks there is an opportunity for the vapour space to be within the flammable range. Although the space between the top of the product and the tank roof is normally vapour rich, an atmosphere in the flammable range can occur when product is first put into an empty tank or as air enters the tank through vents or pressure/vacuum valves when product is withdrawn and as the tank breathes during temperature changes. Cone roof tanks may be connected to vapour recovery systems.
Conservation tanks are a type of cone roof tank with an upper and lower section separated by a flexible membrane designed to contain any vapour produced when the product warms up and expands due to exposure to sunlight in the daytime and to return the vapour to the tank when it condenses as the tank cools down at night. Conservation tanks are typically used to store aviation gasoline and similar products.
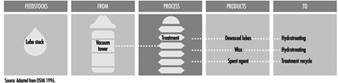
Atmospheric floating roof tanks
Floating roof tanks are aboveground, vertical, open top or covered cylindrical atmospheric vessels that are equipped with floating roofs. The primary purpose of the floating roof is to minimize the vapour space between the top of the product and the bottom of the floating roof so that it is always vapour rich, thus precluding the chance of a vapour-air mixture in the flammable range. All floating roof tanks have external stairways or ladders and platforms, adjustable stairways or ladders for access to the floating roof from the platform, and may have appurtenances such as shunts which electrically bond the roof to the shell, gauging tubes, foam piping and chambers, overflow sensing and signalling systems, automatic gauging systems and so on. Seals or boots are provided around the perimeter of floating roofs to prevent product or vapour from escaping and collecting on the roof or in the space above the roof.
Floating roofs are provided with legs which may be set in high or low positions depending on the type of operation. Legs are normally maintained in the low position so that the greatest possible amount of product can be withdrawn from the tank without creating a vapour space between the top of the product and the bottom of the floating roof. As tanks are brought out of service prior to entry for inspection, maintenance, repair or cleaning, there is a need to adjust the roof legs into the high position to allow room to work under the roof once the tank is empty. When the tank is returned to service, the legs are readjusted into the low position after it is filled with product.
Aboveground floating roof storage tanks are further classified as external floating roof tanks, internal floating roof tanks or covered external floating roof tanks.
External (open top) floating roof tanks are those with floating covers installed on open-top storage tanks. External floating roofs are usually constructed of steel and provided with pontoons or other means of flotation. They are equipped with roof drains to remove water, boots or seals to prevent vapour releases and adjustable stairways to reach the roof from the top of the tank regardless of its position. They may also have secondary seals to minimize release of vapour to the atmosphere, weather shields to protect the seals and foam dams to contain foam in the seal area in case of a fire or seal leak. Entry onto external floating roofs for gauging, maintenance or other activities may be considered confined-space entry, depending on the level of the roof below the top of the tank, the products contained in the tank and government regulations and company policy.
Internal floating roof tanks usually are cone roof tanks which have been converted by installing buoyant decks, rafts or internal floating covers inside the tank. Internal floating roofs are typically constructed of various types of sheet metal, aluminium, plastic or metal-covered plastic expanded foam, and their construction may be of the pontoon or pan type, solid buoyant material, or a combination of these. Internal floating roofs are provided with perimeter seals to prevent vapour from escaping into the portion of the tank between the top of the floating roof and the exterior roof. Pressure/vacuum valves or vents are usually provided at the top of the tank to control any hydrocarbon vapours which may accumulate in the space above the internal floater. Internal floating roof tanks have ladders installed for access from the cone roof to the floating roof. Entry onto internal floating roofs for any purpose should be considered confined-space entry.
Covered (external) floating roof tanks are basically external floating roof tanks that have been retrofitted with a geodesic dome, snow cap or similar semi-fixed cover or roof so that the floating roof is no longer open to the atmosphere. Newly constructed covered external floating roof tanks may incorporate typical floating roofs designed for internal floating roof tanks. Entry onto covered external floating roofs for gauging, maintenance or other activities may be considered confined-space entry, depending on the construction of the dome or cover, the level of the roof below the top of the tank, the products contained in the tank and government regulations and company policy.
Pipeline and marine receipts
An important safety, product quality and environmental concern in tank storage facilities is to prevent intermixing of products and overfilling tanks by developing and implementing safe operating procedures and work practices. Safe operation of storage tanks depends on receiving product into tanks within their defined capacity by designating receiving tanks prior to delivery, gauging tanks to determine the available capacity and ensuring that valves are properly aligned and that only the receiving tank inlet is opened, so the correct amount of product is delivered into the assigned tank. Drains in dyke areas surrounding tanks receiving product should normally be kept closed during receipt in case an overfill or spill occurs. Overfill protection and prevention can be accomplished by a variety of safe operating practices, including manual controls and automatic detection, signalling and shut-down systems and a means of communication, all of which should be mutually understood and acceptable to product transfer personnel at the pipeline, marine vessel and terminal or refinery.
Government regulations or company policy may require that automatic product level detection devices and signal and shut-down systems be installed on tanks receiving flammable liquids and other products from trunk pipelines or marine vessels. Where such systems are installed, electronic system integrity tests should be conducted on a regular basis or prior to product transfer, and if the system fails, transfers should follow manual receipt procedures. Receipts should be monitored manually or automatically, onsite or from a remote control location, to ensure that operations are proceeding as planned. Upon completion of transfer, all valves should be returned to normal operating position or set for the next receipt. Pumps, valves, pipe connections, bleeder and sample lines, manifold areas, drains and sumps should be inspected and maintained to assure good condition and to prevent spills and leakage.
Tank gauging and sampling
Tank storage facilities should establish procedures and safe work practices for gauging and sampling crude oil and petroleum products which take into consideration the potential hazards involved with each product stored and each type of tank in the facility. Although tank gauging is often done using automatic mechanical or electronic devices, manual gauging should be performed at scheduled intervals to assure the accuracy of the automatic systems.
Manual gauging and sampling operations usually require the operator to climb to the top of the tank. When gauging floating roof tanks, the operator then has to descend onto the floating roof unless the tank is fitted with gauging and sampling tubes that are accessible from the platform. With cone roof tanks, the gauger must open a roof hatch in order to lower the gauge into the tank. Gaugers should be aware of the confined-space entry requirements and potential hazards when entering onto covered floating roofs or down upon open-top floating roofs which are below established height levels. This may require the use of monitoring devices, such as oxygen, combustible gas and hydrogen sulphide detectors and personal and respiratory protective equipment.
Product temperatures and samples may be taken at the same time as manual gauging is conducted. Temperatures may also be recorded automatically and samples obtained from built-in sample connections. Manual gauging and sampling should be restricted while tanks are receiving product. Following the completion of receipt, a relaxation period of from 30 minutes to 4 hours, depending on the product and company policy, should be required to allow any electrostatic build-up to dissipate before conducting manual sampling or gauging. Some companies require that communications or visual contact be established and maintained between gaugers and other facility personnel when descending upon floating roofs. Entry onto tank roofs or platforms for gauging, sampling or other activities should be restricted during thunderstorms.
Tank venting and cleaning
Storage tanks are taken out of service for inspection, testing, maintenance, repair, retrofitting and tank cleaning as needed or at regular intervals dependent on government regulations, company policy and operating service requirements. Although tank venting, cleaning and entry is a potentially hazardous operation, this work can be accomplished without incident, provided that proper procedures are established and safe work practices followed. Without such precautions, injury or damage can occur from explosions, fires, lack of oxygen, toxic exposures and physical hazards.
Preliminary preparations
A number of preliminary preparations are required after it has been decided that a tank needs to be taken out of service for inspection, maintenance or cleaning. These include: scheduling storage and supply alternatives; reviewing the tank history to determine whether it has ever contained leaded product or has previously been cleaned and certified lead free; determining the amount and type of products contained and how much residue will remain in the tank; inspecting the outside of the tank, the surrounding area and the equipment to be used for product removal, vapour freeing and cleaning; assuring that personnel are trained, qualified and familiar with facility permit and safety procedures; assigning job responsibilities in accordance with the facility’s confined-space entry and hot- and safe-work permit requirements; and holding a meeting between terminal and tank cleaning personnel or contractors before tank cleaning or construction starts.
Control of ignition sources
After the removal of all available product from the tank through fixed piping, and before any water draws or sample lines are opened, all sources of ignition should be removed from the surrounding area until the tank is declared vapour free. Vacuum trucks, compressors, pumps and other equipment which is electrically or motor driven should be located upwind, either on top of or outside the dyke area, or, if inside the dyke area, at least 20 m from the tank or any other sources of flammable vapours. Tank preparation, venting and cleaning activities should cease during electrical storms.
Removing residue
The next step is to remove as much remaining product or residue in the tank as possible through pipeline and waterdraw connections. A safe-work permit may be issued for this work. Water or distillate fuel may be injected into the tank through fixed connections to help float product out of the tank. Residue removed from tanks that have contained sour crude should be kept wet until disposal to avoid spontaneous combustion.
Isolating the tank
After all available product has been removed through fixed piping, all piping connected to the tank, including product lines, vapour recovery lines, foam piping, sample lines and so on, should be disconnected by closing the valves nearest the tank and inserting blinds in the lines on the tank side of the valve to prevent any vapours from entering the tank from the lines. The portion of piping between the blinds and the tank should be drained and flushed. Valves outside the dyke area should be closed and locked or tagged. Tank pumps, internal mixers, cathodic protection systems, electronic gauging and level detection systems and so on should be disconnected, de-energized and locked or tagged out.
Vapour freeing
The tank is now ready to be made vapour free. Intermittent or continuous vapour testing should be conducted and work in the area restricted during tank ventilation. Natural ventilation, through opening the tank to the atmosphere, is not usually preferred, since it is neither as fast nor as safe as forced ventilation. There are a number of methods of mechanically venting a tank, depending on its size, construction, condition and internal configuration. In one method, cone roof tanks may be vapour freed by placing an eductor (a portable ventilator) at a hatch on the top of the tank, starting it slowly while a hatch at the bottom of the tank is opened and then setting it on high speed to draw air and vapours through the tank.
A safe- or hot-work permit should be issued covering ventilation activities. All blowers and eductors should be securely bonded to the tank shell to prevent electrostatic ignition. For safety purposes, blowers and eductors should preferably be operated by compressed air; however, explosion-proof electric- or steam-driven motors have been used. Internal floating roof tanks may need to have the portions above and below the floating roof vented separately. If vapours are discharged from a bottom hatch, a vertical tube at least 4 m above ground level and no lower than the surrounding dyke wall is needed in order to prevent vapours from collecting at low levels or reaching a source of ignition before dissipating. If necessary, vapours may be directed to the facility vapour recovery system.
As ventilation progresses, the remaining residue can be washed down and removed through the open bottom hatch by water and suction hoses, both of which should be bonded to the tank shell to prevent electrostatic ignition. Tanks which have contained sour crude oil or high-sulphur residual products may generate spontaneous heat and ignite as they dry out during ventilation. This should be avoided by wetting the inside of the tank with water to blanket the deposits from air and prevent a rise in temperature. Any iron sulphide residue should be removed from the open hatch to prevent ignition of vapours during ventilation. Workers engaged in washdown, removal and wetting activities should wear appropriate personal and respiratory protection.
Initial entry, inspection and certification
An indication of the progress being made in vapour freeing the tank can be obtained by monitoring vapours at the point of eduction during ventilation. Once it appears that the flammable vapour level is below that established by regulatory agencies or company policy, entry can be made into the tank for inspection and testing purposes. The entrant should wear appropriate personal and air-supplied respiratory protection; after testing the atmosphere at the hatch and obtaining an entry permit, the worker may enter the tank to continue testing and inspection. Checks for obstructions, falling roofs, weak supports, holes in the floor and other physical hazards should be conducted during the inspection.
Cleaning, maintenance and repair
As ventilation continues and the vapour levels in the tank drop lower, permits may be issued allowing entry by workers with appropriate personal and respiratory equipment, if needed, to start cleaning the tank. Monitoring for oxygen, flammable vapours and toxic atmospheres should continue, and if the levels inside the tank exceed those established for entry, the permit should automatically expire and the entrants should immediately leave the tank until the safe level is again achieved and the permit is reissued. Ventilation should continue during cleaning operations as long as any residue or sludge remains in the tank. Only low-voltage lighting or approved flashlights should be used during inspection and clean-up.
After tanks have been cleaned and dried, a final inspection and testing should be conducted before maintenance, repair or retrofitting work is started. Careful inspection of sumps, wells, floor plates, floating roof pontoons, supports and columns is needed to assure that no leaks have developed which allowed product to enter these spaces or seep beneath the floor. Spaces between foam seals and weather shields or secondary containment should also be inspected and tested for vapours. If the tank has previously contained leaded gasoline, or if no tank history is available, a lead-in-air test should be conducted and the tank certified lead free before workers are allowed inside without air-supplied respiratory equipment.
A hot-work permit should be issued covering welding, cutting and other hot work, and a safe-work permit issued to cover other repair and maintenance activities. Welding or hot work can create toxic or noxious fumes inside the tank, requiring monitoring, respiratory protection and continued ventilation. When tanks are to be retrofitted with double bottoms or internal floating roofs, a large hole is often cut into the side of the tank to provide unrestricted access and avoid the need for confined-space entry permits.
Blast cleaning and painting the outside of tanks usually follows tank cleaning and is completed before the tank is returned to service. These activities, together with cleaning and painting tank farm piping, may be performed while tanks and pipes are in service, by implementing and following prescribed safety procedures, such as conducting monitoring for hydrocarbon vapours and stopping blast cleaning while nearby tanks are receiving flammable liquid products. Blast cleaning with sand has the potential for hazardous exposure to silica; therefore, many government agencies and companies require the use of special non-toxic blast cleaning materials or grit, which may be collected, cleaned and recycled. Special vacuum collection blast cleaning devices may be used in order to avoid contamination when cleaning leaded paint from tanks and piping. Following blast cleaning, spots in the tank walls or piping suspected of having leaks and seeps should be tested and repaired before being painted.
Returning the tank to service
In preparation for return to service upon completion of tank cleaning, inspection, maintenance or repair, the hatches are closed, all blinds are removed and the piping is reconnected to the tank. Valves are unlocked, opened and aligned, and mechanical and electrical devices are reactivated. Many government agencies and companies require tanks to be hydrostatically tested to assure that there are no leaks before they are returned to service. Since a considerable amount of water is required to obtain the necessary pressure head for an accurate test, a water bottom topped with diesel fuel is often used. Upon completion of the testing, the tank is emptied and made ready to receive product. After receipt is completed and a relaxation time has elapsed, the legs on floating roof tanks are reset into the low position.
Fire protection and prevention
Whenever hydrocarbons are present in closed containers such as storage tanks in refineries, terminals and bulk plants, the potential exists for release of liquids and vapours. These vapours could mix with air in the flammable range and, if subjected to a source of ignition, cause an explosion or fire. Regardless of the capability of fire protection systems and personnel in the facility, the key to fire protection is fire prevention. Spills and releases should be stopped from entering sewers and drainage systems. Small spills should be covered with wet blankets, and larger spills with foam, to prevent vapours from escaping and mixing with air. Sources of ignition in areas when hydrocarbon vapours may be present should be eliminated or controlled. Portable fire extinguishers should be carried on service vehicles and located at accessible and strategic positions throughout the facility.
The establishment and implementation of safe work procedures and practices such as hot- and safe- (cold-) work permit systems, electrical classification programmes, lockout/tagout programmes, and employee and contractor training and education is critical to preventing fires. Facilities should develop preplanned emergency procedures, and employees should be knowledgeable in their responsibilities for reporting and responding to fires and evacuation. Telephone numbers of responsible persons and agencies to be notified in case of an emergency should be posted at the facility and a means of communication provided. Local fire departments, emergency response, public safety and mutual aid organizations should also be aware of the procedures and familiar with the facility and its hazards.
Hydrocarbon fires are controlled by one or a combination of methods, as follows:
- Removing fuel. One of the best and easiest methods of controlling and extinguishing a hydrocarbon fire is to shut off the source of fuel by closing a valve, diverting product flow or, if a small amount of product is involved, controlling exposures while allowing the product to burn away. Foam may also be used to cover hydrocarbon spills to prevent vapours from being emitted and mixing with the air.
- Removing oxygen. Another method is to shut off the supply of air or oxygen by smothering fires with foam or water fog, or by using carbon dioxide or nitrogen to displace air in enclosed spaces.
- Cooling. Water fog, mist or spray and carbon dioxide may be used to extinguish certain petroleum product fires by cooling the temperature of the fire below the product’s ignition temperature and by stopping vapours from forming and mixing with air.
- Interrupting combustion. Chemicals such as dry powders and halon extinguish fires by interrupting the chemical reaction of the fire.
Storage tank fire protection
Storage tank fire protection and prevention is a specialized science which depends on the interrelationship of tank type, condition and size; product and amount stored in the tank; tank spacing, dyking and drainage; facility fire protection and response capabilities; outside assistance; and company philosophy, industry standards and government regulations. Storage tank fires may be easy or very difficult to control and extinguish, depending primarily on whether the fire is detected and attacked during its initial inception. Storage tank operators should refer to the numerous recommended practices and standards developed by organizations such as the American Petroleum Institute (API) and the US National Fire Protection Association (NFPA), which cover storage tank fire prevention and protection in great detail.
If open-top floating roof storage tanks are out of round or if the seals are worn or not tight against the tank shells, vapours can escape and mix with air, forming flammable mixtures. In such situations, when lightning strikes, fires may occur at the point where the roof seals meet the shell of the tank. If detected early, small seal fires can often be extinguished by a hand-carried dry powder extinguisher or with foam applied from a foam hose or foam system.
If a seal fire cannot be controlled with hand extinguishers or hose streams, or if a large fire is in progress, foam may be applied onto the roof through fixed or semi-fixed systems or by large foam monitors. Precautions are necessary when applying foam onto the roofs of floating roof tanks; if too much weight is placed on the roof, it may tilt or sink, allowing a large surface area of product to be exposed and become involved in the fire. Foam dams are used on floating roof tanks to trap foam in the area between the seals and the tank shell. As the foam settles, water drains out under the foam dams and should be removed through the tank roof drain system to avoid overweighing and sinking the roof.
Depending on government regulations and company policy, storage tanks may be provided with fixed or semi-fixed foam systems which include: piping to the tanks, foam risers and foam chambers on the tanks; subsurface injection piping and nozzles inside the bottom of tanks; and distribution piping and foam dams on the tops of tanks.With fixed systems, foam-water solutions are generated in centrally located foam houses and pumped to the tank through a piping system. Semi-fixed foam systems typically use portable foam tanks, foam generators and pumps which are brought to the tank involved, connected to a water supply and connected to the tank’s foam piping.
Water-foam solutions may also be centrally generated and distributed within the facility through a system of piping and hydrants, and hoses would be used to connect the nearest hydrant to the tank’s semi-fixed foam system. Where tanks are not provided with fixed or semi-fixed foam systems, foam may be applied onto the tops of tanks, using foam monitors, fire hoses and nozzles. Regardless of the method of application, in order to control a fully involved tank fire, a specific amount of foam must be applied using special techniques at a specific concentration and rate of flow for a minimum amount of time depending primarily on the size of the tank, the product involved and the surface area of the fire. If there is not enough foam concentrate available to meet the required application criteria, the possibility of control or extinguishment is minimal.
Only trained and knowledgeable fire-fighters should be allowed to use water to fight liquid petroleum tank fires. Instantaneous eruptions, or boil-overs, can occur when water turns into steam upon direct application onto tank fires involving crude or heavy petroleum products. As water is heavier than most hydrocarbon fuels, it will sink to the bottom of a tank and, if enough is applied, fill the tank and push the burning product up and over the top of the tank.
Water is typically used to control or extinguish spill fires around the outside of tanks so that valves can be operated to control product flow, to cool the sides of involved tanks to prevent boiling liquid–expanding vapour explosions (BLEVEs—see the section “Fire hazards of LHGs” below) and to reduce the effect of heat and flame impingement on adjacent tanks and equipment. Because of the need for specialized training, materials and equipment, rather than allow employees to attempt to extinguish tank fires, many terminals and bulk plants have established a policy to remove as much product as possible from the involved tank, protect adjacent structures from heat and flame and allow the remaining product in the tank to burn under controlled conditions until the fire burns out.
Terminal and bulk plant health and safety
Storage tank foundations, supports and piping should be regularly inspected for corrosion, erosion, settling or other visible damage to prevent loss or degradation of product. Tank pressure/vacuum valves, seals and shields, vents, foam chambers, roof drains, water draw-off valves and overfill detection devices should be inspected, tested and maintained on a regular schedule, including removal of ice in the winter. Where flame arrestors are installed on tank vents or in vapour recovery lines, they have to be inspected and cleaned regularly and kept free of frost in the winter to ensure proper operation. Valves on tank outlets which close automatically in case of fire or drop in pressure should be checked for operability.
Dyke surfaces should drain or slope away from tanks, pumps and piping to remove any spilled or released product to a safe area. Dyke walls should be maintained in good condition, with drain valves kept closed except when draining water and dyke areas excavated as needed to maintain design capacity. Stairways, ramps, ladders, platforms and railings to loading racks, dykes and tanks should be maintained in a safe condition, free of ice, snow and oil. Leaking tanks and piping should be repaired as soon as possible. The use of victaulic or similar couplings on piping within dyked areas which could be exposed to heat should be discouraged to prevent lines from opening during fires.
Safety procedures and safe work practices should be established and implemented, and training or education provided, so that terminal and bulk plant operators, maintenance personnel, tank truck drivers and contractor personnel can work safely. These should include, as a minimum, information concerning the basics of hydrocarbon fire ignition, control and extinguishment; hazards and protection from exposures to toxic substances such as hydrogen sulphide and polynuclear aromatics in crude oil and residual fuels, benzene in gasoline and additives such as tetraethyl lead and methyl-tert-butyl ether (MTBE); emergency response actions; and normal physical and climatic hazards associated with this activity.
Asbestos or other insulation may be present in the facility as protection for tanks and piping. Appropriate safe-work and personal protective measures should be established and followed for handling, removing and disposing of such materials.
Environmental protection
Terminal operators and employees should be aware of and comply with government regulations and company policies covering environmental protection of ground and surface water, soil and air from pollution by petroleum liquids and vapours, and for handling and removing hazardous waste.
- Water contamination. Many terminals have oil/water separators to handle contaminated water from tank containment areas, run-off from loading racks and parking areas and water drained from tanks and open-top tank roofs. Terminals may be required to meet established water quality standards and obtain permits before discharging water.
- Air pollution. Air pollution prevention includes minimizing releases of vapours from valves and vents. Vapour recovery units collect vapours from loading racks and marine docks, even when tanks are vented prior to entry. These vapours are either processed and returned to storage as liquids or burned.
- Spills on land and water. Government agencies and companies may require that oil storage facilities have spill prevention control and counter-measure plans, and that personnel be trained and aware of the potential hazards, notifications to be made and the actions to take in case of a spill or release. In addition to handling spills within the terminal facility, personnel are often trained and equipped to respond to offsite emergencies, such as a tank truck rollover.
- Sewage and hazardous waste. Terminals may be required to meet regulatory requirements and obtain permits for discharge of sewage and oily waste to public or privately owned treatment works. Various government requirements and company procedures may apply to the onsite storage and handling of hazardous waste such as asbestos insulation, tank cleaning residue and contaminated product. Workers should be trained in this activity and be made aware of the potential hazards from exposures which could occur.
LHG Storage and Handling
Bulk storage tanks
LHGs are stored in large bulk storage tanks at the point of process (gas and oil fields, gas plants and refineries) and at the point of distribution to the consumer (terminals and bulk plants). The two most commonly used methods of bulk storage of LHGs are:
- Under high pressure at ambient temperature. LHG is stored in steel pressure tanks (at 1.6 to 1.8 mPa) or in underground impermeable rock or salt formations.
- Under pressure close to atmospheric at low temperature. LHG is stored in thin-walled, heat-insulated steel storage tanks; in reinforced concrete tanks above and below ground; and in underground cryogenic storage tanks. Pressure is maintained close to atmospheric (0.005 to 0.007 mPa) at a temperature of –160°C for LNG stored in cryogenic underground storage tanks.
LPG bulk storage vessels are either cylindrically (bullet) shaped horizontal tanks (40 to 200 m3) or spheres (up to 8,000 m3). Refrigerated storage is typical for storage in excess of 2,400 m3. Both horizontal tanks, which are fabricated in shops and transported to the storage site, and spheres, which are built onsite, are designed and constructed in accordance with rigid specifications, codes and standards.
The design pressure of storage tanks should not be less than the vapour pressure of the LHG to be stored at the maximum service temperature. Tanks for propane-butane mixtures should be designed for 100% propane pressure. Consideration should be given to additional pressure requirements resulting from the hydrostatic head of the product at maximum fill and the partial pressure of non-condensible gases in the vapour space. Ideally, liquefied hydrocarbon gas storage vessels should be designed for full vacuum. If not, vacuum relief valves must be provided. Design features should also include pressure relief devices, liquid level gauges, pressure and temperature gauges, internal shut-off valves, back flow preventers and excess flow check valves. Emergency fail-safe shut-down valves and high level signals may also be provided.
Horizontal tanks are either installed aboveground, placed on mounds or buried underground, typically downwind from any existing or potential sources of ignition. If the end of a horizontal tank ruptures from over-pressurization, the shell will be propelled in the direction of the other end. Therefore, it is prudent to place an aboveground tank so that its length is parallel to any important structure (and so that neither end points toward any important structure or equipment). Other factors include tank spacing, location, and fire prevention and protection. Codes and regulations specify minimum horizontal distances between pressurized liquefied hydrocarbon gas storage vessels and adjoining properties, tanks and important structures as well as potential sources of ignition, including processes, flares, heaters, power transmission lines and transformers, loading and unloading facilities, internal combustion engines and gas turbines.
Drainage and spill containment are important considerations in designing and maintaining liquid hydrocarbon gas tank storage areas in order to direct spills to a location where they will minimize risk to the facility and surrounding areas. Dyking and impounding may be used where spills present a potential hazard to other facilities or to the public. Storage tanks are not usually dyked, but the ground is graded so that vapours and liquids do not collect underneath or around the storage tanks, in order to keep burning spills from impinging upon storage tanks.
Cylinders
LHGs for use by consumers, either LNG or LPG, are stored in cylinders at temperatures above their boiling points at normal temperature and pressure. All LNG and LPG cylinders are provided with protective collars, safety valves and valve caps. The basic types of consumer cylinders in use are:
- vapour withdrawal (1/2 to 50 kg) cylinders used by consumers, with larger ones usually refillable on an exchange basis with the supplier
- liquid withdrawal cylinders for dispensing into small consumer-owned refillable cylinders
- motor vehicle fuel cylinders, including vehicle cylinders (40 kg) permanently installed as fuel tanks on motor vehicles and filled and used in the horizontal position, and industrial truck cylinders designed to be stored, filled and handled in the upright position, but used in the horizontal position.
Properties of hydrocarbon gases
According to the NFPA, flammable (combustible) gases are those which burn in the normal concentrations of oxygen in air. The burning of flammable gases is similar to flammable hydrocarbon liquid vapours, as a specific ignition temperature is needed to initiate the burning reaction, and each will burn only within a certain defined range of gas-air mixtures. Flammable liquids have a flashpoint, which is the temperature (always below the boiling point) at which they emit sufficient vapours for combustion. There is no apparent flashpoint for flammable gases, since they are normally at temperatures above their boiling points, even when liquefied, and are therefore always at temperatures well in excess of their flashpoints.
The NFPA (1976) defines compressed and liquefied gases as follows:
- “Compressed gases are those which at all normal atmospheric temperatures inside their containers, exist solely in the gaseous state under pressure.”
- “Liquefied gases are those which at normal atmospheric temperatures inside their containers, exist partly in the liquid state and partly in the gaseous state, and are under pressure as long as any liquid remains in the container.”
The major factor which determines the pressure inside the vessel is the temperature of the liquid stored. When exposed to the atmosphere, the liquefied gas very rapidly vaporizes, travelling along the ground or water surface unless dispersed into the air by wind or mechanical air movement. At normal atmospheric temperatures, about one-third of the liquid in the container will vaporize.
Flammable gases are further classified as fuel gas and industrial gas. Fuel gases, including natural gas (methane) and LPGs (propane and butane), are burned with air to produce heat in ovens, furnaces, water heaters and boilers. Flammable industrial gases, such as acetylene, are used in processing, welding, cutting and heat-treating operations. The differences in combustion properties of LNG and LPGs are shown in table 1.
Table 1. Typical approximate combustion properties of liquified hydrocarbon gases.
|
Type gas |
Flammable range |
Vapour pressure |
Normal init. boiling |
Weight (pounds/gal) |
BTU per ft3 |
Specific gravity |
|
LNG |
4.5–14 |
1.47 |
–162 |
3.5–4 |
1,050 |
9.2–10 |
|
LPG (propane) |
2.1–9.6 |
132 |
–46 |
4.24 |
2,500 |
1.52 |
|
LPG (butane) |
1.9–8.5 |
17 |
–9 |
4.81 |
3,200 |
2.0 |
Safety hazards of LPG and LNG
The safety hazards applicable to all LHGs are associated with flammability, chemical reactivity, temperature and pressure. The most serious hazard with LHGs is the unplanned release from containers (canisters or tanks) and contact with an ignition source. Release can occur by failure of the container or valves for a variety of reasons, such as overfilling a container or from overpressure venting when the gas expands due to heating.
The liquid phase of LPG has a high coefficient of expansion, with liquid propane expanding 16 times and liquid butane 11 times as much as water with the same rise in temperature. This property must be considered when filling containers, as free space must be left for the vapour phase. The correct quantity to be filled is determined by a number of variables, including the nature of the liquefied gas, temperature at time of filling and expected ambient temperatures, size, type (insulated or uninsulated) and location of container (above or below ground). Codes and regulations establish allowable quantities, known as “filling densities”, which are specific for individual gases or families of similar gases. Filling densities may be expressed by weight, which are absolute values, or by liquid volume, which must always be temperature corrected.
The maximum amount that LPG pressure containers should be filled with liquid is 85% at 40 ºC (less at higher temperatures). Because LNG is stored under low temperatures, LNG containers may be liquid filled from 90% to 95%. All containers are provided with overpressure relief devices which normally discharge at pressures relating to liquid temperatures above normal atmospheric temperatures. As these valves cannot reduce the internal pressure to atmospheric, the liquid will always be at a temperature above its normal boiling point. Pure compressed and liquefied hydrocarbon gases are non-corrosive to steel and most copper alloys. However, corrosion can be a serious problem when sulphur compounds and impurities are present in the gas.
LPGs are 1-1/2 to 2 times heavier than air and, when released in air, tend to quickly disperse along the ground or water surface and collect in low areas. However, as soon as the vapour is diluted by air and forms a flammable mixture, its density is essentially the same as air, and it disperses differently. Wind will significantly reduce the dispersion distance for any size of leak. LNG vapours react differently from LPG. Because natural gas has a low vapour density (0.6), it will mix and disperse rapidly in open air, reducing the chance of forming a flammable mixture with air. Natural gas will collect in enclosed spaces and form vapour clouds which could be ignited. Figure 4 indicates how a liquefied natural gas vapour cloud spreads downwind in different spill situations.
Figure 4. Extension of LNG vapour cloud downwind from different spills (wind speed 8.05 km/h).
Although LHG is colourless, when released in air its vapours will be noticeable due to the condensation and freezing of water vapour contained in the atmosphere which is contacted by the vapour. This may not occur if the vapour is near ambient temperature and its pressure is relatively low. Instruments are available which can detect the presence of leaking LHG and signal an alarm at levels as low as 15 to 20% of the lower flammable limit (LFL). These devices may also stop all operations and activate suppression systems, should the concentrations of gas reach 40 to 50% of the LFL. Some industrial operations provide forced ventilation to keep leaking fuel-air concentrations below the lower flammable limit. Heater and furnace burners may also have devices which automatically stop the flow of gas if the flame is extinguished.
LHG leakage from tanks and containers may be minimized by the use of limiting and flow control devices. When decompressed and released, LHG will flow out of containers with a low negative pressure and low temperature. The auto refrigeration temperature of the product at the lower pressure must be considered when selecting materials of construction for containers and valves, to prevent metal embrittlement followed by rupture or failure due to exposure to low temperatures.
LHG can contain water in both its liquid and gaseous phases. Water vapour can saturate gas in a specific amount at a given temperature and pressure. If the temperature or pressure changes, or the water vapour content exceeds the evaporation limits, the water condenses. This can create ice plugs in valves and regulators and form hydrocarbon hydrate crystals in pipelines, devices and other apparatus. These hydrates can be decomposed by heating the gas, lowering the gas pressure or introducing materials, such as methanol, which reduce the water vapour pressure.
There are differences in the characteristics of compressed and liquefied gases which must be considered from safety, health and fire aspects. As an example, the differences in the characteristics of compressed natural gas and LNG are illustrated in table 2.
Table 2. Comparison of characteristics of compressed and liquified gas.
|
Type gas |
Flammable range |
Heat release rate (BTU/gal) |
Storage condition |
Fire risks |
Health risks |
|
Compressed natural gas |
5.0–15 |
19,760 |
Gas at 2,400 to 4,000 psi |
Flammable gas |
Asphyxiant; overpressure |
|
LNG |
4.5–14 |
82,450 |
Liquid at 40–140 psi |
Flammable gas 625:1 expansion ratio; BLEVE |
Asphyxiant; cryogenic liquid |
Health hazards of LHGs
The primary occupational injury concern in handling LHGs is the potential hazard of frostbite to the skin and eyes from contact with liquid during handling and storage activities including sampling, measuring, filling, receiving and delivery. As with other fuel gases, when improperly burned, compressed and liquefied hydrocarbon gases will emit undesirable levels of carbon monoxide.
Under atmospheric pressures and low concentrations, compressed and liquefied hydrocarbon gases are normally non-toxic, but they are asphyxiants—they will displace oxygen (air) if released in enclosed or confined spaces. Compressed and liquefied hydrocarbon gases may be toxic if they contain sulphur compounds, especially hydrogen sulphide. Because LHGs are colourless and odourless, safeguards include adding odourants, such as mercaptans, to consumer fuel gases to aid in leak detection. Safe work practices should be implemented to protect workers from exposure to mercaptans and other additives during storage and injection. Exposure to LPG vapours in concentrations at or above the LFL may cause a general central nervous system depression similar to anaesthesia gases or intoxicants.
Fire hazards of LHGs
Failure of liquefied gas (LNG and LPG) containers constitutes a more severe hazard than failure of compressed gas containers, as they release greater quantities of gas. When heated, liquefied gases react differently from compressed gases, because they are two-phase (liquid-vapour) products. As the temperature rises, the vapour pressure of the liquid is increased, resulting in increased pressure inside the container. The vapour phase first expands, followed by expansion of the liquid, which then compresses the vapour. The design pressure for LHG vessels is therefore assumed to be near that of the gas pressure at maximum possible ambient temperature.
When a liquefied gas container is exposed to fire, a serious condition can occur if the metal in the vapour space is allowed to heat. Unlike the liquid phase, the vapour phase absorbs little heat. This allows the metal to heat rapidly until a critical point is reached at which an instantaneous, catastrophic explosive failure of the container occurs. This phenomenon is known as a BLEVE. The magnitude of a BLEVE depends on the amount of liquid vaporizing when the container fails, the size of the pieces of exploded container, the distance they travel and the areas they impact. Uninsulated LPG containers may be protected against a BLEVE by applying cooling water to those areas of the container which are in the vapour phase (not in contact with LPG).
Other more common fire hazards associated with compressed and liquefied hydrocarbon gases include electrostatic discharge, combustion explosions, large open-air explosions and small leaks from pump seals, containers, valves, pipes, hoses and connections.
- Electrostatic charges may be generated when LHG is shipped in pipelines, when loaded and unloaded, in blending and filtering and during tank cleaning.
- Combustion explosions result when escaping gas or vapour is contained in a confined space or structure and combines with air to create a flammable mixture. When this flammable mixture contacts a source of ignition, it burns instantaneously and rapidly, producing extreme heat. The very hot air expands quickly, causing a considerable rise in pressure. If the space or structure is not strong enough to contain this pressure, a combustion explosion occurs.
- Flammable gas fires result when there is no confinement of the escaping gas or vapours, or ignition occurs when only a small amount of gas has been released.
- Large open-air explosions occur when a massive failure of a container releases a large vapour cloud of gas which is ignited before it disperses.
Controlling sources of ignition in hazardous areas is essential for the safe handling of compressed and liquefied hydrocarbon gases. This may be accomplished by establishing a permit system to authorize and control hot work, smoking, operation of motor vehicles or other internal combustion engines, and the use of open flames in areas where compressed and liquefied hydrocarbon gas is transported, stored and handled. Other safeguards include the use of properly classified electrical equipment and bonding and grounding systems to neutralize and dissipate static electricity.
The best means of reducing the fire hazard of leaking compressed or liquefied hydrocarbon gas is to stop the release, or shut off the flow of product, if possible. Although most LHGs will vaporize upon contact with air, lower vapour pressure LPGs, such as butane, and even some higher vapour pressure LPGs, such as propane, will pool if ambient temperatures are low. Water should not be applied to these pools, as it will create turbulence and increase the rate of vaporization. Vaporization from pool spills can be controlled by the careful application of foam. Water, if correctly applied against a leaking valve or small rupture, can freeze upon contact with the cold LHG and block the leak. LHG fires require controlling heat impingement upon storage tanks and containers by the application of cooling water. While compressed and liquefied hydrocarbon gas fires can be extinguished by the use of water spray and dry powder extinguishers, it is often more prudent to allow controlled burning so that a combustible explosive vapour cloud does not form and re-ignite should the gas continue to escape after the fire is extinguished.
Motor Vehicle Fueling and Servicing Operations
Petroleum-based fuels and lubricants are sold directly to consumers at full-service and self-service (with or without repair bays) service stations, car washes, automotive service centres, motor vehicle agencies, truck stops, repair garages, automotive parts stores and convenience stores. Service station attendants, mechanics and other employees who fuel, lubricate and service motor vehicles should be aware of the physical and chemical hazards of the petroleum fuels, lubricants, additives and waste products they come into contact with and follow appropriate safe work procedures and personal protection measures. The same physical and chemical hazards and exposures are present at commercial facilities, such as those operated by motor truck fleets, automobile rental agencies and bus companies for fuelling and servicing their own vehicles.
Because they are the facilities where motor fuels are delivered direct to the user’s vehicle, service stations, particularly those where drivers fuel their own vehicles, are where employees and the general public are most likely to come into direct contact with hazardous petroleum products. Other than those drivers who change their own oil and lubricate their own vehicles, the likelihood of contact with lubricants or used oil by motorists, except for incidental contact when checking fluid levels, is very small.
Service Station Operations
Fuel island area and dispensing system
Employees should be aware of the potential fire, safety and health hazards of gasoline, kerosene, diesel and other fuels dispensed at service stations. They should also be aware of suitable precautions. These include: safe dispensing of fuels into vehicles and containers, clean-up and disposal of spills, fighting incipient fires and draining fuels safely. Service stations should provide fuel-dispenser pumps which operate only when the fuel-hose nozzles are removed from the dispensers’ brackets and the switches are manually or automatically activated. Fuel-dispensing devices should be mounted on islands or protected against collision damage by barriers or curbs. Dispensing equipment, hoses and nozzles should be inspected regularly for leaks, damage and malfunctions. Safety features may be installed on fuel dispensers such as emergency breakaway devices on hoses, which retain liquid on each side of the break point, and impact valves with fusible links at the base of dispensers, which close automatically in event of severe impact or fire.
Government regulations and company policies may require that signs be posted in dispensing areas similar to the following signs, which are required in the United States:
- “No Smoking—Shut off engine”
- “WARNING: It is unlawful and dangerous to dispense gasoline into unapproved containers”
- “Federal Law prohibits the introduction of any gasoline containing lead or phosphorus into any motor vehicle labelled UNLEADED GASOLINE ONLY”
- “UNLEADED GASOLINE”, posted at unleaded gasoline dispensers and “CONTAINS LEAD ANTIKNOCK COMPOUNDS”, posted at leaded gasoline dispensers.
Fuelling vehicles
Service station employees should know where the fuel dispenser pump emergency shut-off switches are located and how to activate them, and should be aware of potential hazards and procedures for safely dispensing fuel into vehicles, such as the following:
- Vehicle engines should be shut off and smoking prohibited while fuelling to reduce the hazards of accidental vehicle movement, spills and fuel vapour ignition.
- When fuel is dispensed, the nozzle should be inserted into the vehicle’s fill pipe and contact between the nozzle and the fill pipe maintained to provide for an electric bond until the delivery has been completed. Nozzles should not be blocked open with fuel caps or other objects. Where allowed, approved latches should be used to hold open automatic nozzles.
- Vehicles such as cement mixers and recreation vehicles with auxiliary internal combustion engines should not be fuelled until both the vehicle’s engines and auxiliary engines are shut off. Care should be taken when fuelling recreational or other vehicles equipped with gas-fired stoves, refrigerators and water heaters to ensure that fuel vapours are not ignited by pilot lights. Employees should not fuel trucks while standing on the side rail, truck bed or fuel tank.
- Fuel tanks on motorcycles, motor bicycles, fork-lift trucks and similar vehicles should not be filled while the engine is running or when anyone is seated on the vehicle. The tanks should be filled at a slow rate to prevent fuel spills that could run onto hot engines and start fires.
- After fuelling, hose nozzles should be promptly replaced on the dispensers, pumps turned off and caps replaced on fill pipes or containers.
Filling portable fuel containers
Service stations should establish procedures such as the following for safely dispensing fuel into portable containers:
- Where required by government regulation or company policies, fuel should be dispensed only into approved, properly identified and labelled portable containers, with or without dispensing spouts, nozzles or hoses and equipped with vents and screw tops or self-closing gravity, spring action or combination fusible link covers designed to provide pressure relief.
- Containers should be placed on the ground and filled slowly to avoid splash filling and overfills and to provide for grounding (earthing). Containers should not be filled while in a vehicle or in the bed of a truck, particularly one with a plastic liner, as proper grounding cannot be achieved. Bonding wires and clamps should be provided and used, or contact should be maintained between dispenser nozzles and containers to provide a bond while filling, and between container spouts or funnels and tanks during refuelling from containers.
- When pouring fuel from containers which do not have built-in spouts, funnels should be used to minimize spillage and avoid splash filling.
- Portable containers which contain fuel or vapours should be properly stored in approved storage cabinets or rooms away from sources of heat and ignition.
Storage tanks, fill pipes, fill caps and vents
Service station underground and aboveground storage-tank gauge and fill-caps should be kept closed except when filling and gauging to minimize release of fuel vapours. When tank-gauge openings are located inside buildings, spring-loaded check valves or similar devices should be provided to protect each of the openings against fluid overflow and possible vapour release. Storage-tank vents should be located in accordance with government regulations and company policy. Where venting to open air is permitted, vent-pipe openings from both underground and aboveground storage tanks should be located at a high level so that flammable vapours are directed away from potential sources of ignition and will not enter windows or air intakes or doors or become trapped under eaves or overhangs.
Improper mixing of different products during deliveries may be caused by lack of identification or improper colour coding or markings on storage tanks. Storage-tank covers, fill pipes, caps and fill-box rims or pads should be properly identified as to products and grades so as to reduce the potential of a delivery into the wrong tank. Identification symbols and colour coding should conform to government regulations, company policies or industry standards, such as the American Petroleum Institute’s (API) Recommended Practice 1637, Using the API Color Symbol System to Mark Equipment and Vehicles for Product Identification at Service Stations and Distribution Terminals. A chart indicating the symbols or colour codes in use should be available at the service station during deliveries.
Delivery of fuel to service stations
Service stations should establish and implement procedures such as the following, for the safe delivery of fuel into aboveground and underground service station storage tanks:
Prior to delivery
- Vehicles and other objects should be moved from the area where the delivery tank truck and delivery hoses will be located.
- Delivery tank trucks should be positioned away from traffic areas, and vehicles should be restricted from driving near the unloading area or over hoses by the use of traffic cones or barriers.
- Receiving storage tanks should be gauged prior to delivery to determine if there is sufficient capacity, and checked to see if any water is in the tank.
- Drivers should assure that fuel is delivered into the correct tanks, that gauge caps are replaced before starting delivery and that all tank openings not being used for delivery are covered.
- Where required by company policies or government regulation, the tank truck vapour recovery system should be connected to the receiving storage tank prior to starting delivery.
During delivery
- Drivers should monitor the area near the receiving tank’s vents for potential ignition sources and check that the vents operate properly during delivery.
- Drivers should remain where they can observe the delivery and be able to stop delivery or take other appropriate action in event of an emergency, such as ejection of liquid from vents or if an overfill device or tank vent alarm activates.
After delivery
- Storage tanks may be gauged after delivery to verify that specific tanks have received the correct products and the proper amount of products as indicated on the delivery ticket or record. Samples may be taken from the tanks after delivery for quality-control purposes.
- After delivery, spill containment devices should be drained if necessary and the correct fill and gauge caps and storage tank covers replaced on the proper tanks.
Other Service Station Functions
Storage of flammable and combustible liquids
Government regulations and company policies may control the storage, handling and dispensing of flammable and combustible liquids and automotive chemicals such as paints, starter fluids, antifreeze, battery acids, window washer fluids, solvents and lubricants in service stations. Service stations should store aerosols and flammable liquids in closed containers in approved, well-ventilated areas, away from sources of heat or ignition, in appropriate flammable liquid rooms, lockers or cabinets, or in separate, outside buildings.
Electrical safety and lighting
Service station employees should be familiar with electrical safety fundamentals applicable to service stations, such as the following:
- Lighting and electrical installations, equipment and fixtures of the proper electrical classification should be provided and maintained in accordance with codes and regulations and should not be replaced by equipment of lesser classification.
- Electric tools, water coolers, ice machines, refrigerators and similar electrical equipment should be properly grounded (earthed). Portable lights should be protected against breakage to minimize the chance that a spark might ignite flammable vapours in case bulbs break.
Adequate illumination should be provided at appropriate locations in service stations to reduce the potential for accidents and injuries. Government regulations, company policies or voluntary standards may be used to determine appropriate illumination levels. See table 1.
Table 1. Illumination levels for service station areas.
|
Service station area |
Suggested foot candles |
|
Active traffic areas |
20 |
|
Storage areas and stockrooms |
10–20 |
|
Washrooms and waiting areas |
30 |
|
Dispenser islands, work benches and cashier areas |
50 |
|
Service, repair, lubrication and washing areas |
100 |
|
Offices |
100–150 |
Source: ANSI 1967.
Lockout/tagout
Service stations should establish and implement lockout/tagout procedures to prevent the release of potentially hazardous energy while performing maintenance, repair and service work on electrical, mechanical, hydraulic and pneumatic powered tools, equipment, machinery and systems such as lifts, hoists and jacks, lubrication equipment, fuel-dispenser pumps and compressors. Safe work procedures to prevent the accidental start-up of vehicle engines during servicing or repair should include disconnecting the battery or removing the key from the ignition.
Service station fluids
Fluid and coolant levels
Before working under a hood (bonnet), employees should assure that it will stay open by testing the tension or using a rod or brace. Employees should exercise caution when checking vehicle engine fluids to avoid burns from exhaust manifolds and to prevent contact between dipsticks and electrical terminals or wires; care is also necessary when checking transmission fluid levels (since the engine must be running). Employees should follow safe work procedures when opening radiators, such as allowing pressurized radiators to cool and covering radiator caps with a heavy cloth when opening, using PPE and standing with face turned away from radiators so as to not inhale any escaping steam or vapours.
Antifreeze and window washer fluids
Employees servicing vehicles should be aware of the hazards of both glycol and alcohol antifreezes and window washer fluid concentrates and how to safely handle them. This includes precautions such as storing alcohol-based products in tightly closed drums or packaged containers, in separate rooms or lockers, away from all heating equipment, and providing containment to prevent contamination of drains and ground in the event of a spill or leak of glycol-type antifreeze. Antifreeze or washer fluid should be dispensed from upright drums by using tightly connected hand pumps equipped with drip returns, rather than by using faucets or valves on horizontal drums, which may leak or be knocked open or broken off, causing spills. Air pressure should not be used to pump antifreeze or washer fluid concentrates from drums. Empty portable antifreeze and washer fluid concentrate containers should be completely drained prior to disposal, and applicable regulations governing the disposal of glycol antifreeze solutions should be followed.
Lubrication
Service stations should ensure that employees are aware of the characteristics and uses of the different fuels, oils, lubricants, greases, automotive fluids and chemicals available in the facility and their correct selection and application. The proper tools should be used to remove crankcase, transmission and differential drains, test plugs and oil filters so as to not damage vehicles or equipment. Pipe wrenches, extenders and chisels should be used only by employees who know how to safely remove frozen or rusted plugs. Because of the potential hazards involved, high-pressure lubricating equipment should not be started until the nozzles are set firmly against grease fittings. If testing is to be done prior to use, the nozzle should be aimed into an empty drum or similar receptacle, and not into a hand-held rag or cloth.
Lift operations
Employees working in and around vehicle service areas should be aware of unsafe conditions and follow safe work practices such as not standing in front of vehicles while they are being driven into service bays, over lubrification pits or onto lifts, or when vehicles are being lifted.
- Vehicles should be properly aligned on two-rail, free-wheel or frame-contact lifts, since an off-centre position may cause a vehicle to fall.
- Lifts should not be raised until occupants have left the vehicles and a check of overhead clearance has been made.
- Once the vehicle is in position, the emergency stop device should be set so the lift will not fall in the event of a pressure drop. If a lift is in a position where the emergency stop device cannot be engaged, blocks or safety stands should be placed under the lift or vehicle.
- A hydraulic lift may be equipped with a low-oil control valve, which prevents operation if the oil in the supply tank falls below a minimum level, since the lift can drop accidentally under those conditions.
When wheel-bearing lubrication, brake repair, tyre changing or other services are performed on free-wheel or frame-contact lifts, vehicles should be raised slightly above the floor to allow employees to work from a squatting position, to reduce the possibility of back strain. After vehicles are raised, the wheels should be blocked to prevent rolling, and safety stands should be placed underneath for support in case of jack or lift failure. When removing wheels from vehicles on drive-on lifts, the vehicles should be blocked securely to prevent rolling. If jacks or stands are used to lift and support vehicles, they should be of the proper capacity, placed at appropriate lift points on the vehicles and checked for stability.
Servicing tyres
Employees should be aware of how to safely check pressures and inflate tyres; tyres should be inspected for excessive wear, maximum tyre pressures should not be exceeded, and the worker should stand or kneel to the side and turn the face when inflating tyres. Employees should be aware of hazards and follow safe work practices when servicing wheels with multi-piece and single-piece rims and lock-ring-rim wheels on trucks and trailers. When repairing tyres with flammable or toxic patching compounds or liquids, precautions such as controlling ignition sources, using PPE and providing adequate ventilation, should be observed.
Parts cleaning
Service station employees should be aware of the fire and health hazards of using gasoline or low-flashpoint solvents to clean parts and should follow safe practices such as using approved solvents with a flashpoint above 60ºC. Parts washers should have a protective cover that is kept closed when the washer is not in use; when the washer is open, there should be a hold-open device such as fusible links, which allows the cover to close automatically in case of fire.
Employees should take precautions so that gasoline or other flammable liquids do not contaminate the cleaning solvent and lower its flashpoint to create a fire hazard. Contaminated cleaning solvent should be removed and placed in approved containers for proper disposal or recycling. Employees who clean parts and equipment using cleaning solvents should avoid skin and eye contact and use appropriate PPE. Solvents should not be used for hand-washing and other personal hygiene.
Compressed air
Safe work practices should be established by service stations for the operation of air compressors and the use of compressed air. The air hoses should be used only for inflating tyres and for lubrication, maintenance and auxiliary services. Employees should be aware of the hazards of pressurizing fuel tanks, air horns, water tanks and other non-air pressure containers. Compressed air should not be used for cleaning or to blow residue from vehicle brake systems, since many brake linings, especially on older model vehicles, contain asbestos. Safer methods such as cleaning with vacuums or liquid solutions should be used.
Storage battery service and handling
Service stations should establish procedures to ensure that storage, handling and disposal of batteries and battery electrolyte fluids follow government regulations and company policies. Employees should be aware of the hazards of electrical short circuits when charging, removing, installing or handling batteries; disconnect the ground (negative) cable first before removing batteries; and reconnect the ground (negative) cable last when installing batteries. When removing and replacing batteries, a carrier may be used to facilitate lifting and to avoid touching the battery.
Employees should be aware of safe practices such as the following for handling battery solution:
- Containers of electrolyte solution should be stored at temperature ranges between 16 and
32ºC in safe areas where they cannot overturn. Any electrolyte solution spilled on the batteries or in the filling area should be flushed with water. Baking soda (sodium bicarbonate) may be used on spills, since it is an effective neutralizer for battery electrolyte solution.
- New batteries should be placed on the floor or work table when being filled with electrolyte solution, and the caps should be replaced prior to installation. New batteries should not be filled when they are inside vehicles.
- Face shields and chemical goggles, aprons and gloves may be used to minimize exposure to battery solution. Battery solution should be handled and dispensed where a supply of potable water or eye wash fluid is available, in case the battery solution spills or contacts an employee’s skin or eyes. Do not use neutralizing solutions on skin or eyes.
- When servicing batteries, corrosive particles which accumulate around the terminals should be brushed away, washed with clean water, neutralized with baking soda or other similar agents and prevented from contacting eyes or clothing.
Employees should check fluid levels in batteries prior to charging and periodically check them during charging to determine whether batteries are overheating. Chargers should be turned off before disconnecting cables from batteries, to avoid creating sparks which may ignite hydrogen gas generated during the charge. When “quick charging” batteries are installed in vehicles, the vehicles should be moved away from the fuel-dispensing islands, and the battery ground (negative) cables should be disconnected before connecting the charger units. If the batteries are located within passenger compartments or under vehicle floorboards, they should be removed before charging.
Employees should be familiar with the hazards and safe procedures to “jump start” vehicles that have dead batteries, in order to avoid electrical system damage or injury from exploding batteries if the jumper cables are hooked up incorrectly. Employees should never jump start or charge frozen batteries.
Driving vehicles and towing
Employees should be trained, qualified and have proper motor vehicle operator’s licences to drive customer or company vehicles, service trucks or towing equipment either on or off the premises. All vehicles should be operated in compliance with government regulations and company policies. Operators should check the vehicle’s brakes immediately, and vehicles with faulty brakes should not be driven. Employees operating tow trucks should be familiar with safe operating procedures, such as operating the hoist, checking the transmission and frame of the vehicle to be towed and not exceeding the tow truck’s maximum lifting capacity.
Confined spaces in service stations
Service station employees should be aware of the hazards associated with entry into confined spaces such as aboveground and underground tanks, sumps, pump pits, waste containment tanks, septic tanks and environmental collection wells. Unauthorized entry should not be allowed, and confined-space entry permit procedures should be established that apply to both employee and contractor entrants.
Emergency procedures
Service stations should develop emergency procedures, and employees should know how to sound the alarms, how to notify authorities of emergencies when and how to evacuate and what appropriate response actions should be taken (such as shutting off emergency switches in the event of spills or fires in the dispensing pump areas). Service stations may establish security programmes to familiarize employees with robbery and violence prevention, depending on the service station’s location, hours of operation and potential threats.
Service Station Health and Safety
Fire protection
Gasoline vapours are heavier than air and may travel long distances to reach sources of ignition when released during fuel filling, spills, overflows or repairs. Proper ventilation should be provided in enclosed areas to allow for dissipation of gasoline vapours. Fires may occur from spills and overflows when fuelling or servicing vehicles or delivering product into service station tanks, particularly if smoking is not restricted or if vehicle engines remain running during fuelling. To avoid fires, vehicles should be pushed away from spill areas or the spilled gasoline should be cleaned from under or around vehicles before starting their engines. Vehicles should not be permitted to enter or drive through spills.
Employees should be aware of other causes of fires in service stations, such as improper handling, transfer and storage of flammable and combustible liquids, accidental releases during fuel system repairs, electrostatic discharge when changing filters on gasoline dispensers and the use of improper or unprotected work lights. Draining gasoline from vehicle fuel tanks could be very hazardous due to the potential for release of fuel and vapours, especially in enclosed service areas when sources of ignition may be present.
Hot-work permits should be issued when work other than vehicle repair and servicing is performed which introduces sources of ignition in areas where flammable vapours may be present. Employees should be aware that carburettor priming should not be attempted while vehicle engines are running or being turned over with their starters, since flashbacks could ignite the fuel vapours. Employees should follow safe procedures, such as using starter fluid and not gasoline for priming carburettors and using clamps to hold the chokes open while attempting to start the engine.
Although government regulations or company policies may require the installation of fixed fire-protection systems, fire extinguishers are usually the primary means of fire protection in service stations. Service stations should provide fire extinguishers of the proper classification for the expected hazards. Fire extinguishers and fixed fire protection systems should be regularly inspected, maintained and serviced, and employees should know when, where and how to use the fire extinguishers and how to activate the fixed systems.
Service stations should install fuel-dispenser emergency shut-down controls at clearly identified and accessible locations and ensure that employees know the purpose, location and operation of these controls. To prevent spontaneous combustion, oily rags should be kept in covered metal containers until they are recycled or discarded.
Safety
Employee injuries at service stations may result from improper use of tools, equipment and ladders; not wearing PPE; falling or tripping; working in awkward positions; and lifting or carrying cases of materials incorrectly. Injuries and accidents may also occur from not following safe practices when working on hot radiators, transmissions, engines and exhaust systems, servicing tyres and batteries, and working with lifts, jacks, electrical equipment and machinery; from robbery and assault; and from improper use of or exposure to automotive cleaners, solvents and chemicals.
Service stations should develop and implement programmes to prevent accidents and incidents which can be attributed to problems associated with service station physical conditions, such as poor maintenance, storage and housekeeping practices. Other factors contributing toward accidents in service stations include employees’ lack of attention, training or skills, which may result in the improper use of equipment, tools, automotive parts, supplies and maintenance materials. Figure 1 provides a safety checklist.
Figure 1. Service station safety and health checklist.
Robberies are a major safety hazard in service stations. Appropriate precautions and training are discussed in the accompanying box and elsewhere in this Encyclopaedia.
Health
Employees should be aware of health hazards associated with working in service stations, such as the following:
Carbon monoxide. Internal combustion engine exhaust gases contain carbon monoxide, a highly toxic, odourless and colourless gas. Employees should be aware of the dangers of exposure to carbon monoxide, particularly when vehicles are inside service bays, garages or car washes with their engines running. Vehicle exhaust gases should be piped outside through flexible hoses, and ventilation should be provided to assure an adequate supply of fresh air. Fuel oil appliances and heaters should be checked to assure that carbon monoxide is not vented to inside areas.
Toxicity of petroleum fuels. Employees who come in contact with gasoline, diesel fuel, heating oil or kerosene should be aware of the potential hazards of exposure and know how to handle these fuels safely. Inhaling sufficient concentrations of petroleum fuel vapours for extended periods of time may result in mild intoxication, anaesthesia or more serious conditions. Short exposure to high concentrations will cause dizziness, headaches and nausea, and irritate the eyes, nose and throat. Gasoline, solvents or fuel oils should never be siphoned from containers or tanks by mouth, since the toxicity of low viscosity liquid hydrocarbons aspired directly into the lungs is 200 times greater than if they are ingested. Aspiration into the lungs may cause pneumonia with extensive pulmonary oedema and haemorrhage, leading to serious injury or death. Vomiting should not be induced. Immediate medical assistance should be sought.
Benzene. Service station employees should be aware of the potential hazards of benzene, which is found in gasoline, and avoid inhaling gasoline vapours. Although gasoline contains benzene, low-level exposure to gasoline vapours is unlikely to cause cancer. Numerous scientific studies have shown that service station employees are not exposed to excessive levels of benzene during the course of their normal work activities; however, there is always the possibility that overexposure could occur.
Dermatitis hazards. Employees who handle and come into contact with petroleum products as part of their jobs should be aware of the hazards of dermatitis and other skin disorders and the personal hygiene and personal protective measures needed to control exposure. If eye contact with gasoline, lubricants or antifreeze occurs, the eyes should be flushed with clean, lukewarm potable water, and medical assistance should be provided.
Lubricants, used motor oil and automotive chemicals. Employees who change oil and other motor vehicle fluids, including antifreeze, should be aware of the hazards and know how to minimize exposure to products such as gasoline in used motor oil, glycol in antifreeze and other contaminants in transmission fluids and gear lubricants by the use of PPE and good hygiene practices. If high-pressure lubricating guns are discharged against an employee’s body, the affected area should be examined immediately to see if petroleum products have penetrated the skin. These injuries cause little pain or bleeding, but involve almost instant separation of the skin tissues and possible deeper damage, which should receive immediate medical attention. The attending physician should be informed of the cause and the product involved in the injury.
Welding. Welding, besides being a fire hazard, can involve exposure to lead pigments from welding on car exteriors, as well as metal fumes and welding gases. Local exhaust ventilation or respiratory protection is needed.
Spray painting and auto body fillers. Spray painting can involve exposure to solvent vapours and pigment particulates (e.g., lead chromate). Auto body fillers often are epoxy or polyester resins and can involve skin and respiratory hazards. Drive-in spray booths for spray painting, local exhaust ventilation and skin and eye protection are recommended while using auto body fillers.
Storage batteries. Batteries contain corrosive electrolyte solutions of sulphuric acid that can cause burns and other injuries to the eyes or skin. Exposure to battery solution should be minimized by the use of PPE, including rubber gloves and eye protection. Employees should immediately flush electrolyte solution from the eyes or skin with clean potable water or eye wash fluid for at least 15 minutes and seek immediate medical attention. Employees should thoroughly wash their hands after servicing batteries and keep their hands away from the face and eyes. Employees should be aware that overcharging batteries can create explosive and toxic quantities of hydrogen gas. Because of the potential harmful effects of exposure to lead, used storage batteries should be properly disposed of or recycled in accordance with government regulations or company policies.
Asbestos. Employees who check and service brakes should be aware of the hazards of asbestos, know how to recognize whether brake shoes contain asbestos and take appropriate protective measures to reduce exposure and contain waste for proper disposal (see figure 2).
Figure 2. Portable enclosure for preventing exposure to asbestos dust from brake drums It is equipped with an enclosed compressed-air gun with a cotton sleeve and is connected to a HEPA vacuum cleaner.
Courtesy of Nilfisk of America, Inc.
Personal protective equipment (PPE)
Injuries to employees may occur from contact with automotive fuels, solvents and chemicals or from chemical burns caused by exposure to battery acids or caustic solutions. Service station employees should be familiar with the need to use and wear PPE such as the following:
- Work shoes with oil- and slip-resistant soles should be worn for general work in service stations, and approved protective-toe safety shoes with oil/slip-resistant soles should be worn where there is a danger of foot injuries due to rolling or falling objects or equipment.
- Safety goggles and respiratory protection should be used for protection against exposures to chemicals, dust or steam, such as when painting or working around batteries and radiators. Industrial safety glasses or face shields with goggles should be worn when the potential exists for exposure to impact materials, such as working with grinders or wire buffers, repairing or mounting tyres, or replacing exhaust systems. Welding glasses should be worn when cutting or welding to prevent flash burns and injuries from particles.
- Impervious gloves, aprons, footwear, face shields and chemical goggles should be worn when handling automotive chemicals and solvents, battery acid and caustic solutions and when cleaning up chemical or fuel spills. Leather work gloves should be worn when handling sharp objects such as broken glass, motor vehicle parts or tyre rims and while emptying trash cans.
- Head protection may be needed when working beneath vehicles in pits or changing overhead signage or lights and in other areas where a potential exists for injury to the head.
- Employees working on vehicles should not wear rings, wristwatches, bracelets or long chains, since the jewellery may contact the vehicle’s moving parts or electrical system and cause injury.
To prevent fires, dermatitis or chemical burns to the skin, clothing that is soaked with gasoline, antifreeze or oil should be immediately removed in an area or room with good ventilation and where no sources of ignition, such as electric heaters, engines, cigarettes, lighters or electric hand dryers, are present. The affected areas of the skin should then be thoroughly washed with soap and warm water to remove all traces of contamination. Clothing should be air dried outside or in well-ventilated areas away from sources of ignition before laundering to minimize contamination of wastewater systems.
Service Station Environmental Issues
Storage tank inventory control
Service stations should maintain and reconcile accurate inventory records on all gasoline and fuel oil storage tanks on a regular basis to control losses. Manual stick gauging may be used to provide a check of the integrity of underground storage tanks and connecting pipes. Where automatic gauging or leak detection equipment is installed, its accuracy should be verified regularly by manual stick gauging. Any storage tank or system suspected of leaking should be investigated, and if leakage is detected, the tank should be made safe or emptied and repaired, removed or replaced. Service station employees should be aware that leaking gasoline can travel long distances underground, contaminate water supplies, enter sewer and drainage systems and cause fires and explosions.
Handling and disposal of waste materials
Waste lubricants and automotive chemicals, used motor oil and solvents, spilled gasoline and fuel oil and glycol-type antifreeze solutions should be drained into approved, properly labelled tanks or containers and stored until disposed of or recycled in accordance with government regulations and company policies.
Because engines with worn cylinders or other defects may allow small amounts of gasoline to enter their crankcases, precautions are needed to prevent vapours which could be released from tanks and containers with crankcase drainings from reaching sources of ignition.
Used oil filters and transmission fluid filters should be drained of oil prior to disposal. Used fuel filters which have been removed from vehicles or fuel dispenser pumps should be drained into approved containers and stored in well-ventilated locations away from sources of ignition until dry before disposal.
Used battery-electrolyte containers should be thoroughly rinsed with water before discarding or recycling. Used batteries contain lead and should be properly disposed of or recycled.
Cleaning large spills may require special training and PPE. Recovered spilled fuel may be returned to the terminal or bulk plant or otherwise disposed of according to government regulations or company policy. Lubricants, used oil, grease, antifreeze, spilled fuel and other materials should not be swept, washed or flushed into floor drains, sinks, toilets, sewers, sumps or other drains or the street. Accumulated grease and oil should be removed from floor drains and sumps to prevent these materials from flowing into sewers. Asbestos dust and used asbestos brake linings should be handled and disposed of according to government regulations and company policies. Employees should be aware of the environmental impact and potential health, safety and fire hazards of these wastes.
Industrial Lubricants, Metal Working Fluids and Automotive Oils
The industrial revolution could not have occurred without the development of refined petroleum-based industrial oils, lubricants, cutting oils and greases. Prior to the discovery in the 1860s that a superior lubricant could be produced by distilling crude oil in a vacuum, industry depended on naturally occurring oils and animal fats such as lard and whale sperm oil for lubricating moving parts. These oils and animal products were especially susceptible to melting, oxidation and breakdown from exposure to heat and moisture produced by the steam engines which powered almost all industrial equipment at that time. The evolution of petroleum-based refined products has continued from the first lubricant, which was used to tan leather, to modern synthetic oils and greases with longer service life, superior lubricating qualities and better resistance to change under varying temperatures and climatic conditions.
Industrial Lubricants
All moving parts on machinery and equipment require lubrication. Although lubrication may be provided by dry materials such as Teflon or graphite, which are used in parts such as small electrical motor bearings, oils and greases are the most commonly used lubricants. As the complexity of the machinery increases, the requirements for lubricants and metal process oils become more stringent. Lubricating oils now range from clear, very thin oils used to lubricate delicate instruments, to thick, tar-like oils used on large gears such as those which turn steel mills. Oils with very specific requirements are used both in the hydraulic systems and to lubricate large computer-operated machine tools such as those used in the aerospace industry to produce parts with extremely close tolerances. Synthetic oils, fluids and greases, and blends of synthetic and petroleum-based oils, are used where extended lubricant life is desired, such as sealed-for-life electric motors, where the increased time between oil changes offsets the difference in cost; where extended temperature and pressure ranges exist, such as in aerospace applications; or where it is difficult and expensive to re-apply the lubricant.
Industrial Oils
Industrial oils such as spindle and lubricating oils, gear lubricants, hydraulic and turbine oils and transmission fluids are designed to meet specific physical and chemical requirements and to operate without discernible change for extended periods under varying conditions. Lubricants for aerospace use must meet entirely new conditions, including cleanliness, durability, resistance to cosmic radiation and the ability to operate in extremely cold and hot temperatures, without gravity and in a vacuum.
Transmissions, turbines and hydraulic systems contain fluids which transfer force or power, reservoirs to hold the fluids, pumps to move the fluids from one place to another and auxiliary equipment such as valves, piping, coolers and filters. Hydraulic systems, transmissions and turbines require fluids with specific viscosities and chemical stability to operate smoothly and provide the controlled transfer of power. The characteristics of good hydraulic and turbine oils include a high viscosity index, thermal stability, long life in circulating systems, deposit resistance, high lubricity, anti-foam capabilities, rust protection and good demulsibility.
Gear lubricants are designed to form strong, tenacious films which provide lubrication between gears under extreme pressure. The characteristics of gear oils include good chemical stability, demulsibility and resistance to viscosity increase and deposit formation. Spindle oils are thin, extremely clean and clear oils with lubricity additives. The most important characteristics for way oils—used to lubricate two flat sliding surfaces where there is high pressure and slow speed—are lubricity and tackiness to resist squeezing out and resistance to extreme pressure.
Cylinder and compressor oils combine the characteristics of both industrial and automotive oils. They should resist accumulation of deposits, act as a heat transfer agent (internal combustion engine cylinders), provide lubrication for cylinders and pistons, provide a seal to resist blow-back pressure, have chemical and thermal stability (especially vacuum pump oil), have a high viscosity index and resist water wash (steam-operated cylinders) and detergency.
Automotive Engine Oils
Manufacturers of internal combustion engines and organizations, such as the Society of Automotive Engineers (SAE) in the United States and Canada, have established specific performance criteria for automotive engine oils. Automotive gasoline and diesel engine oils are subjected to a series of performance tests to determine their chemical and thermal stability, corrosion resistance, viscosity, wear protection, lubricity, detergency and high and low temperature performance. They are then classified according to a code system which allows consumers to determine their suitability for heavy-duty use and for different temperatures and viscosity ranges.
Oils for automotive engines, transmissions and gear cases are designed with high viscosity indexes to resist changes in viscosity with temperature changes. Automotive engine oils are especially formulated to resist breakdown under heat as they lubricate internal combustion engines. Internal combustion engine oils must not be too thick to lubricate the internal moving parts when an engine starts up in cold weather, and they must not thin out as the engine heats up when operating. They should resist carbon build-up on valves, rings and cylinders and the formation of corrosive acids or deposits from moisture. Automotive engine oils contain detergents designed to hold carbon and metallic wear particles in suspension so that they can be filtered out as the oil circulates and not accumulate on internal engine parts and cause damage.
Cutting Fluids
The three types of cutting fluids used in industry are mineral oils, soluble oils and synthetic fluids. Cutting oils are typically a blend of high-quality, high-stability mineral oils of various viscosities together with additives to provide specific characteristics depending on the type of material being machined and the work performed. Soluble water-in-oil cutting fluids are mineral oils (or synthetic oils) which contain emulsifiers and special additives including defoamants, rust inhibitors, detergents, bactericides and germicides. They are diluted with water in varying ratios before being used. Synthetic cutting fluids are solutions of non-petroleum-based fluids, additives and water, rather than emulsions, some of which are fire resistant for machining specific metals. Semi-synthetic fluids contain 10 to 15% mineral oil. Some special fluids have both lubricating oil and cutting fluid characteristics due to the tendency of fluids to leak and intermix in certain machine tools such as multi-spindle, automatic screw machines.
The desired characteristics of cutting fluids depend on the composition of the metal being worked on, the cutting tool being used and the type of cutting, planing or shaping operation performed. Cutting fluids improve and enhance the metal working process by cooling and lubrication (i.e., protecting the edge of the cutting tool). For example, when working on a soft metal which creates a lot of heat, cooling is the most important criterion. Improved cooling is provided by using a light oil (such as kerosene) or water-based cutting fluid. Control of the built-up edge on cutting tools is provided by anti-weld or anti-wear additives such as sulphur, chlorine or phosphorus compounds. Lubricity, which is important when working on steel to overcome the abrasiveness of iron sulphide, is provided by synthetic and animal fats or sulphurized sperm oil additives.
Other Metal Working and Process Oils
Grinding fluids are designed to provide cooling and prevent metal build-up on grinding wheels. Their characteristics include thermal and chemical stability, rust protection (soluble fluids), preventing gummy deposits upon evaporation and a safe flashpoint for the work performed.
Quench oils, which require high stability, are used in metal treating to control the change of the molecular structure of steel as it cools. Quenching in lighter oil is used to case harden small, inexpensive steel parts. A slower quench rate is used to produce machine tool steels which are fairly hard on the outside with lower internal stress. A gapped or multi-phase quenching oil is used to treat high carbon and alloy steels.
Roll oils are specially formulated mineral or soluble oils which lubricate and provide a smooth finish to metal, particularly aluminium, copper and brass, as it goes through hot and cold rolling mills. Release oils are used to coat dies and moulds to facilitate the release of the formed metal parts. Tanning oils are still used in the felt and leather-making industry. Transformer oils are specially formulated dielectric fluids used in transformers and large electric breakers and switches.
Heat transfer oils are used in open or closed systems and may last up to 15 years in service. The primary characteristics are good thermal stability as systems operate at temperatures from 150 to 315°C, oxidation stability and high flashpoint. Heat transfer oils are normally too viscous to be pumped at ambient temperatures and must be heated to provide fluidity.
Petroleum solvents are used to clean parts by spraying, dripping or dipping. The solvents remove oil and emulsify dirt and metal particles. Rust preventive oils may be either solvent or water based. They are applied to stainless steel coils, bearings and other parts by dipping or spraying, and leave polarized or wax films on the metal surfaces for fingerprint and rust protection and water displacement.
Greases
Greases are mixtures of fluids, thickeners and additives used to lubricate parts and equipment which cannot be made oil-tight, which are hard to reach or where leaking or splashed liquid lubricants might contaminate products or create a hazard. They have a wide range of applications and performance requirements, from lubricating jet engine bearings at sub-zero temperatures to hot rolling mill gears, and resisting acid or water washout, as well as the continuous friction created by railroad car wheel roller bearings.
Grease is made by the blending of metallic soaps (salts of long-chained fatty acids) into a lubricating oil medium at temperatures of 205 to 315°C. Synthetic greases may use di-esters, silicone or phosphoric esters and polyalkyl glycols as fluids. The characteristics of the grease depend to a great extent upon the particular fluid, metallic element (e.g., calcium, sodium, aluminium, lithium and so on) in the soap and the additives used to improve performance and stability and to reduce friction. These additives include extreme-pressure additives which coat the metal with a thin layer of non-corrosive metallic sulphur compounds, lead naphthenate or zinc dithiophosphate, rust inhibitors, anti-oxidants, fatty acids for added lubricity, tackiness additives, colour dyes for identification and water inhibitors. Some greases may contain graphite or molybdenum fillers which coat the metallic parts and provide lubrication after the grease has run out or decomposed.
Industrial Lubricants, Grease and Automotive Engine Oil Additives
In addition to using high-quality lubricant base stocks with chemical and thermal stability and high viscosity indexes, additives are needed to enhance the fluid and provide specific characteristics required in industrial lubricants, cutting fluids, greases and automotive engine oils. The most commonly used additives include but are not limited to the following:
- Anti-oxidants. Oxidation inhibitors, such as 2,6-ditertiary butyl, paracresol and phenyl naphthylamine, reduce the rate of deterioration of oil by breaking up the long-chain molecules which form when exposed to oxygen. Oxidation inhibitors are used to coat metals such as copper, zinc and lead to prevent contact with the oil so they will not act as catalysts, speeding up oxidation and forming acids which attack other metals.
- Foam inhibitors. Defoamants, such as silicones and polyorganic silioxanes, are used in hydraulic oils, gear oils, transmission fluids and turbine oils to reduce surface film tension and remove air entrapped in the oil by pumps and compressors, in order to maintain constant hydraulic pressure and prevent cavitation.
- Corrosion inhibitors. Anti-rust additives, such as lead naphthenate and sodium sulphonate, are used to prevent rust from forming on metallic parts and systems where circulating oil has been contaminated with water or by moist air which entered system reservoirs as they cooled down when the equipment or machinery was not in use.
- Anti-wear additives. Anti-wear additives, such as tricresylphosphate, form polar compounds which are attracted to metal surfaces and provide a physical layer of additional protection in the event that the oil film is not sufficient.
- Viscosity index improvers. Viscosity index improvers help oils resist the effects of temperature changes. Unfortunately, their effectiveness diminishes with extended use. Synthetic oils are designed with very high viscosity indexes, allowing them to maintain their structure over wider temperature ranges and for much longer periods of time than mineral oils with viscosity index improver additives.
- Demulsifiers. Water inhibitors and special compounds separate water out of oil and prevent gum formation; they contain waxy oils which provide added lubricity. They are used where equipment is subject to water wash or where a large amount of moisture is present, such as in steam cylinders, air compressors and gear cases contaminated by soluble cutting fluids.
- Colour dyes. Dyes are used to assist users to identify different oils used for specific purposes, such as transmission fluids and gear oils, in order to prevent misapplication.
- Extreme pressure additives. Extreme pressure additives, such as non-corrosive sulphurized fatty compounds, zinc dithiophosphate and lead naphthenate, are used in automotive, gear and transmission oils to form coatings which protect metal surfaces when the protective oil film thins or is squeezed out and cannot prevent metal to metal contact.
- Detergents. Metal sulphonate and metal phenate detergents are used to hold dirt, carbon and metallic wear particles in suspension in hydraulic oils, gear oils, engine oils and transmission fluids. These contaminants are typically removed when the oil passes through a filter to prevent their being recirculated through the system where they could cause damage.
- Tackiness additives. Adhesive or tackiness additives are used to enable oils to adhere to and resist leakage from bearing assemblies, gear cases, large open gears on mills and construction equipment, and overhead machinery. Their tackiness diminishes with extended service.
- Emulsifiers. Fatty acids and fatty oils are used as emulsifiers in soluble oils to help form solutions with water.
- Lubricity additives. Fat, lard, tallow, sperm and vegetable oils are used to provide a higher degree of oiliness in cutting oils and some gear oils.
- Bactericides. Bactericides and germicides, such as phenol and pine oil, are added to soluble cutting oils to prolong the life of the fluid, maintain stability, reduce odours and prevent dermatitis.
Manufacturing Industrial Lubricants and Automotive Oils
Industrial lubricants and oils, grease, cutting fluids and automotive engine oils are manufactured in blending and packaging facilities, also called “lube plants” or “blending plants”. These facilities may be located either in or adjacent to refineries which produce lubricant base stocks, or they may be some distance away and receive the base stocks by marine tankers or barges, railroad tank cars or tank trucks. Blending and packaging plants blend and compound additives into lubricating oil base stocks to manufacture a wide range of finished products, which are then shipped in bulk or in containers.
The blending and compounding processes used to manufacture lubricants, fluids and greases depend on the age and sophistication of the facility, the equipment available, the types and formulation of the additives used and the variety and volume of products produced. Blending may require only physical mixing of base stocks and additive packages in a kettle using mixers, paddles or air agitation, or auxiliary heat from electric or steam coils may be needed to help dissolve and blend in the additives. Other industrial fluids and lubricants are produced automatically by mixing base stocks and pre-blended additive and oil slurries through manifold systems. Grease may be either batch produced or continuously compounded. Lube plants may compound their own additives from chemicals or purchase pre-packaged additives from specialty companies; a single plant may use both methods. When lube plants manufacture their own additives and additive packages, there may be a need for high temperatures and pressures in addition to chemical reactions and physical agitation to compound the chemicals and materials.
After production, fluids and lubricants may be held in the blending kettles or placed in holding tanks to ensure that the additives remain in suspension or solution, to allow time for testing to determine whether the product meets quality specifications and certification requirements, and to allow process temperatures to return to ambient levels before products are packaged and shipped. When testing is completed, finished products are released for bulk shipment or packaging into containers.
Finished products are shipped in bulk in railroad tank cars or in tank trucks directly to consumers, distributors or outside packaging plants. Finished products also are shipped to consumers and distributors in railroad box cars or package delivery trucks in a variety of containers, as follows:
- Metal, plastic and combination metal/plastic or plastic/fibre intermediate bulk containers, which range in size from 227 l to approximately 2,840 l, are shipped as individual units on built-in or separate pallets, stacked 1 or 2 high.
- Metal, fibre or plastic drums with a capacity of 208 l, 114 l or 180 kg are typically shipped 4 to a pallet.
- Metal or plastic drums with a capacity of 60 l or 54 kg, and 19 l or 16 kg metal or plastic pails, are stacked on pallets and banded or stretch wrapped to maintain stability.
- Metal or plastic containers with a capacity of 8 l or 4 l, 1 l plastic, metal and fibre bottles and cans and 2 kg grease cartridges are packaged in cartons which are stacked on pallets and banded or stretch wrapped for shipment.
Some blending and packaging plants may ship pallets of mixed products and mixed sizes of containers and packages directly to small consumers. For example, a single-pallet shipment to a service station could include 1 drum of transmission fluid, 2 kegs of grease, 8 cases of automotive engine oil and 4 pails of gear lubricant.
Product Quality
Lubricant product quality is important to keep machines and equipment operating properly and to produce quality parts and materials. Blending and packaging plants manufacture finished petroleum products to strict specifications and quality requirements. Users should maintain the level of quality by establishing safe practices for the handling, storage, dispensing and transfer of lubricants from their original containers or tanks to the dispensing equipment and to the point of application on the machine or equipment to be lubricated or the system to be filled. Some industrial facilities have installed centralized dispensing, lubrication and hydraulic systems which minimize contamination and exposure. Industrial oils, lubricants, cutting oils and grease will deteriorate from water or moisture contamination, exposure to excessively high or low temperatures, inadvertent mixing with other products and long-term storage which allows additive drop-out or chemical changes to occur.
Health and Safety
Because they are used and handled by consumers, finished industrial and automotive products must be relatively free of hazards. There is a potential for hazardous exposures when blending and compounding products, when handling additives, when using cutting fluids and when operating oil mist lubrication systems.
The chapter Oil and natural gas refineries in this Encyclopaedia gives information regarding potential hazards associated with auxiliary facilities at blending and packaging plants such as boiler rooms, laboratories, offices, oil-water separators and waste treatment facilities, marine docks, tank storage, warehouse operations, railroad tank car and tank truck loading racks and railroad box car and package truck loading and unloading facilities.
Safety
Manufacturing additives and slurries, batch compounding, batch blending and in-line blending operations require strict controls to maintain desired product quality and, along with the use of PPE, to minimize exposure to potentially hazardous chemicals and materials as well as contact with hot surfaces and steam. Additive drums and containers should be stored safely and kept tightly sealed until ready for use. Additives in drums and bags need to be handled properly to avoid muscular strain. Hazardous chemicals should be properly stored, and incompatible chemicals should not be stored where they can mix with one another. Precautions to be taken when operating filling and packaging machinery include using gloves and avoiding catching fingers in devices which crimp covers on kegs and pails. Machine guards and protective systems should not be removed, disconnected or by-passed to expedite work. Intermediate bulk containers and drums should be inspected before filling to make sure they are clean and suitable.
A confined-space permit system should be established for entry into storage tanks and blending kettles for cleaning, inspection, maintenance or repair. A lockout/tagout procedure should be established and implemented before working on packaging machinery, blending kettles with mixers, conveyors, palletizers and other equipment with moving parts.
Leaking drums and containers should be removed from the storage area and spills cleaned up to prevent slips and falls. Recycling, burning and disposal of waste, spilled and used lubricants, automotive engine oils and cutting fluids should be in accordance with government regulations and company procedures. Workers should use appropriate PPE when cleaning spills and handling used or waste products. Drained motor oil, cutting fluids or industrial lubricants which may be contaminated with gasoline and flammable solvents should be stored in a safe place away from sources of ignition, until proper disposal.
Fire protection
While the potential for fire is less in industrial and automotive lubricant blending and compounding than in refining processes, care must be taken when manufacturing metal working oils and greases due to the use of high blending and compounding temperatures and lower flashpoint products. Special precautions should be taken to prevent fires when products are dispensed or containers filled at temperatures above their flashpoints. When transferring flammable liquids from one container to another, proper bonding and grounding techniques should be applied to prevent static build-up and electrostatic discharge. Electrical motors and portable equipment should be properly classified for the hazards present in the area in which they are installed or used.
The potential for fire exists if a leaking product or vapour release in the lube blending and grease processing or storage areas reaches a source of ignition. The establishment and implementation of a hot-work permit system should be considered to prevent fires in blending and packaging facilities. Storage tanks installed inside buildings should be constructed, vented and protected in accordance with government requirements and company policy. Products stored on racks and in piles should not block fire protection systems, fire doors or exit routes.
Storage of finished products, both in bulk and in containers and packages, should be in accordance with recognized practices and fire prevention regulations. For example, flammable liquids and additives which are in solutions of flammable liquids may be stored in outside buildings or separate, specially designed inside or attached storage rooms. Many additives are stored in warm rooms (38 to 65°C) or in hot rooms (over 65°C) in order to keep the ingredients in suspension, to reduce the viscosity of thicker products or to provide for easier blending or compounding. These storage rooms should comply with electrical classification, drainage, ventilation and explosion venting requirements, especially when flammable liquids or combustible liquids are stored and dispensed at temperatures above their flashpoints.
Health
When blending, sampling and compounding, personal and respiratory protective equipment should be considered to prevent exposures to heat, steam, dusts, mists, vapours, fumes, metallic salts, chemicals and additives. Safe work practices, good hygiene and appropriate personal protection may be needed for exposure to oil mists, fumes and vapours, additives, noise and heat when conducting inspection and maintenance activities while sampling and handling hydrocarbons and additives during the production and packaging and when cleaning up spills and releases:
- Work shoes with oil- or slip-resistant soles should be worn for general work, and approved protective toe safety shoes with oil- or slip-resistant soles should be worn where hazards of foot injuries from rolling or falling objects or equipment exist.
- Safety goggles and respiratory protection may be needed for hazardous exposures to chemicals, dust or steam.
- Impervious gloves, aprons, footwear, face shields and chemical goggles should be worn when handling hazardous chemicals, additives and caustic solutions and when cleaning up spills.
- Head protection may be needed when working in pits or areas where the potential exists for injury to the head.
- Ready access to appropriate cleaning and drying facilities to handle splashes and spills should be provided.
Oil is a common cause of dermatitis, which can be controlled through the use of PPE and good personal hygiene practices. Direct skin contact with any formulated greases or lubricants should be avoided. Lighter oils such as kerosene, solvents and spindle oils defat the skin and cause rashes. Thicker products, such as gear oils and greases, block the pores of the skin, leading to folliculitis.
Health hazards due to microbial contamination of oil may be summarized as follows:
- Pre-existing skin conditions may be aggravated.
- Lubricant aerosols of respirable size may cause respiratory illness.
- Organisms may change the composition of the product so that it becomes directly injurious.
- Harmful bacteria from animals, birds or humans may be introduced.
Contact dermatitis may occur when employees are exposed to cutting fluids during production, work or maintenance and when they wipe oil-covered hands with rags embedded with minute metal particles. The metal causes small lacerations in the skin which may become infected. Water-based cutting fluids on skin and clothing may contain bacteria and cause infections, and the emulsifiers may dissolve fats from the skin. Oil folliculitis is caused by prolonged exposure to oil-based cutting fluids, such as from wearing oil-soaked clothing. Employees should remove and launder clothing that is soaked with oil before wearing it again. Dermatitis may also be caused by using soaps, detergents or solvents to clean the skin. Dermatitis is best controlled by good hygiene practices and minimizing exposure. Medical advice should be sought when dermatitis persists.
In the extensive review conducted as a basis for its criteria document, the US National Institute for Occupational Safety and Health (NIOSH) found an association between exposure to metal working fluids and the risk of developing cancer at several organ sites, including the stomach, pancreas, larynx and rectum (NIOSH 1996). The specific formulations responsible for the elevated cancer risks remain to be determined.
Occupational exposure to oil mists and aerosols is associated with a variety of non-malignant respiratory effects, including lipoid pneumonia, asthma, acute airways irritation, chronic bronchitis and impaired pulmonary function (NIOSH 1996).
Metal working fluids are readily contaminated by bacteria and fungi. They may affect the skin or, when inhaled as contaminated aerosols, they may have systemic effects.
Refinery processes such as hydrofinishing and acid treatment are used to remove aromatics from industrial lubricants, and the use of naphthenic base stocks has been restricted in order to minimize carcinogenicity. Additives introduced in blending and compounding may also create a potential risk to health. Exposures to chlorinated compounds and leaded compounds, such as those used in some gear lubricants and greases, cause irritation of the skin and may be potentially hazardous. Tri-orthocresyl phosphate has caused outbreaks of nerve palsies when lubricating oil was accidentally used for cooking. Synthetic oils consist mainly of sodium nitrite and triethanolamine and additives. Commercial triethanolamine contains diethanolamine, which can react with sodium nitrite to form a relatively weak carcinogen, N-nitrosodiethanolamine, which may create a hazard. Semi-synthetic lubricants present the hazards of both products, as well as the additives in their formulations.
Product safety information is important to employees of both manufacturers and users of lubricants, oils and greases. Manufacturers should have material safety data sheets (MSDSs) or other product information available for all of the additives and base stocks used in blending and compounding. Many companies have conducted epidemiological and toxicological testing to determine the degree of hazards associated with any acute and chronic health effects of their products. This information should be available to workers and users through warning labels and product safety information.
Exploration, Drilling and Production of Oil and Natural Gas
General Profile
Crude oils and natural gases are mixtures of hydrocarbon molecules (organic compounds of carbon and hydrogen atoms) containing from 1 to 60 carbon atoms. The properties of these hydrocarbons depend on the number and arrangement of the carbon and hydrogen atoms in their molecules. The basic hydrocarbon molecule is 1 carbon atom linked with 4 hydrogen atoms (methane). All other variations of petroleum hydrocarbons evolve from this molecule. Hydrocarbons containing up to 4 carbon atoms are usually gases; those with 5 to 19 carbon atoms are usually liquids; and those with 20 or more are solids. In addition to hydrocarbons, crude oils and natural gases contain sulphur, nitrogen and oxygen compounds together with trace quantities of metals and other elements.
Crude oil and natural gas are believed to have been formed over millions of years by the decay of vegetation and marine organisms, compressed under the weight of sedimentation. Because oil and gas are lighter than water, they rose up to fill the voids in these overlying formations. This upward movement stopped when the oil and gas reached dense, overlying, impervious strata or nonporous rock. The oil and gas filled the spaces in porous rock seams and natural underground reservoirs, such as saturated sands, with the lighter gas on top of the heavier oil. These spaces were originally horizontal, but shifting of the earth’s crust created pockets, called faults, anticlines, salt domes and stratigraphic traps, where the oil and gas collected in reservoirs.
Shale Oil
Shale oil, or kerogen, is a mixture of solid hydrocarbons and other organic compounds containing nitrogen, oxygen and sulphur. It is extracted, by heating, from a rock called oil shale, yielding from 15 to 50 gallons of oil per ton of rock.
Exploration and production is the common terminology applied to that portion of the petroleum industry which is responsible for exploring for and discovering new crude oil and gas fields, drilling wells and bringing the products to the surface. Historically, crude oil, which had naturally seeped to the surface, was collected for use as medicine, protective coatings and fuel for lamps. Natural gas seepage was recorded as fires burning on the surface of the earth. It was not until 1859 that methods of drilling and obtaining large commercial quantities of crude oil were developed.
Crude oil and natural gas are found throughout the world, beneath both land and water, as follows:
- Western Hemisphere Intercontinental Basin (US Gulf Coast, Mexico, Venezuela)
- Middle East (Arabian Peninsula, Persian Gulf, Black and Caspian Seas)
- Indonesia and South China Sea
- North and West Africa (Sahara and Nigeria)
- North America (Alaska, Newfoundland, California and Mid-continent United States and Canada)
- Far East (Siberia and China)
- North Sea.
Figure 1 and figure 2 show world crude oil and natural gas production for 1995.
Figure 1. World crude oil production for 1995
Figure 2. World natural gas plant liquids production - 1995
The names of crude oils often identify both the type of crude and areas where they were originally discovered. For example, the first commercial crude oil, Pennsylvania Crude, is named after its place of origin in the United States. Other examples are Saudi Light and Venezuelan Heavy. Two benchmark crudes used to set world crude prices are Texas Light Sweet and North Sea Brent.
Classification of crude oils
Crude oils are complex mixtures containing many different, individual hydrocarbon compounds; they differ in appearance and composition from one oil field to another, and sometimes are even different from wells relatively near one another. Crude oils range in consistency from watery to tar-like solids, and in colour from clear to black. An “average” crude oil contains about 84% carbon; 14% hydrogen; 1 to 3% sulphur; and less than 1% of nitrogen, oxygen, metals and salts. See table 1 and table 2.
Table 1. Typical approximate characteristics and properties and gasoline potential of various typical crude oils.
|
Crude source and name * |
Paraffins |
Aromatics |
Naphthenes |
Sulphur |
API gravity |
Naphthene yield |
Octane number |
|
Nigerian Light |
37 |
9 |
54 |
0.2 |
36 |
28 |
60 |
|
Saudi Light |
63 |
19 |
18 |
2 |
34 |
22 |
40 |
|
Saudi Heavy |
60 |
15 |
25 |
2.1 |
28 |
23 |
35 |
|
Venezuela Heavy |
35 |
12 |
53 |
2.3 |
30 |
2 |
60 |
|
Venezuela Light |
52 |
14 |
34 |
1.5 |
24 |
18 |
50 |
|
USA Midcontinental Sweet |
— |
— |
— |
0.4 |
40 |
— |
— |
|
USA West Texas Sour |
46 |
22 |
32 |
1.9 |
32 |
33 |
55 |
|
North Sea Brent |
50 |
16 |
34 |
0.4 |
37 |
31 |
50 |
* Representative average numbers.
Table 2. Composition of crude oil and natural gas
Hydrocarbons
Paraffins: The paraffinic saturated chain type hydrocarbon (aliphatic) molecules in crude oil have the formula CnH2n+2, and can be either straight chains (normal) or branched chains (isomers) of carbon atoms. The lighter, straight chain paraffin molecules are found in gases and paraffin waxes. The branched chain paraffins are usually found in heavier fractions of crude oil and have higher octane numbers than normal paraffins.
Aromatics: Aromatics are unsaturated ring type hydrocarbon (cyclic) compounds. Naphthalenes are fused double ring aromatic compounds. The most complex aromatics, polynuclears (three or more fused aromatic rings), are found in heavier fractions of crude oil.
Naphthenes: Naphthenes are saturated ring type hydrocarbon groupings, with the formula
CnH2n, arranged in the form of closed rings (cyclic), found in all fractions of crude oil except the very lightest. Single ring naphthenes (mono-cycloparaffins) with 5 and 6 carbon atoms predominate, with two ring naphthenes (dicycloparaffins) found in the heavier ends of naphtha.
Non-hydrocarbons
Sulphur and sulphur compounds: Sulphur is present in natural gas and crude oil as hydrogen sulphide (H2S), as compounds (thiols, mercaptans, sulphides, polysulphides, etc.) or as elemental sulphur. Each gas and crude oil has different amounts and types of sulphur compounds, but as a rule the proportion, stability and complexity of the compounds are greater in heavier crude oil fractions.
Sulphur compounds called mercaptans, which exhibit distinct odours detectable at very low concentrations, are found in gas, petroleum crude oils and distillates. The most common are methyl and ethyl mercaptans. Mercaptans are often added to commercial gas (LNG and LPG) to provide an odour for leak detection.
The potential for exposure to toxic levels of H2S exists when working in drilling, production, transportation and processing crude oil and natural gas. The combustion of petroleum hydrocarbons containing sulphur produces undesirables such as sulphuric acid and sulphur dioxide.
Oxygen compounds: Oxygen compounds, such as phenols, ketones and carboxylic acids, are found in crude oils in varying amounts.
Nitrogen compounds: Nitrogen is found in lighter fractions of crude oil as basic compounds, and more often in heavier fractions of crude oil as non-basic compounds which may also include trace metals.
Trace metals: Trace amounts, or small quantities of metals, including copper, nickel, iron, arsenic and vanadium, are often found in crude oils in small quantities.
Inorganic salts: Crude oils often contain inorganic salts, such as sodium chloride, magnesium chloride and calcium chloride, suspended in the crude or dissolved in entrained water (brine).
Carbon dioxide: Carbon dioxide may result from the decomposition of bicarbonates present in, or added to crude, or from steam used in the distillation process.
Naphthenic acids: Some crude oils contain naphthenic (organic) acids, which may become corrosive at temperatures above 232 °C when the acid value of the crude is above a certain level.
Normally occurring radioactive materials: Normally occurring radioactive materials (NORMs) are often present in crude oil, in the drilling deposits and in the drilling mud, and can present a hazard from low levels of radioactivity.
Relatively simple crude-oil assays are used to classify crude oils as paraffinic, naphthenic, aromatic or mixed, based on the predominant proportion of similar hydrocarbon molecules. Mixed-base crudes have varying amounts of each type of hydrocarbon. One assay method (US Bureau of Mines) is based on distillation, and another method (UOP “K” factor) is based on gravity and boiling points. More comprehensive crude assays are conducted to determine the value of the crude (i.e., its yield and quality of useful products) and processing parameters. Crude oils are usually grouped according to yield structure, with high-octane gasoline being one of the more desirable products. Refinery crude oil feedstocks usually consist of mixtures of two or more different crude oils.
Crude oils are also defined in terms of API (specific) gravity. For example, heavier crude oils have low API gravities (and high specific gravities). A low-API gravity crude oil may have either a high or low flashpoint, depending on its lightest ends (more volatile constituents). Because of the importance of temperature and pressure in the refining process, crude oils are further classified as to viscosity, pour points and boiling ranges. Other physical and chemical characteristics, such as colour and carbon residue content, are also considered. Crude oils with high carbon, low hydrogen and low API gravity are usually rich in aromatics; while those with low carbon, high hydrogen and high API gravity are usually rich in paraffins.
Crude oils which contain appreciable quantities of hydrogen sulphide or other reactive sulphur compounds are called “sour.” Those with less sulphur are called “sweet.” Some exceptions to this rule are West Texas crudes (which are always considered “sour” regardless of their H2S content) and Arabian high-sulphur crudes (which are not considered “sour” because their sulphur compounds are not highly reactive).
Compressed Natural Gas and Liquefied Hydrocarbon Gases
The composition of naturally occurring hydrocarbon gases is similar to crude oils in that they contain a mixture of different hydrocarbon molecules depending on their source. They can be extracted as natural gas (almost free of liquids) from gas fields; petroleum-associated gas which is extracted with oil from gas and oil fields; and gas from gas condensate fields, where some of the liquid components of oil convert into the gaseous state when pressure is high (10 to 70 mPa). When the pressure is decreased (to 4 to 8 mPa) condensate containing heavier hydrocarbons separates from the gas by condensation. Gas is extracted from wells reaching up to 4 miles (6.4 km) or more in depth, with seam pressures varying from 3 mPa up to as high as 70 mPa. (See figure 3.)
Figure 3. Offshore natural gas well set in 87.5 metres of water in the Pitas Point area of the Santa Barbara Channel, Southern California
American Petroleum Institute
Natural gas contains 90 to 99% hydrocarbons, which consist predominately of methane (the simplest hydrocarbon) together with smaller amounts of ethane, propane and butane. Natural gas also contains traces of nitrogen, water vapour, carbon dioxide, hydrogen sulphide and occasional inert gases such as argon or helium. Natural gases containing more than 50 g/m3 of hydrocarbons with molecules of three or more carbon atoms (C3 or higher) are classified as “lean” gases.
Depending how it is used as a fuel, natural gas is either compressed or liquefied. Natural gas from gas and gas condensate fields is processed in the field to meet specific transportation criteria before being compressed and fed into gas pipelines. This preparation includes removal of water with driers (dehydrators, separators and heaters), oil removal using coalescing filters, and the removal of solids by filtration. Hydrogen sulphide and carbon dioxide are also removed from natural gas, so that they do not corrode pipelines and transportation and compression equipment. Propane, butane and pentane, present in natural gas, are also removed before transmission so they will not condense and form liquids in the system. (See the section “Natural gas production and processing operations.”)
Natural gas is transported by pipeline from gas fields to liquefication plants, where it is compressed and cooled to approximately –162 ºC to produce liquefied natural gas (LNG) (see figure 4). The composition of LNG is different from natural gas due to the removal of some impurities and components during the liquefaction process. LNG is primarily used to augment natural gas supplies during peak demand periods and to supply gas in remote areas away from major pipelines. It is regasified by adding nitrogen and air to make it comparable to natural gas before being fed into gas supply lines. LNG is also used as a motor-vehicle fuel as an alternative to gasoline.
Figure 4. World’s largest LNG plant at Arzew, Algeria
American Petroleum Institute
Petroleum-associated gases and condensate gases are classified as “rich” gases, because they contain significant amounts of ethane, propane, butane and other saturated hydrocarbons. Petroleum-associated and condensate gases are separated and liquefied to produce liquefied petroleum gas (LPG) by compression, adsorption, absorption and cooling at oil and gas process plants. These gas plants also produce natural gasoline and other hydrocarbon fractions.
Unlike natural gas, petroleum-associated gas and condensate gas, oil processing gases (produced as by-products of refinery processing) contain considerable amounts of hydrogen and unsaturated hydrocarbons (ethylene, propylene and so on). The composition of oil processing gases depends upon each specific process and the crude oils used. For example, gases obtained as a result of thermal cracking usually contain significant amounts of olefins, while those obtained from catalytic cracking contain more isobutanes. Pyrolysis gases contain ethylene and hydrogen. The composition of natural gases and typical oil processing gases is shown in table 3.
Table 3. Typical approximate composition of natural and oil processing gases (per cent by volume)
|
Type gas |
H2 |
CH4 |
C2H6 |
C3H4 |
C3H8 |
C3H6 |
C4H10 |
C4H8 |
N2+CO2 |
C5+ |
|
Natural gas |
n/a |
98 |
0.4 |
n/a |
0.15 |
n/a |
0.05 |
n/a |
1.4 |
n/a |
|
Petroleum- |
n/a |
42 |
20 |
n/a |
17 |
n/a |
8 |
n/a |
10 |
3 |
|
Oil processing gases |
|
|
|
|
|
|
|
|
|
|
Combustible natural gas, with a calorific value of 35.7 to 41.9 MJ/m3 (8,500 to 10,000 kcal/m3), is primarily used as a fuel to produce heat in domestic, agricultural, commercial and industrial applications. The natural gas hydrocarbon also is used as feedstock for petrochemical and chemical processes. Synthesis gas (CO + H2) is processed from methane by oxygenation or water vapour conversion, and used to produce ammonia, alcohol and other organic chemicals. Compressed natural gas (CNG) and liquefied natural gas (LNG) are both used as fuel for internal combustion engines. Oil processing liquefied petroleum gases (LPG) have higher calorific values of 93.7 MJ/m3 (propane) (22,400 kcal/m3) and 122.9 MJ/m3 (butane) (29,900 kcal/m3) and are used as fuel in homes, businesses and industry as well as in motor vehicles (NFPA 1991). The unsaturated hydrocarbons (ethylene, propylene and so on) derived from oil processing gases may be converted into high-octane gasoline or used as raw materials in the petrochemical and chemical-processing industries.
Properties of Hydrocarbon Gases
According to the US National Fire Protection Association, flammable (combustible) gases are those which burn in the concentrations of oxygen normally present in air. The burning of flammable gases is similar to that of flammable hydrocarbon liquid vapours, as a specific ignition temperature is needed to initiate the burning reaction and each will burn only within a certain defined range of gas-air mixtures. Flammable liquids have a flashpoint (the temperature (always below the boiling point) at which they emit sufficient vapours for combustion). There is no apparent flashpoint for flammable gases, as they are normally at temperatures above their boiling points, even when liquefied, and are therefore always at temperatures well in excess of their flashpoints.
The US National Fire Protection Association (1976) defines compressed and liquefied gases, as follows:
- “Compressed gases are those which at all normal atmospheric temperatures inside their containers, exist solely in the gaseous state under pressure.”
- “Liquefied gases are those which at normal atmospheric temperatures inside their containers, exist partly in the liquid state and partly in the gaseous state, and are under pressure as long as any liquid remains in the container.”
The major factor which determines the pressure inside the vessel is the temperature of the liquid stored. When exposed to the atmosphere, the liquefied gas very rapidly vaporizes, travelling along the ground or water surface unless dispersed into the air by wind or mechanical air movement. At normal atmospheric temperatures, about one-third of the liquid in the container will vaporize.
Flammable gases are further classified as fuel gas and industrial gas. Fuel gases, including natural gas and liquefied petroleum gases (propane and butane), are burned with air to produce heat in ovens, furnaces, water heaters and boilers. Flammable industrial gases, such as acetylene, are used in processing, welding, cutting and heat treating operations. The differences in properties of liquefied natural gas (LNG) and liquefied petroleum gases (LPG) are shown in table 3.
Searching for Oil and Gas
The search for oil and gas requires a knowledge of geography, geology and geophysics. Crude oil is usually found in certain types of geological structures, such as anticlines, fault traps and salt domes, which lie under various terrains and in a wide range of climates. After selecting an area of interest, many different types of geophysical surveys are conducted and measurements performed in order to obtain a precise evaluation of the subsurface formations, including:
- Magnetometric surveys. Magnetometers hung from airplanes measure variations in the earth’s magnetic field in order to locate sedimentary rock formations which generally have low magnetic properties when compared to other rocks.
- Aerial photogrammetric surveys. Photographs taken with special cameras in airplanes, provide three-dimensional views of the earth which are used to determine land formations with potential oil and gas deposits.
- Gravimetric surveys. Because large masses of dense rock increase the pull of gravity, gravimeters are used to provide information regarding underlying formations by measuring minute differences in gravity.
- Seismic surveys. Seismic studies provide information on the general characteristics of the subsurface structure (see figure 5). Measurements are obtained from shock waves generated by setting off explosive charges in small-diameter holes, from the use of vibrating or percussion devices on both land and in water, and from underwater blasts of compressed air. The elapsed time between the beginning of the shock wave and the return of the echo is used to determine the depth of the reflecting substrata. The recent use of super-computers to generate three-dimensional images greatly improves evaluation of seismic test results.
Figure 5. Saudi Arabia, seismic operations
American Petroleum Institute
- Radiographic surveys. Radiography is the use of radio waves to provide information similar to that obtained from seismic surveys.
- Stratigraphic surveys. Stratigraphic sampling is the analysis of cores of subsurface rock strata for traces of gas and oil. A cylindrical length of rock, called a core, is cut by a hollow bit and pushed up into a tube (core barrel) attached to the bit. The core barrel is brought to the surface and the core is removed for analysis.
When the surveys and measurements indicate the presence of formations or strata which may contain petroleum, exploratory wells are drilled to determine whether or not oil or gas is actually present and, if so, whether it is available and obtainable in commercially viable quantities.
Offshore Operations
Although the first offshore oil well was drilled in the early 1900s off of the coast of California, the beginning of modern marine drilling was in 1938, with a discovery in the Gulf of Mexico, 1 mile (1.6 km) from the US coastline. After the Second World War, offshore drilling expanded quickly, first in shallow waters adjacent to known land-based production areas, and then to other shallow and deep water areas around the world, and in climates varying from the Arctic to the Persian Gulf. In the beginning, offshore drilling was possible only in water depths of about 91 m; however, modern platforms are now able drill in waters over 3.2 km deep. Offshore oil activities include exploration, drilling, production, processing, underwater construction, maintenance and repair, and the transport of the oil and gas to shore by ship or pipeline.
Offshore platforms
Drilling platforms support drilling rigs, supplies and equipment for offshore or inland water operations, and range from floating or submergible barges and ships, to fixed-in-place platforms on steel legs used in shallow waters, to large, buoyant, reinforced concrete, gravity-type platforms used in deep waters. After the drilling is completed, marine platforms are used to support production equipment. The very largest production platforms have accommodations for over 250 crew members and other support personnel, heliports, processing plants and crude oil and gas condensate storage capability (see figure 6).
Figure 6. Drilling vessels; drill ship Ben Ocean Laneer
American Petroleum Institute
Typically, with deep water floating platform drilling, the wellhead equipment is lowered to the ocean floor and sealed to the well casing. The use of fibre-optic technology allows a large, central platform to remotely control and operate smaller satellite platforms and sub-sea templates. Production facilities on the large platform process the crude oil, gas and condensate from the satellite facilities, before it is shipped on-shore.
The type of platform used in underwater drilling is often determined by the type of well to be drilled (exploratory or production) and by the depth of the water (see table 4).
Table 4. Platform types for underwater drilling
|
Platform type |
Depth (m) |
Description |
|
Submersible barges and platforms |
15–30 |
Barges or platforms, towed to the site and sunk to rest on the bottom. Lower buoyant column keeps rigs afloat |
|
Jack-ups (on legs) |
30–100 |
Mobile, self-elevating buoyant platforms whose legs are jacked up for towing. At the site, the legs are lowered to |
|
Floating platforms |
100–3,000+ |
Large, self-contained, multi-level, reinforced concrete gravity structures, towed to the site, submerged with |
|
Smaller floating platforms, similarly suspended, which support only the drilling rig and are serviced by a floating |
||
|
Drilling barges |
30–300 |
Self-propelled, floating or semi-submersible barges. |
|
Drill ships |
120–3,500+ |
Highly sophisticated, specially designed, floating or semi-submersible ships. |
|
Fixed on site platforms |
0–250 |
Platforms built on steel supports (jackets) which are sunk and fixed in place, and artificial islands used as |
|
Sub-sea templates |
n/a |
Underwater production installations. |
Types of Wells
Exploratory wells.
Following the analysis of geological data and geophysical surveys, exploratory wells are drilled, either on land or offshore. Exploratory wells which are drilled in areas where neither oil nor gas has been previously found are called “wildcats.” Those wells which strike oil or gas are called “discovery wells.” Other exploratory wells, known as “step-out” or “appraisal” wells, are drilled to determine the limits of a field following discovery, or to search for new oil- and gas-bearing formations next to, or beneath, those already known to contain product. A well which does not find any oil or gas, or finds too little to produce economically, is called a “dry hole”.
Developmental wells.
After a discovery, the area of the reservoir is roughly determined with a series of step-out or appraisal wells. Developmental wells are then drilled to produce gas and oil. The number of developmental wells to be drilled is determined by the expected definition of the new field, both in size and in productivity. Because of the uncertainty as to how reservoirs are shaped or confined, some developmental wells may turn out to be dry holes. Occasionally, drilling and producing occurs simultaneously.
Geopressure/geothermal wells.
Geopressure/geothermal wells are those which produce extremely high-pressure (7,000 psi) and high-temperature (149 ºC) water which may contain hydrocarbons. The water becomes a rapidly expanding cloud of hot steam and vapours upon release to the atmosphere from a leak or rupture.
Stripper wells.
Stripper wells are those which produce less than ten barrels of oil a day from a reservoir.
Multiple completion wells.
When multiple producing formations are discovered when drilling a single well, a separate string of pipe may be run into a single well for each individual formation. Oil and gas from each formation is directed into its respective piping and isolated from one another by packers, which seal the annular spaces between the piping string and the casing. These wells are known as multiple completion wells.
Injection wells.
Injection wells pump air, water, gas or chemicals into reservoirs of producing fields, either to maintain pressure or move oil toward producing wells by hydraulic force or increased pressure.
Service wells.
Service wells include those used for fishing and wire-line operations, packer/plug placement or removal and reworking. Service wells are also drilled for underground disposal of salt water, which is separated from crude oil and gas.
Drilling Methods
Drilling rigs.
Basic drilling rigs contain a derrick (tower), a drilling pipe, a large winch to lower and lift out the drilling pipe, a drilling table which rotates the drilling pipe and bit, a mud mixer and pump and an engine to drive the table and winch (see figure 7). Small drilling rigs used to drill exploratory or seismic wells may be mounted on trucks for movement from site to site. Larger drilling rigs are either erected onsite or have portable, hinged (jack knife) derricks for easy handling and erection.
Figure 7. Drilling rig on Elf Ringnes Island in the Canadian Arctic
American Petroleum Institute
Percussion or cable drilling.
The oldest drilling technique is percussion or cable drilling. This slow, limited depth method, which is seldom used, involves crushing rock by raising and dropping a heavy chisel bit and stem on the end of a cable. At intervals, the bit is removed and the cuttings are suspended in water and removed by flushing or pumping to the surface. As the hole deepens, it is lined with steel casing to prevent cave-in and protect against contamination of groundwater. Considerable work is required to drill even a shallow well, and upon striking oil or gas, there is no way to control the immediate flow of product to the surface.
Rotary drilling.
Rotary drilling is the most common method and is used to drill both exploratory and production wells at depths over 5 miles (7,000 m). Lightweight drills, mounted on trucks, are used to drill low-depth seismic wells on land. Medium and heavy rotary mobile and floating drills are used for drilling exploration and production wells. Rotary drilling equipment is mounted on a drilling platform with a 30- to 40-m-high derrick, and includes a rotary table, engine, mud mixer and injector pump, a wire-line drum hoist or winch, and many sections of pipe, each approximately 27 m long. The rotary table turns a square kelly connected to the drilling pipe. The square kelly has a mud swivel on the top which is connected to blowout preventors. The drill pipe rotates at a speed of from 40 to 250 rpm, turning either a drill which has drag bits with fixed chisel-like cutting edges or a drill whose bit has rolling cutters with hardened teeth.
Rotary percussion drilling.
Rotary percussion drilling is a combination method whereby a rotary drill uses a circulating hydraulic fluid to operate a hammer-like mechanism, thereby creating a series of rapid percussion blows which allow the drill to simultaneously bore and pound into the earth.
Electro and turbo drilling.
Most rotary tables, winches and pumps of heavy drills are usually driven by electric motors or turbines, which allows for increased flexibility in operations and remote-controlled drilling. Electro drill and turbo drill are newer methods which provide more direct power to the drill bit by connecting the drilling motor just above the bit at the bottom of the hole.
Directional drilling.
Directional drilling is a rotary drilling technique which directs the drill string along a curved path as the hole deepens. Directional drilling is used to reach deposits which are inaccessible by vertical drilling. It also reduces costs, as a number of wells can be drilled in different directions from a single platform. Extended-reach drilling allows tapping into undersea reservoirs from the shore. Many of these techniques are possible by using computers to direct automatic drilling machines and flexible pipe (coiled tubing), which is raised and lowered without connecting and disconnecting sections.
Other drilling methods.
Abrasive drilling uses an abrasive material under pressure (instead of using a drill stem and bit) to cut through the substrata. Other drilling methods include explosive drilling and flame piercing.
Abandonment.
When oil and gas reservoirs are no longer productive, the wells are typically plugged with cement to prevent flow or leakage to the surface and to protect the underground strata and water. Equipment is removed and the sites of abandoned wells are cleaned up and returned to normal conditions.
Drilling Operations
Drilling techniques
The drilling platform provides a base for workers to couple and uncouple the sections of drilling pipe which are used to increase the depth of drilling. As the hole deepens, additional lengths of pipe are added and the drilling string is suspended from the derrick. When a drilling bit needs to be changed, the entire drilling string of pipe is pulled out of the hole, and each section is detached and stacked vertically inside the derrick. After the new bit is fitted in place, the process is reversed, and the pipe is returned to the hole to continue drilling.
Care is needed to assure that the drilling string pipe does not split apart and drop into the hole, as it may be difficult and costly to fish out and may even result in the loss of the well. Another potential problem is if drilling tools stick in the hole when drilling stops. For this reason, once drilling begins, it usually continues until the well is completed.
Drilling mud
Drilling mud is a fluid composed of water or oil and clay with chemical additives (e.g., formaldehyde, lime, sodium hydrazide, barite). Caustic soda is often added to control the pH (acidity) of drilling mud and to neutralize potentially hazardous mud additives and completion fluids. Drilling mud is pumped into the well under pressure from the mixing tank on the drilling platform, down the inside of the drilling pipe to the drill bit. It then rises between the outside of the drill pipe and the sides of the hole, returning to the surface, where it is filtered and recirculated.
Drilling mud is used to cool and lubricate the drilling bit, lubricate the pipe and flush the rock cuttings from the drill hole. Drilling mud is also used to control flow from the well by lining the sides of the hole and resisting the pressure of any gas, oil or water which is met by the drill bit. Jets of mud may be applied under pressure at the bottom of the hole to aid in drilling.
Casing and cementation
The casing is a special heavy steel pipe which lines the well hole. It is used to prevent cave-in of the drill hole walls and protect fresh water strata by preventing leakage from the returning flow of mud during drilling operations. The casing also seals off water-permeated sands and high-pressure gas zones. Casing is initially used near the surface and is cemented into place to guide the drill pipe. A cement slurry is pumped down the drilling pipe and forced back up through the gap between the casing and the walls of the well hole. Once the cement sets and the casing is place, drilling continues using a smaller diameter bit.
After the surface casing is placed in the well, blowout preventors (large valves, bags or rams) are attached to the top of the casing, in what is called a stack. Following discovery of oil or gas, casing is set into the bottom of the well to keep dirt, rocks, salt water and other contaminants out of the well hole and to provide a conduit for the crude oil and gas extraction lines.
Completion, Enhanced Recovery and Workover Operations
Completion
Completion describes the process of bringing a well into production after the well has been drilled to the depth where oil or gas is expected to be found. Completion involves a number of operations, including penetration of the casing and cleaning out water and sediment from the pipeline so that flow is unimpeded. Special core bits are used to drill and extract cores up to 50 m long for analysis during the drilling operation to determine when penetration should be performed. The drill pipe and bit are first removed and the final string of casing is cemented into place. A perforating gun, which is a metal tube containing sockets holding either bullets or shaped explosive charges, is then lowered into the well. The charges are discharged by electrical impulse through the casing into reservoir to create openings for the oil and gas to flow into the well and to the surface.
The flow of crude oil and natural gas is controlled by a series of valves, called “Christmas trees”, which are placed at the top of the well head. Monitors and controls are installed to automatically or manually operate surface and subsurface safety valves, in the event of a change in pressure, fire or other hazardous condition. Once the oil and gas are produced they are separated, and water and sediment are removed from the crude oil.
Crude oil and gas production and conservation
Producing oil is basically a matter of displacement by either water or gas. At the time of initial drilling, almost all crude oil is under pressure. This natural pressure decreases as oil and gas is removed from the reservoir, during the three phases of a reservoir’s life.
- During the first phase, flush production, the flow is governed by the natural pressure in the reservoir which comes from dissolved gas in the oil, gas trapped under pressure above the oil and hydraulic pressure from water trapped under the oil.
- Artificial lift, the second phase, involves pumping pressurized gas into the reservoir when the natural pressure is expended.
- Phase three, stripper or marginal production, occurs when wells only produce intermittently.
Originally there was little understanding of the forces which affected oil and gas production. The study of oil and gas reservoir behaviour began at the beginning of the 20th century, when it was discovered that pumping water into a reservoir increased production. At that time, the industry was recovering between 10 and 20% of reservoir capacity, as compared to recent recovery rates of over 60% before wells become unproductive. The concept of control is that a faster rate of production more quickly dissipates the pressure in the reservoir, thereby reducing the total amount of oil which can be eventually recovered. Two measures used to conserve petroleum reservoirs are unitization and well spacing.
- Unitization is the operation of a field as one unit in order to apply secondary recovery methods and maintain pressure, even through a number of different operators may be involved. The total production is allocated on an equitable basis among the operators.
- Well spacing is the limiting and proper location of wells so as to achieve maximum production without dissipating a field due to overdrilling.
Methods of Recovering Additional Product
Productivity of oil and gas reservoirs is improved by a variety of recovery methods. One method is either to chemically or physically open passages in the strata to allow oil and gas to move more freely through reservoirs to the well. Water and gas are injected into reservoirs to maintain working pressure by natural displacement. Secondary recovery methods, including displacement by pressure, artificial lift and flooding, improve and restore reservoir pressure. Enhanced recovery is the use of various secondary recovery methods in multiple and different combinations. Enhanced recovery also includes more advanced methods of obtaining additional product from depleted reservoirs, such as thermal recovery, which uses heat instead of water or gas to force more crude oil out of reservoirs.
Acidizing
Acidizing is a method of increasing the output of a well by pumping acid directly into a producing reservoir to open flow channels through the reaction of chemicals and minerals. Hydrochloric (or regular) acid, was first used to dissolve limestone formations. It is still most commonly used; however, various chemicals are now added to the hydrochloric acid to control its reaction and to prevent corrosion and formation of emulsions.
Hydrofluoric acid, formic acid and acetic acid are also used, together with hydrochloric acid, depending on the type of rock or minerals in the reservoir. Hydrofluoric acid is always combined with one of the other three acids, and was originally used to dissolve sandstone. It is often called “mud acid”, as it is now used to clean perforations which have been plugged with drilling mud and to restore damaged permeability near the well hole. Formic and acetic acids are used in deep, ultra-hot limestone and dolomite reservoirs and as breakdown acids prior to perforation. Acetic acid is also added to wells as a neutralizing buffer agent to control the pH of well stimulation fluids. Almost all acids have additives, such as inhibitors to prevent reaction with the metal casings and surfactants to prevent formation of sludge and emulsions.
Fracturing
Fracturing describes the method used to increase the flow of oil or gas through a reservoir and into wells by force or pressure. Production may decrease because the reservoir formation is not permeable enough to allow the oil to flow freely toward the well. Fracturing forces open underground channels by pumping a fluid treated with special propping agents (including sand, metal, chemical pellets and shells) into the reservoir under high pressure to open fissures. Nitrogen may be added to the fluid to stimulate expansion. When the pressure is released, the fluid withdraws and the propping agents remain in place, holding the fissures open so that oil can flow more freely.
Massive fracturing (mass frac) involves pumping large amounts of fluid into wells to hydraulically create fissures which are thousands of feet in length. Massive fracturing is typically used to open gas wells where the reservoir formations are so dense that even gas cannot pass through them.
Pressure maintenance
Two common pressure maintenance techniques are the injection of water and gas (air, nitrogen, carbon dioxide and natural gas) into reservoirs where natural pressures are reduced or insufficient for production. Both methods require drilling auxiliary injection wells at designated locations to achieve the best results. The injection of water or gas to maintain the working pressure of the well is called natural displacement. The use of pressurized gas to increase the pressure in the reservoir is called artificial (gas) lift.
Water flooding
The most commonly used secondary enhanced recovery method is pumping water into an oil reservoir to push product toward producing wells. In five-spot water flooding, four injection wells are drilled to form a square with the producing well at the center. The injection is controlled to maintain an even advance of the water front through the reservoir toward the producing well. Some of the water used is salt water, obtained from the crude oil. In low-tension water flooding, a surfactant is added to the water to assist the flow of oil through the reservoir by reducing its adhesion to rock.
Miscible flooding
Miscible fluid and miscible polymer flooding are enhanced recovery methods used to improve water injection by reducing the surface tension of crude oil. A fluid miscible (one that can be dissolved in the crude) is injected into a reservoir. This is followed by an injection of another fluid which pushes the crude and miscible fluid mixture toward the producing well. Miscible polymer flooding involves the use of a detergent to wash the crude oil from the strata. A gel or thickened water is injected behind the detergent to move the crude toward the producing well.
Fire flooding
Fire flooding, or in situ (in place) combustion, is an expensive thermal recovery method wherein large quantities of air or oxygen-containing gas is injected into the reservoir and a portion of the crude oil is ignited. The heat from the fire reduces the viscosity of the heavy crude oil so that it flows more easily. Hot gases, produced by the fire, increase the pressure in the reservoir and create a narrow burning front which pushes the thinner crude from the injection well to the producing well. The heavier crude remains in place, providing additional fuel as the flame front moves slowly forward. The burning process is closely monitored and controlled by regulating the injected air or gas.
Steam injection
Steam injection, or steam flooding, is a thermal recovery method which heats heavy crude oil and lowers its viscosity by injecting super-hot steam into the lowest stratum of relatively shallow reservoir. The steam is injected over a period of 10 to 14 days, and the well is shut for another week or so to allow the steam to thoroughly heat the reservoir. At the same time the increased heat expands reservoir gases, thereby increasing the pressure in the reservoir. The well is then reopened and the heated, less viscous crude flows up into the well. A newer method injects low-heat steam at lower pressure into larger sections of two, three or more zones simultaneously, developing a “steam chest” which squeezes down the oil in each of the zones. This provides a greater flow of oil to the surface, while using less steam.
Natural Gas Production and Processing Operations
There are two types of wells producing natural gas. Wet gas wells produce gas which contains dissolved liquids, and dry gas wells produce gas which cannot be easily liquefied
After natural gas is withdrawn from producing wells, it is sent to gas plants for processing. Gas processing requires a knowledge of how temperature and pressure interact and affect the properties of both fluids and gases. Almost all gas-processing plants handle gases that are mixtures of various hydrocarbon molecules. The purpose of gas processing is to separate these gases into components of similar composition by various processes such as absorption, fractionation and cycling, so they can be transported and used by consumers.
Absorption processes
Absorption involves three processing steps: recovery, removal and separation.
Recovery.
Removes undesirable residue gases and some methane by absorption from the natural gas. Absorption takes place in a counterflow vessel, where the well gas enters the bottom of the vessel and flows upward through absorption oil, which is flowing downward. The absorption oil is “lean” as it enters the top of the vessel, and “rich” as it leaves the bottom as it has absorbed the desirable hydrocarbons from the gas. The gas leaving the top of the unit is called “residue gas.”
Absorption may also be accomplished by refrigeration. The residue gas is used to pre-cool the inlet gas, which then passes through a gas chiller unit at temperatures from 0 to –40 ºC. Lean absorber oil is pumped through an oil chiller, before contacting the cool gas in the absorber unit. Most plants use propane as the refrigerant in the cooler units. Glycol is injected directly into the inlet gas stream to mix with any water in the gas in order to prevent freezing and formation of hydrates. The glycol-water mixture is separated from the hydrocarbon vapour and liquid in the glycol separator, and then reconcentrated by evaporating the water in a regenerator unit.
Removal.
The next step in the absorption process is removal, or demethanization. The remaining methane is removed from the rich oil in ethane recovery plants. This is usually a two-phase process, which first rejects at least one-half of the methane from the rich oil by reducing pressure and increasing temperature. The remaining rich oil usually contains enough ethane and propane to make reabsorption desirable. If not sold, the overhead gas is used as plant fuel or as a pre-saturator, or is recycled to the inlet gas in the main absorber.
Separation.
The final step in the absorption process, distillation, uses vapours as a medium to strip the desirable hydrocarbons from the rich absorption oil. Wet stills use steam vapours as the stripping medium. In dry stills, hydrocarbon vapours, obtained from partial vaporization of the hot oil pumped through the still reboiler, are used as the stripping medium. The still controls the final boiling point and molecular weight of the lean oil, and the boiling point of the final hydrocarbon product mix.
Other Processes
Fractionation.
Is the separation of the desirable hydrocarbon mixture from absorption plants, into specific, individual, relatively pure products. Fractionation is possible when the two liquids, called top product and bottom product, have different boiling points. The fractionation process has three parts: a tower to separate products, a reboiler to heat the input and a condenser to remove heat. The tower has an abundance of trays so that a lot of vapour and liquid contact occurs. The reboiler temperature determines the composition of the bottom product.
Sulphur recovery.
Hydrogen sulphide must be removed from gas before it is shipped for sale. This is accomplished in sulphur recovery plants.
Gas cycling.
Gas cycling is neither a means of pressure maintenance nor a secondary method of recovery, but is an enhanced recovery method used to increase production of natural gas liquids from “wet gas” reservoirs. After liquids are removed from the “wet gas” in cycling plants, the remaining “dry gas” is returned to the reservoir through injection wells. As the “dry gas” recirculates through the reservoir it absorbs more liquids. The production, processing and recirculation cycles are repeated until all of the recoverable liquids have been removed from the reservoir and only “dry gas” remains.
Site Development for Producing Oil and Gas Fields
Extensive site development is required to bring a new oil or gas field into production. Site access may be limited or constrained by both climatic and geographic conditions. The requirements include transportation; construction; maintenance, housing and administrative facilities; oil, gas and water separation equipment; crude oil and natural gas transport; water and waste disposal facilities; and many other services, facilities and kinds of equipment. Most of these are not readily available at the site and must be provided by either the drilling or producing company or by outside contractors.
Contractor activities
Contractors are typically used by oil and gas exploration and producing companies to provide some or all of the following supporting services required to drill and develop producing fields:
- Site preparation - brush clearing, road construction, ramps and walkways, bridges, aircraft landing areas, marine harbour, wharfs, docks and landings
- Erection and installation - drilling equipment, power and utilities, tanks and pipeline, housing, maintenance buildings, garages, hangers, service and administration buildings
- Underwater work - installation, inspection, repair and maintenance of underwater equipment and structures
- Maintenance and repair - drilling and production equipment preventive maintenance, vehicles and boats, machinery and buildings
- Contract services - food service; housekeeping; facility and perimeter protection and security; janitorial, recreation and support activity; warehousing and distribution of protective equipment, spare parts and disposable supplies
- Engineering and technical - testing and analyses, computer services, inspections, laboratories, non-destructive analysis, explosives storage and handling, fire protection, permits, environmental, medical and health, industrial hygiene and safety and spill response
- Outside services - telephone, radio and television, sewerage and garbage
- Transportation and material handling equipment - aircraft and helicopter, marine services, heavy-duty construction and materials handling equipment
Utilities
Whether exploration, drilling and producing operations take place on land or offshore, power, light electricity and other support utilities are required, including:
- Power generation - gas, electricity and steam
- Water - fresh water supply, purification and treatment and process water
- Sewerage and drainage - storm water, sanitary treatment and waste (oily) water treatment and disposal
- Communications - telephone, radio and television, computer and satellite communication
- Utilities - light, heat, ventilation and cooling.
Working Conditions, Health and Safety
Work on drilling rigs usually involves a minimum crew of 6 people (primary and secondary drillers, three assistant drillers or helpers (roughnecks) and a cathead person) reporting to a site supervisor or foreman (tool pusher) who is responsible for the drilling progression. The primary and secondary drillers have overall responsibility for drilling operations and supervision of the drilling crew during their respective shifts. Drillers should be familiar with the capabilities and limitations of their crews, as work can progress only as fast as the slowest crew member.
Assistant drillers are stationed on the platform to operate equipment, read instruments and perform routine maintenance and repair work. The cathead person is required to climb up near the top of the derrick when drill pipe is being fed into or drawn out of the well hole and assist in moving the sections of pipe into and out of the stack. During drilling, the cathead person also operates the mud pump and provides general assistance to the drilling crew.
Persons who assemble, place, discharge and retrieve perforating guns should be trained, familiar with the hazards of explosives and qualified to handle explosives, primer cord and blasting caps. Other personnel working in and around oil fields include geologists, engineers, mechanics, drivers, maintenance personnel, electricians, pipeline operators and labourers.
Wells are drilled around the clock, on either 8- or 12-hour shifts, and workers require considerable experience, skill and stamina to meet the rigorous physical and mental demands of the job. Overextending a crew may result in a serious accident or injury. Drilling requires close teamwork and coordination in order to accomplish the tasks in a safe and timely fashion. Because of these and other requirements, consideration must be given to the morale and health and safety of workers. Adequate periods of rest and relaxation, nutritious food and appropriate hygiene and living quarters, including air conditioning in hot, humid climates and heating in cold-weather areas, are essential.
The primary occupational hazards associated with exploration and production operations include illnesses from exposure to geographical and climatic elements, stress from travelling long distances over water or harsh terrain and personal injury. Psychological problems may result from the physical isolation of exploratory sites and their remoteness from base camps and the extended work periods required on offshore drilling platforms and at remote onshore sites. Many other hazards particular to offshore operations, such as underwater diving, are covered elsewhere in this Encyclopaedia.
Offshore work is dangerous at all times, both when on and off the job. Some workers cannot handle the stress of working offshore at a demanding pace, for extended periods of time, under relative confinement and subject to ever changing environmental conditions. The signs of stress in workers include unusual irritability, other signs of mental distress, excessive drinking or smoking and use of drugs. Problems of insomnia, which may be aggravated by high levels of vibration and noise, have been reported by workers on platforms. Fraternization among workers and frequent shore leave may reduce stress. Seasickness and drowning, as well as exposure to severe weather conditions, are other hazards in offshore work.
Illnesses such as respiratory tract diseases result from exposure to harsh climates, infections or parasitic diseases in areas where these are endemic. Although many of these diseases are still in need of epidemiological study in drilling workers, it is known that oil workers have experienced periarthritis of the shoulder and shoulder blade, humeral epicondylitis, arthrosis of the cervical spine and polyneuritis of the upper limbs. The potential for illnesses as a result of exposure to noise and vibration is also present in drilling operations. The severity and frequency of these drilling-related illnesses appears to be proportional to the length of service and exposure to adverse working conditions (Duck 1983; Ghosh 1983; Montillier 1983).
Injuries while working in drilling and production activities may result from many causes, including slips and falls, pipe handling, lifting pipe and equipment, misuse of tools and mishandling explosives. Burns may be caused by steam, fire, acid or mud containing chemicals such as sodium hydroxide. Dermatitis and skin injuries may result from exposure to crude oil and chemicals.
The possibility exists for acute and chronic exposure to a wide variety of unhealthful materials and chemicals which are present in oil and gas drilling and production. Some chemicals and materials which may be present in potentially hazardous amounts are listed in table 2 and include:
- Crude oil, natural gas and hydrogen sulphide gas during drilling and blowouts
- Heavy metals, benzene and other contaminants present in crude
- Asbestos, formaldehyde, hydrochloric acid and other hazardous chemicals and materials
- Normally occurring radioactive materials (NORMs) and equipment with radioactive sources.
Safety
Drilling and production take place in all types of climates and under varying weather conditions, from tropical jungles and deserts to the frozen Arctic, and from dry land to the North Sea. Drilling crews have to work in difficult conditions, subject to noise, vibration, inclement weather, physical hazards and mechanical failures. The platform, rotary table and equipment are usually slippery and vibrate from the engine and drilling operation, requiring workers to make deliberate and careful movements. The hazard exists for slips and falls from heights when climbing the rig and derrick, and there is risk of exposure to crude oil, gas, mud and engine exhaust fumes. The operation of rapidly disconnecting and then reconnecting drill pipe requires training, skill and precision by workers in order to be done safely time after time.
Construction, drilling and production crews working offshore have to contend with the same hazards as crews working on land, and with the additional hazards specific to offshore work. These include the possibility of collapse of the platform at sea and provisions for specialized evacuation procedures and survival equipment in event of an emergency. Another important consideration when working offshore is the requirement for both deep-sea and shallow-water diving to install, maintain and inspect equipment.
Fire and explosion
There is always a risk of blowout when perforating a well, with a gas or vapour cloud release, followed by explosion and fire. Additional potential for fire and explosion exists in gas process operations.
Offshore platform and drilling rig workers should be carefully evaluated after having a thorough physical examination. The selection of offshore crew members with a history or evidence of pulmonary, cardiovascular or neurological diseases, epilepsy, diabetes, psychological disturbances and drug or alcohol addiction requires careful consideration. Because workers will be expected to use respiratory protection equipment and, in particular, those trained and equipped to fight fires, they must be physically and mentally evaluated for capability of carrying out these tasks. The medical examination should include psychological evaluation reflective of the particular job requirements.
Emergency medical services on offshore drilling rigs and production platforms should include provisions for a small dispensary or clinic, staffed by a qualified medical practitioner on board at all times. The type of medical service provided will be determined by the availability, distance and quality of the available onshore services. Evacuation may be by ship or helicopter, or a physician may travel to the platform or provide medical advice by radio to the onboard practitioner, when needed. A medical ship may be stationed where a number of large platforms operate in a small area, such as the North Sea, to be more readily available and quickly provide service to a sick or injured worker.
Persons not actually working on drilling rigs or platforms should also be given pre-employment and periodic medical examinations, particularly if they are employed to work in abnormal climates or under harsh conditions. These examinations should take into consideration the particular physical and psychological demands of the job.
Personal protection
An occupational hygiene monitoring and sampling programme, in conjunction with a medical surveillance programme, should be implemented to evaluate systematically the extent and effect of hazardous exposures to workers. Monitoring for flammable vapours and toxic exposures, such as hydrogen sulphide, should be implemented during exploration, drilling and production operations. Virtually no exposure to H2S should be permitted, especially on offshore platforms. An effective method of controlling exposure is by using properly weighted drilling mud to keep H2S from entering the well and by adding chemicals to the mud to neutralize any entrapped H2S. All workers should be trained to recognize the presence of H2S and take immediate preventive measures to reduce the possibility of toxic exposure and explosions.
Persons engaged in exploration and production activities should have available and use appropriate personal protective equipment including:
- Head protection (hard hats and weather-proof liners)
- Gloves (oil-resistant, non-slip work gloves, fire insulated or thermal where needed)
- Arm protection (long sleeves or oil-proof gauntlets)
- Foot and leg protection (weather-protected, oil-impervious safety boots with steel toes and non-skid soles)
- Eye and face protection (safety glasses, goggles and face shield for acid handling)
- Skin protection from heat and cold (sun screen ointment and cold-weather face masks)
- Climatized and weather-proof clothing (parkas, rain gear)
- Where required, firefighting gear, flame-resistant clothing and acid-resistant aprons or suits.
Control rooms, living quarters and other spaces on large offshore platforms are usually pressurized to prevent the entry of harmful atmospheres, such as hydrogen sulphide gas, which may be released upon penetration or in an emergency. Respiratory protection may be needed in the event pressure fails, and when there is a possibility of exposure to toxic gases (hydrogen sulphide), asphyxiants (nitrogen, carbon dioxide), acids (hydrogen fluoride) or other atmospheric contaminants when working outside of pressurized areas.
When working around geopressure/geothermal wells, insulated gloves and full heat- and steam-protective suits with supplied breathing air should be considered, as contact with hot steam and vapours can cause burns to skin and lungs.
Safety harnesses and lifelines should be used when on catwalks and gangways, especially on offshore platforms and in inclement weather. When climbing rigs and derricks, harnesses and lifelines with an attached counterweight should be used. Personnel baskets, carrying four or five workers wearing personal flotation devices, are often used to transfer crews between boats and offshore platforms or drilling rigs. Another means of transfer is by “swing ropes.” Ropes used to swing from boats to platforms are hung directly above the edge of the boat landings, while those from platforms to boats should hang 3 or 4 feet from the outer edge.
Providing washing facilities for both workers and clothing and following proper hygiene practices are fundamental measures to control dermatitis and other skin diseases. Where needed, emergency eye wash stations and safety showers should be considered.
Safety protection measures
Oil and gas platform safety shutdown systems use various devices and monitors to detect leaks, fires, ruptures and other hazardous conditions, activate alarms and shut down operations in a planned, logical sequence. Where needed due to the nature of the gas or crude, non-destructive testing methods, such as ultrasonic, radiography, magnetic particle, liquid dye penetrant or visual inspections, should be used to determine the extent of corrosion of piping, heater tubes, treaters and vessels used in crude oil, condensate and gas production and processing.
Surface and sub-surface safety shut-in valves protect onshore installations, single wells in shallow water and multi-well offshore deep-water drilling and production platforms, and are automatically (or manually) activated in the event of fire, critical pressure changes, catastrophic failure at the well head or other emergency. They are also used to protect small injection wells and gas lift wells.
Inspection and care of cranes, winches, drums, wire rope and associated appurtenances is an important safety consideration in drilling. Dropping a pipeline string inside a well is a serious incident, which may result in the loss of the well. Injuries, and sometimes fatalities, can occur when personnel are struck by a wire rope which breaks while under tension. Safe operation of the drilling rig is also dependent on a smooth-running, well maintained draw works, with properly adjusted catheads and braking systems. When working on land, keep cranes a safe distance from electric power lines.
Handling of explosives during exploration and drilling operations should be under the control of a specifically qualified person. Some safety precautions to be considered while using a perforating gun include:
- Never strike or drop a loaded gun, or drop piping or other materials on a loaded gun.
- Clear the line of fire and evacuate unnecessary personnel from the drilling rig floor and the floor below as the perforating gun is lowered into and retrieved from the well hole.
- Control work on or around the wellhead while the gun is in the well.
- Restrict use of radios and prohibit arc welding while the gun is attached to the cable to prevent discharge from an inadvertent electric impulse.
Emergency preparedness planning and drills are important to the safety of workers on oil and gas drilling and production rigs and offshore platforms. Each different type of potential emergency (e.g., fire or explosion, flammable or toxic gas release, unusual weather conditions, worker overboard, and the need to abandon a platform) should be evaluated and specific response plans developed. Workers need to be trained in the correct actions to be taken in emergencies, and familiar with the equipment to be used.
Helicopter safety and survival in the event of dropping into water are important considerations for offshore platform operations and emergency preparedness. Pilots and passengers should wear seat-belts and, where required, survival gear during flight. Life vests should be worn at all times, both during flight and when transferring from helicopter to platform or ship. Careful attention to keep bodies and materials beneath the path of the rotor blade is required when entering, leaving or working around a helicopter.
Training of both onshore and offshore workers is essential to a safe operation. Workers should be required to attend regularly scheduled safety meetings, covering both mandatory and other subjects. Statutory regulations have been enacted by government agencies, including the US Occupational Safety and Health Administration, the US Coast Guard for offshore operations, and the equivalents in the United Kingdom, Norway and elsewhere, which regulate the safety and health of exploration and production workers, both onshore and offshore. The International Labour Organization Code of Practice Safety and Health in the Construction of Fixed Offshore Installations in the Petroleum Industry (1982) provides guidance in this area. The American Petroleum Institute has a number of standards and recommended practices covering safety and health related to exploration and production activities.
Fire protection and prevention measures
Fire prevention and protection, especially on offshore drilling rigs and production platforms, is an important element in the safety of the workers and continued operations. Workers should be trained and educated to recognize the fire triangle, as discussed in the Fire chapter, as it applies to flammable and combustible hydrocarbon liquids, gases and vapours and the potential hazards of fires and explosions. An awareness of fire prevention is essential and includes a knowledge of ignition sources such as welding, open flames, high temperatures, electrical energy, static sparks, explosives, oxidizers and incompatible materials.
Both passive and active fire-protection systems are used onshore and offshore.
- Passive systems include fireproofing, layout and spacing, equipment design, electrical classification and drainage.
- Detectors and sensors are installed which activate alarms, and may also activate automatic protection systems, upon detecting heat, flame, smoke, gas or vapours.
- Active fire protection includes fire water systems, fire water supply, pumps, hydrants, hoses and fixed sprinkler systems; dry chemical automatic systems and manual extinguishers; halon and carbon dioxide systems for confined or enclosed areas such as control rooms, computer rooms and laboratories; and foam water systems.
Employees who are expected to fight fires, from small fires in the incipient stages to large fires in enclosed spaces, such as on offshore platforms, must be properly trained and equipped. Workers assigned as fire brigade leaders and incident commanders need leadership capabilities and additional specialized training in advanced firefighting and fire-control techniques.
Environmental Protection
The major sources of air, water and ground pollution in oil and natural gas production are from oil spills or gas leaks on land or sea, hydrogen sulphide present in oil and gas escaping into the atmosphere, hazardous chemicals present in drilling mud contaminating water or land and combustion products of oil well fires. The potential public health effects of inhalation of smoke particulates from large-scale oil field fires has been of great concern since the oil well fires that occurred in Kuwait during the Persian Gulf War in 1991.
Pollution controls typically include:
- API separators and other waste and water treatment facilities
- Spill control, including booms for spills on water
- Spill containment, dikes and drainage to control oil spills and divert oily water to treatment facilities.
Gas dispersion modelling is conducted to ascertain the probable area which would be affected by a cloud of escaping toxic or flammable gas or vapour. Groundwater table studies are conducted to project the maximum extent of water pollution should oil contamination occur.
Workers should be trained and qualified to provide first aid response to mediate spills and leakage. Contractors who specialize in pollution remediation are usually engaged to manage large spill responses and remediation projects.
Petroleum Refining Process
General Profile
Petroleum refining begins with the distillation, or fractionation, of crude oils into separate hydrocarbon groups. The resultant products are directly related to the characteristics of the crude oil being processed. Most of these products of distillation are further converted into more useable products by changing their physical and molecular structures through cracking, reforming and other conversion processes. These products are subsequently subjected to various treatment and separation processes, such as extraction, hydrotreating and sweetening, in order to produce finished products. Whereas the simplest refineries are usually limited to atmospheric and vacuum distillation, integrated refineries incorporate fractionation, conversion, treatment and blending with lubricant, heavy fuels and asphalt manufacturing; they may also include petrochemical processing.
The first refinery, which opened in 1861, produced kerosene by simple atmospheric distillation. Its by-products included tar and naphtha. It was soon discovered that high-quality lubricating oils could be produced by distilling petroleum under vacuum. However, for the next 30 years, kerosene was the product consumers wanted most. The two most significant events which changed this situation were:
- the invention of the electric light, which decreased the demand for kerosene
- the invention of the internal-combustion engine, which created a demand for diesel fuel and gasoline (naphtha).
With the advent of mass production and the First World War, the number of gasoline-powered vehicles increased dramatically, and the demand for gasoline grew accordingly. However, only a certain amount of gasoline could be obtained from crude oil through atmospheric and vacuum distillation processes. The first thermal cracking process was developed in 1913. Thermal cracking subjected heavy fuels to both pressure and intense heat, physically breaking their large molecules into smaller ones, producing additional gasoline and distillate fuels. A sophisticated form of thermal cracking, visbreaking, was developed in the late 1930s to produce more desirable and valuable products.
As higher-compression gasoline engines were developed, there was a demand for higher-octane gasoline with better anti-knock characteristics. The introduction of catalytic cracking and poly- merization processes in the mid- to late 1930s met this demand by providing improved gasoline yields and higher octane numbers. Alkylation, another catalytic process, was developed in the early 1940s to produce more high-octane aviation gasoline and petrochemical feedstocks, the starting materials, for explosives and synthetic rubber. Subsequently, catalytic isomerization was developed to convert hydrocarbons to produce increased quantities of alkylation feedstocks.
Following the Second World War, various reforming processes were introduced which improved gasoline quality and yield, and produced higher-quality products. Some of these involved the use of catalysts and/or hydrogen to change molecules and remove sulphur. Improved catalysts, and process methods such as hydrocracking and reforming, were developed throughout the 1960s to increase gasoline yields and improve anti-knock characteristics. These catalytic processes also produced molecules with a double bond (alkenes), forming the basis of the modern petrochemical industry.
The numbers and types of different processes used in modern refineries depend primarily on the nature of the crude feedstock and finished product requirements. Processes are also affected by economic factors including crude costs, product values, availability of utilities and transportation. The chronology of the introduction of various processes is given in table 1.
Table 1. Summary of the history of refining processing
|
Year |
Process name |
Process purpose |
Process by-products |
|
1862 |
Atmospheric distillation |
Produce kerosene |
Naphtha, tar, etc. |
|
1870 |
Vacuum distillation |
Lubricants (original) |
Asphalt, residual |
|
1913 |
Thermal cracking |
Increase gasoline |
Residual, bunker fuel |
|
1916 |
Sweetening |
Reduce sulphur and odour |
Sulphur |
|
1930 |
Thermal reforming |
Improve octane number |
Residual |
|
1932 |
Hydrogenation |
Remove sulphur |
Sulphur |
|
1932 |
Coking |
Produce gasoline base stocks |
Coke |
|
1933 |
Solvent extraction |
Improve lubricant viscosity index |
Aromatics |
|
1935 |
Solvent dewaxing |
Improve pour point |
Waxes |
|
1935 |
Catalytic polymerization |
Improve gasoline yield and octane number |
Petrochemical feedstocks |
|
1937 |
Catalytic cracking |
Higher octane gasoline |
Petrochemical feedstocks |
|
1939 |
Visbreaking |
Reduce viscosity |
Increased distillate, tar |
|
1940 |
Alkylation |
Increase gasoline octane and yield |
High-octane aviation gasoline |
|
1940 |
Isomerization |
Produce alkylation feedstock |
Naphtha |
|
1942 |
Fluid catalytic cracking |
Increase gasoline yield and octane |
Petrochemical feedstocks |
|
1950 |
Deasphalting |
Increase cracking feedstock |
Asphalt |
|
1952 |
Catalytic reforming |
Convert low-quality naphtha |
Aromatics |
|
1954 |
Hydrodesulphurization |
Remove sulphur |
Sulphur |
|
1956 |
Inhibitor sweetening |
Remove mercaptan |
Disulphides |
|
1957 |
Catalytic isomerization |
Convert to molecules with high octane number |
Alkylation feedstocks |
|
1960 |
Hydrocracking |
Improve quality and reduce sulphur |
Alkylation feedstocks |
|
1974 |
Catalytic dewaxing |
Improve pour point |
Wax |
|
1975 |
Residual hydrocracking |
Increase gasoline yield from residual |
Heavy residuals |
Basic refining processes and operations
Petroleum refining processes and operations can be classified into the following basic areas: separation, conversion, treatment, formulating and blending, auxiliary refining operations and refining non-process operations. See figure 1 for a simplified flow chart.
Figure 1. Refinery process chart
Separation. Crude oil is physically separated by fractionation in atmospheric and vacuum distillation towers, into groups of hydrocarbon molecules with various boiling-point ranges, called “fractions” or “cuts”.
Conversion. Conversion processes used to change the size and/or structure of hydrocarbon molecules include:
- decomposition (dividing) by hydro-, thermal and catalytic cracking, coking and visbreaking
- unification (combining) through alkylation and polymerization
- alteration (rearranging) with isomerization and catalytic reforming
- treatment.
Since the beginning of refining, various treatment methods have been used to remove non-hydrocarbons, impurities and other constituents that adversely affect the performance properties of finished products or reduce the efficiency of the conversion processes. Treatment involves both chemical reactions and physical separation, such as dissolving, absorption or precipitation, using a variety and combination of processes. Treatment methods include removing or separating aromatics and naphthenes, as well as removing impurities and undesirable contaminants. Sweetening compounds and acids are used to desulphurize crude oil before processing, and to treat products during and after processing. Other treatment methods include crude desalting, chemical sweetening, acid treating, clay contacting, hydrodesulphurizing, solvent refining, caustic washing, hydrotreating, drying, solvent extraction and solvent dewaxing.
Formulating and blending is the process of mixing and combining hydrocarbon fractions, additives and other components to produce finished products with specific desired performance properties.
Auxiliary refining operations. Other refinery operations which are required to support hydrocarbon processing include light ends recovery; sour water stripping; solid waste, waste water and process water treatment and cooling; hydrogen production; sulphur recovery; and acid and tail gas treatment. Other process functions are providing catalysts, reagents, steam, air, nitrogen, oxygen, hydrogen and fuel gases.
Refinery non-process facilities. All refineries have a multitude of facilities, functions, equipment and systems which support the hydrocarbon process operations. Typical support operations are heat and power generation; product movement; tank storage; shipping and handling; flares and relief systems; furnaces and heaters; alarms and sensors; and sampling, testing and inspecting. Non-process facilities and systems include firefighting, water and protection systems, noise and pollution controls, laboratories, control rooms, warehouses, maintenance and administrative facilities.
Major Products of Crude Oil Refining
Petroleum refining has evolved continuously in response to changing consumer demand for better and different products. The original process requirement was to produce kerosene as a cheaper and better source of fuel for lighting than whale oil. The development of the internal combustion engine led to the production of benzene, gasoline and diesel fuels. The evolution of the airplane created a need for high-octane aviation gasoline and jet fuel, which is a sophisticated form of the original refinery product, kerosene. Present-day refineries produce a variety of products, including many which are used as feedstocks for cracking processes and lubricant manufacturing, and for the petrochemical industry. These products can be broadly classified as fuels, petrochemical feedstocks, solvents, process oils, lubricants and special products such as wax, asphalt and coke. (See table 2.)
Table 2. Principal products of crude oil refining
|
Hydrocarbon gases |
Uses |
|
Liquified gases |
Cooking and industrial gas |
|
Chemical industry feedstock |
Rubber products |
|
Carbon black |
Printing inks |
|
Light distillates |
|
|
Light naphthas |
Olefins |
|
Intermediate naphthas |
Aviation and motor gasoline |
|
Heavy naphthas |
Military jet fuel |
|
Gas oil |
Cracking stock |
|
Heavy distillates |
|
|
Technical oils |
Textile oils |
|
Lubricating oils |
Transformer and spindle oils |
|
Paraffin wax |
Rubber industry |
|
Residues |
|
|
Petrolatum |
Petroleum jelly |
|
Residual fuel oil |
No. 6 boiler and process fuel oil |
|
Asphalts |
Paving asphalt |
|
Refinery by-products |
|
|
Coke |
Electrodes and fuel |
|
Sulphonates |
Emulsifiers |
|
Sulphuric acid |
Synthetic fertilizer |
|
Sulphur |
Chemicals |
|
Hydrogen |
Hydrocarbon reformation |
A number of chemicals are used in, or formed as a result of, hydrocarbon processing. A brief description of those which are specific and pertinent to refining follows:
Sulphur Dioxide
Flue gas from burning high-sulphur-content fuels usually contains high levels of sulphur dioxide, which usually is removed by water scrubbing.
Caustics
Caustics are added to desalting water to neutralize acids and reduce corrosion. Caustics are also added to desalted crude in order to reduce the amount of corrosive chlorides in the tower overheads. They are used in refinery treating processes to remove contaminants from hydrocarbon streams.
Nitrogen oxides and carbon monoxide
Flue gas contains up to 200 ppm of nitric oxide, which reacts slowly with oxygen to form nitrogen dioxide. Nitric oxide is not removed by water scrubbing, and nitrogen dioxide can dissolve in water to form nitrous and nitric acid. Flue gas normally contains only a slight amount of carbon monoxide, unless combustion is abnormal.
Hydrogen sulphide
Hydrogen sulphide is found naturally in most crude oils and is also formed during processing by the decomposition of unstable sulphur compounds. Hydrogen sulphide is an extremely toxic, colourless, flammable gas which is heavier than air and soluble in water. It has a rotten egg odour which is discernible at concentrations well below its very low exposure limit. This smell cannot be relied upon to provide adequate warning as the senses are almost immediately desensitized upon exposure. Special detectors are required to alert workers to the presence of hydrogen sulphide, and proper respiratory protection should be used in the presence of the gas. Exposure to low levels of hydrogen sulphide will cause irritation, dizziness and headaches, while exposure to levels in excess of the prescribed limits will cause nervous system depression and eventually death.
Sour water
Sour water is process water which contains hydrogen sulphide, ammonia, phenols, hydrocarbons and low-molecular-weight sulphur compounds. Sour water is produced by steam stripping hydrocarbon fractions during distillation, regenerating catalyst, or steam stripping hydrogen sulphide during hydrotreating and hydrofinishing. Sour water is also generated by the addition of water to processes to absorb hydrogen sulphide and ammonia.
Sulphuric acid and hydrofluoric acid
Sulphuric acid and hydrofluoric acid are used as catalysts in alkylation processes. Sulphuric acid is also used in some of the treatment processes.
Solid catalysts
A number of different solid catalysts in many forms and shapes, from pellets to granular beads to dusts, made of various materials and having various compositions, are used in refining processes. Extruded pellet catalysts are used in moving and fixed bed units, while fluid bed processes use fine, spherical particulate catalysts. Catalysts used in processes which remove sulphur are impregnated with cobalt, nickel or molybdenum. Cracking units use acid-function catalysts, such as natural clay, silica alumina and synthetic zeolites. Acid-function catalysts impregnated with platinum or other noble metals are used in isomerization and reforming. Used catalysts require special handling and protection from exposures, as they may contain metals, aromatic oils, carcinogenic polycyclic aromatic compounds or other hazardous materials, and may also be pyrophoric.
Fuels
The principal fuel products are liquefied petroleum gas, gasoline, kerosene, jet fuel, diesel fuel and heating oil and residual fuel oils.
Liquefied petroleum gas (LPG), which consists of mixtures of paraffinic and olefinic hydrocarbons such as propane and butane, is produced for use as a fuel, and is stored and handled as liquids under pressure. LPG has boiling points ranging from about –74 °C to
38 °C, is colourless, and the vapours are heavier than air and extremely flammable. The important qualities from an occupational health and safety perspective of LPGs are vapour pressure and control of contaminants.
Gasoline. The most important refinery product is motor gasoline, a blend of relatively low-boiling hydrocarbon fractions, including reformate, alkylate, aliphatic naphtha (light straight-run naphtha), aromatic naphtha (thermal and catalytic cracked naphtha) and additives. Gasoline blending stocks have boiling points which range from ambient temperatures to about 204 °C, and a flashpoint below –40 °C. The critical qualities for gasoline are octane number (anti-knock), volatility (starting and vapour lock) and vapour pressure (environmental control). Additives are used to enhance gasoline performance and provide protection against oxidation and rust formation. Aviation gasoline is a high-octane product, specially blended to perform well at high altitudes.
Tetra ethyl lead (TEL) and tetra methyl lead (TML) are gasoline additives which improve octane ratings and anti-knock performance. In an effort to reduce lead in automotive exhaust emissions, these additives are no longer in common use, except in aviation gasoline.
Ethyl tertiary butyl ether (ETBE), methyl tertiary butyl ether (MTBE), tertiary amyl methyl ether (TAME) and other oxygenated compounds are used in lieu of TEL and TML to improve unleaded gasoline anti-knock performance and reduce carbon monoxide emissions.
Jet fuel and kerosene. Kerosene is a mixture of paraffins and naphthenes with usually less than 20% aromatics. It has a flashpoint above 38 °C and a boiling range of 160 °C to 288 °C, and is used for lighting, heating, solvents and blending into diesel fuel. Jet fuel is a middle distillate kerosene product whose critical qualities are freezepoint, flashpoint and smokepoint. Commercial jet fuel has a boiling range of about 191 °C to 274 °C, and military jet fuel from 55 °C to 288 °C.
Distillate fuels. Diesel fuels and domestic heating oils are light-coloured mixtures of paraffins, naphthenes and aromatics, and may contain moderate quantities of olefins. Distillate fuels have flashpoints above 60 °C and boiling ranges of about 163 °C to 371 °C, and are often hydrodesulphurized for improved stability. Distillate fuels are combustible and when heated may emit vapours which can form ignitable mixtures with air. The desirable qualities required for distillate fuels include controlled flash- and pourpoints, clean burning, no deposit formation in storage tanks, and a proper diesel fuel cetane rating for good starting and combustion.
Residual fuels. Many ships and commercial and industrial facilities use residual fuels or combinations of residual and distillate fuels, for power, heat and processing. Residual fuels are dark- coloured, highly viscous liquid mixtures of large hydrocarbon molecules, with flashpoints above 121 °C and high boiling points. The critical specifications for residual fuels are viscosity and low sulphur content (for environmental control).
Health and safety considerations
The primary safety hazard of LPG and gasoline is fire. The high volatility and high flammability of the lower-boiling-point products allows vapours to evaporate readily into air and form flammable mixtures which can be easily ignited. This is a recognized hazard that requires specific storage, containment and handling precautions, and safety measures to assure that releases of vapours and sources of ignition are controlled so that fires do not occur. The less volatile fuels, such as kerosene and diesel fuel, should be handled carefully to prevent spills and possible ignition, as their vapours are also combustible when mixed with air in the flammable range. When working in atmospheres containing fuel vapours, concentrations of highly volatile, flammable product vapours in air are often restricted to no more than 10% of the lower flammable limits (LFL), and concentrations of less volatile, combustible product vapours to no more than 20% LFL, depending on applicable company and government regulations, in order to reduce the risk of ignition.
Although gasoline vapour levels in air mixtures are typically maintained below 10% of the LFL for safety purposes, this concentration is considerably above the exposure limits to be observed for health reasons. When inhaled, small amounts of gasoline vapour in air, well below the lower flammable limit, can cause irritation, headaches and dizziness, while inhalation of larger concentrations can cause loss of consciousness and eventually death. Long-term health effects may also be possible. Gasoline contains benzene, for example, a known carcinogen with allowable exposure limits of only a few parts per million. Therefore, even working in gasoline vapour atmospheres at levels below 10% LFL requires appropriate industrial hygiene precautions, such as respiratory protection or local exhaust ventilation.
In the past, many gasolines contained tetra-ethyl or tetra methyl alky lead anti-knock additives, which are toxic and present serious lead absorption hazards by skin contact or inhalation. Tanks or vessels which contained leaded gasoline at any time during their use must be vented, thoroughly cleaned, tested with a special “lead-in-air” test device and certified to be lead-free to assure that workers can enter without using self-contained or supplied breathing air equipment, even though oxygen levels are normal and the tanks now contain unleaded gasoline or other products.
Gaseous petroleum fractions and the more highly volatile fuel products have a mild anaesthetic effect, generally in inverse ratio to molecular weight. Lower-boiling-point liquid fuels, such as gasoline and kerosene, produce a severe chemical pneumonitis if inhaled, and should not be siphoned by mouth or accidentally ingested. Gases and vapours may also be present in sufficiently high concentrations to displace oxygen (in the air) below normal breathing levels. Maintaining vapour concentrations below the exposure limits and oxygen levels at normal breathing ranges, is usually accomplished by purging or ventilation.
Cracked distillates contain small amounts of carcinogenic polycyclic aromatic hydrocarbons (PAHs); therefore, exposure should be limited. Dermatitis may also develop from exposure to gasoline, kerosene and distillate fuels, as they have a tendency to defat the skin. Prevention is accomplished by use of personal protective equipment, barrier creams or reduced contact and good hygienic practices, such as washing with warm water and soap instead of cleaning hands with gasoline, kerosene or solvents. Some persons have skin sensitivity to the dyes used to colour gasoline and other distillate products.
Residual fuel oils contain traces of metals and may have entrained hydrogen sulphide, which is extremely toxic. Residual fuels which have high cracked stocks boiling above 370 °C contain carcinogenic PAHs. Repeated exposure to residual fuels without appropriate personal protection, should be avoided, especially when opening tanks and vessels, as hydrogen sulphide gas may be emitted.
Petrochemical feedstocks
Many products derived from crude-oil refining, such as ethylene, propylene and butadiene, are olefinic hydrocarbons derived from refinery cracking processes, and are intended for use in the petrochemical industry as feedstocks for the production of plastics, ammonia, synthetic rubber, glycol and so on.
Petroleum solvents
A variety of pure compounds, including benzene, toluene, xylene, hexane and heptane, whose boiling points and hydrocarbon composition are closely controlled, are produced for use as solvents. Solvents may be classified as aromatic or non-aromatic, depending on their composition. Their use as paint thinners, dry-cleaning fluids, degreasers, industrial and pesticide solvents and so on, is generally determined by their flashpoints, which vary from well below –18 °C to above 60 °C.
The hazards associated with solvents are similar to those of fuels in that the lower flashpoint solvents are flammable and their vapours, when mixed with air in the flammable range, are ignitable. Aromatic solvents will usually have more toxicity than non-aromatic solvents.
Process oils
Process oils include the high boiling range, straight run atmospheric or vacuum distillate streams and those which are produced by catalytic or thermal cracking. These complex mixtures, which contain large paraffinic, naphthenic and aromatic hydrocarbon molecules with more than 15 carbon atoms, are used as feedstocks for cracking or lubricant manufacturing. Process oils have fairly high viscosities, boiling points ranging from 260 °C to 538 °C, and flashpoints above 121 °C.
Process oils are irritating to the skin and contain high concentrations of PAHs as well as sulphur, nitrogen and oxygen compounds. Inhalation of vapours and mists should be avoided, and skin exposure should be controlled by the use of personal protection and good hygienic practices.
Lubricants and greases
Lubricating oil base stocks are produced by special refining processes to meet specific consumer requirements. Lubricating base stocks are light- to medium-coloured, low-volatile, medium- to high-viscous mixtures of paraffinic, naphthenic and aromatic oils, with boiling ranges from 371 °C to 538 °C. Additives, such as demulsifiers, anti-oxidants and viscosity improvers, are blended into the lubricating oil base stocks to provide the characteristics required for motor oils, turbine and hydraulic oils, industrial greases, lubricants, gear oils and cutting oils. The most critical quality for lubricating oil base stock is a high viscosity index, providing for less change in viscosity under varying temperatures. This characteristic may be present in the crude oil feed stock or attained through the use of viscosity index improver additives. Detergents are added to keep in suspension any sludge formed during the use of the oil.
Greases are mixtures of lubricating oils and metallic soaps, with the addition of special-purpose materials such as asbestos, graphite, molybdenum, silicones and talc to provide insulation or lubricity. Cutting and metal-process oils are lubricating oils with special additives such as chlorine, sulphur and fatty-acid additives which react under heat to provide lubrication and protection to the cutting tools. Emulsifiers and bacteria prevention agents are added to water-soluble cutting oils.
Although lubricating oils by themselves are non-irritating and have little toxicity, hazards may be presented by the additives. Users should consult supplier material safety data information to determine the hazards of specific additives, lubricants, cutting oils and greases. The primary lubricant hazard is dermatitis, which can usually be controlled by the use of personal protective equipment together with proper hygienic practices. Occasionally workers may develop a sensitivity to cutting oils or lubricants which will require reassignment to a job where contact cannot occur. There are some concerns about carcinogenic exposure to mists from naphthenic-based cutting and light spindle oils, which can be controlled by substitution, engineering controls or personal protection. The hazards of exposure to grease are similar to those of lubricating oil, with the addition of any hazards presented by the grease materials or additives. Most of these hazards are discussed elsewhere in this Encyclopaedia.
Special products
Wax is used for protecting food products; in coatings; as an ingredient in other products such as cosmetics and shoe polish and for candles.
Sulphur is produced as a result of petroleum refining. It is stored either as a heated, molten liquid in closed tanks or as a solid in containers or outdoors.
Coke is almost pure carbon, with a variety of uses from electrodes to charcoal briquettes, depending on its physical characteristics, which result from the coking process.
Asphalt, which is primarily used for paving roads and roofing materials, should be inert to most chemicals and weather conditions.
Waxes and asphalts are solid at ambient temperatures, and higher temperatures are needed for storage, handling and transportation, with the resulting hazard of burns. Petroleum wax is so highly refined that it usually does not present any hazards. Skin contact with wax can lead to plugging of pores, which can be controlled by proper hygienic practices. Exposure to hydrogen sulphide when asphalt and molten sulphur tanks are opened can be controlled by the use of appropriate engineering controls or respiratory protection. Sulphur is also readily ignitable at elevated temperatures. Asphalt is discussed elsewhere in the Encyclopaedia.
Petroleum Refining Processes
Hydrocarbon refining is the use of chemicals, catalysts, heat and pressure to separate and combine the basic types of hydrocarbon molecules naturally found in crude oil into groups of similar molecules. The refining process also rearranges the structures and bonding patterns of the basic molecules into different, more desirable hydrocarbon molecules and compounds. The type of hydrocarbon (paraffinic, naphthenic or aromatic) rather than the specific chemical compounds present, is the most significant factor in the refining process.
Throughout the refinery, operations procedures, safe work practices and the use of appropriate personal protective clothing and equipment, including approved respiratory protection, is needed for fire, chemical, particulate, heat and noise exposures and during process operations, sampling, inspection, turnaround and maintenance activities. As most refinery processes are continuous and the process streams are contained in enclosed vessels and piping, there is limited potential for exposure. However, the potential for fire exists because even though refinery operations are closed processes, if a leak or release of hydrocarbon liquid, vapour or gas occurs, the heaters, furnaces and heat exchangers throughout the process units are sources of ignition.
Crude oil pretreatment
Desalting
Crude oil often contains water, inorganic salts, suspended solids and water-soluble trace metals. The first step in the refining process is to remove these contaminants by desalting (dehydration) in order to reduce corrosion, plugging and fouling of equipment, and to prevent poisoning the catalysts in processing units. Chemical desalting, electrostatic separation and filtering are three typical methods of crude-oil desalting. In chemical desalting, water and chemical surfactants (demulsifiers) are added to the crude oil, heated so that salts and other impurities dissolve into the water or attach to the water, and are then held in a tank where they settle out. Electrical desalting applies high-voltage electrostatic charges in order to concentrate suspended water globules in the bottom portion of the settling tank. Surfactants are added only when the crude oil has a large amount of suspended solids. A third, less common process involves filtering heated crude oil using diatomaceous earth as a filtration medium.
In chemical and electrostatic desalting, the crude feedstock is heated to between 66 °C and 177 °C, to reduce viscosity and surface tension for easier mixing and separation of the water. The temperature is limited by the vapour pressure of the crude-oil feedstock. Both methods of desalting are continuous. Caustic or acid may be added to adjust the pH of the water wash, and ammonia added to reduce corrosion. Waste water, together with contaminants, is discharged from the bottom of the settling tank to the waste water treatment facility. The desalted crude oil is continuously drawn from the top of the settling tanks and sent to an atmospheric crude distillation (fractionating) tower. (See figure 2.)
Figure 2. Desalting (pre-treatment) process
Inadequate desalting causes fouling of heater tubes and heat exchangers in all refinery process units, restricting product flow and heat transfer, and resulting in failures due to increased pressures and temperatures. Overpressuring the desalting unit will cause failure.
Corrosion, which occurs due to the presence of hydrogen sulphide, hydrogen chloride, naphthenic (organic) acids and other contaminants in the crude oil, also causes equipment failure. Corrosion occurs when neutralized salts (ammonium chlorides and sulphides) are moistened by condensed water. Because desalting is a closed process, there is little potential for exposure to crude oil or process chemicals, unless a leak or release occurs. A fire may occur as a result of a leak in the heaters, allowing a release of low-boiling-point components of crude oil.
There is the possibility of exposure to ammonia, dry chemical demulsifiers, caustics and/or acids during desalting. Where elevated operating temperatures are used when desalting sour crude oils, hydrogen sulphide will be present. Depending on the crude feedstock and the treatment chemicals used, the waste water will contain varying amounts of chlorides, sulphides, bicarbonates, ammonia, hydrocarbons, phenol and suspended solids. If diatomaceous earth is used in filtration, exposures should be minimized or controlled since diatomaceous earth can contain silica with a very fine particle size, making it a potential respiratory hazard.
Crude oil separation processes
The first step in petroleum refining is the fractionation of crude oil in atmospheric and vacuum distillation towers. Heated crude oil is physically separated into various fractions, or straight-run cuts, differentiated by specific boiling-point ranges and classified, in order of decreasing volatility, as gases, light distillates, middle distillates, gas oils and residuum. Fractionation works because the gradation in temperature from the bottom to the top of the distillation tower causes the higher-boiling-point components to condense first, while the lower-boiling-point fractions rise higher in the tower before they condense. Within the tower, the rising vapours and the descending liquids (reflux) mix at levels where they have compositions in equilibrium with each other. Special trays are located at these levels (or stages) which remove a fraction of the liquid which condenses at each level. In a typical two-stage crude unit, the atmospheric tower, producing light fractions and distillate, is immediately followed by a vacuum tower which processes the atmospheric residuals. After distillation, only a few hydrocarbons are suitable for use as finished products without further processing.
Atmospheric distillation
In atmospheric distillation towers, the desalted crude feedstock is preheated using recovered process heat. It then flows to a direct-fired crude charge heater, where it is fed into the vertical distillation column just above the bottom at pressures slightly above atmosphere and at temperatures from 343 °C to 371 °C, to avoid undesirable thermal cracking at higher temperatures. The lighter (lower boiling point) fractions diffuse into the upper part of the tower, and are continuously drawn off and directed to other units for further processing, treating, blending and distribution.
Fractions with the lowest boiling points, such as fuel gas and light naphtha, are removed from the top of the tower by an overhead line as vapours. Naphtha, or straight-run gasoline, is taken from the upper section of the tower as an overhead stream. These products are used as petrochemical and reformer feedstocks, gasoline blending stocks, solvents and LPGs.
Intermediate boiling range fractions, including gas oil, heavy naphtha and distillates, are removed from the middle section of the tower as side streams. These are sent to finishing operations for use as kerosene, diesel fuel, fuel oil, jet fuel, catalytic cracker feedstock and blending stocks. Some of these liquid fractions are stripped of their lighter ends, which are returned to the tower as downflowing reflux streams.
The heavier, higher-boiling-point fractions (called residuum, bottoms or topped crude) which condense or remain at the bottom of the tower, are used for fuel oil, bitumen manufacturing or cracking feedstock, or are directed to a heater and into the vacuum distillation tower for further fractionation. (See figure 3 and figure 4.)
Figure 3. Atmospheric distillation process
Figure 4. Schematic of atmospheric distrillation process
Vacuum distillation
Vacuum distillation towers provide the reduced pressure required to prevent thermal cracking when distilling the residuum, or topped crude, from the atmospheric tower at higher temperatures. The internal designs of some vacuum towers are different from atmospheric towers in that random packing and demister pads are used instead of trays. Larger diameter towers may also be used to keep velocities lower. A typical first-phase vacuum tower may produce gas oils, lubricating oil base stocks and heavy residual for propane deasphalting. A second-phase tower, operating at a lower vacuum, distills surplus residuum from the atmospheric tower which is not used for lube stock processing, and surplus residuum from the first vacuum tower not used for deasphalting.
Vacuum towers are typically used to separate catalytic cracker feedstocks from surplus residuum. Vacuum tower bottoms may also be sent to a coker, used as lubricant or asphalt stock or desulphurized and blended into low-sulphur fuel oil. (See figure 5 and figure 6.)
Figure 5. Vacuum distillation process
Figure 6. Schematic of vacuum distillation process
Distillation columns
Within refineries there are numerous other smaller distillation towers, called columns, designed to separate specific and unique products, which all work on the same principles as atmospheric towers. For example, a depropanizer is a small column designed to separate propane from isobutane and heavier components. Another larger column is used to separate ethyl benzene and xylene. Small “bubbler” towers, called strippers, use steam to remove trace amounts of light products (gasoline) from heavier product streams.
Control temperatures, pressures and reflux must be maintained within operating parameters to prevent thermal cracking from taking place within distillation towers. Relief systems are provided because excursions in pressure, temperature or liquid levels may occur if automatic control devices fail. Operations are monitored in order to prevent crude from entering the reformer charge. Crude feedstocks may contain appreciable amounts of water in suspension which separate during start-up and, along with water remaining in the tower from steam purging, settle in the bottom of the tower. This water may heat to the boiling point and create an instantaneous vaporization explosion upon contact with the oil in the unit.
The preheat exchanger, preheat furnace and bottoms exchanger, atmospheric tower and vacuum furnace, vacuum tower and overhead are susceptible to corrosion from hydrochloric acid (HCl), hydrogen sulphide (H2S), water, sulphur compounds and organic acids. When processing sour crudes, severe corrosion can occur in both atmospheric and vacuum towers where metal temperatures exceed 232 °C, and in furnace tubing. Wet H2S will also cause cracks in steel. When processing high-nitrogen crudes, nitrogen oxides, which are corrosive to steel when cooled to low temperatures in the presence of water, form in the flue gases of furnaces.
Chemicals are used to control corrosion by hydrochloric acid produced in distillation units. Ammonia may be injected into the overhead stream prior to initial condensation, and/or an alkaline solution may be carefully injected into the hot crude oil feed. If sufficient wash water is not injected, deposits of ammonium chloride can form, causing serious corrosion.
Atmospheric and vacuum distillation are closed processes, and exposures are minimal. When sour (high sulphur) crudes are processed, there may be potential exposure to hydrogen sulphide in the preheat exchanger and furnace, tower flash zone and overhead system, vacuum furnace and tower, and bottoms exchanger. Crude oils and distillation products all contain high-boiling aromatic compounds, including carcinogenic PAHs. Short-term exposure to high concentrations of naphtha vapour can result in headaches, nausea and dizziness, and long-term exposure can result in loss of consciousness. Benzene is present in aromatic naphthas, and exposure must be limited. The dehexanizer overhead may contain large amounts of normal hexane, which can affect the nervous system. Hydrogen chloride may be present in the preheat exchanger, tower top zones and overheads. Waste water may contain water-soluble sulphides in high concentrations and other water-soluble compounds, such as ammonia, chlorides, phenol and mercaptan, depending upon the crude feedstock and the treatment chemicals.
Crude oil conversion processes
Conversion processes, such as cracking, combining and rearranging, change the size and structure of hydrocarbon molecules in order to convert fractions into more desirable products. (See table 3.)
Table 3. Overview of petroleum refining processes
|
Process name |
Action |
Method |
Purpose |
Feedstocks |
Products |
|
Fractionation processes |
|||||
|
Atmospheric distillation |
Separation |
Thermal |
Separate fractions |
Desalted crude oil |
Gas, gas oil, distillate, residual |
|
Vacuum distillation |
Separation |
Thermal |
Separate without cracking |
Atmospheric tower residual |
Gas oil, lube stock, residual |
|
Conversion processes—Decomposition |
|||||
|
Catalytic cracking |
Alteration |
Catalytic |
Upgrade gasoline |
Gas oil, coke distillate |
Gasoline, petrochemical feedstock |
|
Coking |
Polymerization |
Thermal |
Convert vacuum residuals |
Residual, heavy oil, tar |
Naphtha, gas oil, coke |
|
Hydrocracking |
Hydrogenation |
Catalytic |
Convert to lighter hydrocarbons |
Gas oil, cracked oil, residuals |
Lighter, higher quality products |
|
Hydrogen steam reforming |
Decomposition |
Thermal/catalytic |
Produce hydrogen |
Desulphurized gas, O2 ,steam |
Hydrogen, CO,CO2 |
|
Steam cracking |
Decomposition |
Thermal |
Crack large molecules |
Atmospheric tower heavy fuel/distillate |
Cracked naphtha, coke, residuals |
|
Visbreaking |
Decomposition |
Thermal |
Reduce viscosity |
Atmospheric tower residual |
Distillate, car |
|
Conversion processes—Unification |
|||||
|
Alkylation |
Combining |
Catalytic |
Unite olefins and isoparaffins |
Tower isobutane/cracker olefin |
Iso-octane (alkylate) |
|
Grease compounding |
Combining |
Thermal |
Combine soaps and oils |
Lube oil, catty acid, alkymetal |
Lubricating grease |
|
Polymerization |
Polymerization |
Catalytic |
Unite two or more olefins |
Cracker olefins |
High octane naphtha, petrochemical stocks |
|
Conversion processes—Alteration/rearrangement |
|||||
|
Catalytic reforming |
Alteration/ |
Catalytic |
Upgrade low-octane naphtha |
Coker/hydrocracker naphtha |
High-octane reformate/aromatic |
|
Isomerization |
Rearrangement |
Catalytic |
Convert straight chain to branch |
Butane, centane, cexane |
Isobutane/pentane/hexane |
|
Treatment processes |
|||||
|
Amine treating |
Treatment |
Absorption |
Remove acidic contaminants |
Sour gas, cydrocarbons with CO2 and H2S |
Acid-free gases and liquid hydrocarbons |
|
Desalting (pre-treatment) |
Dehydration |
Absorption |
Remove contaminants |
Crude oil |
Desalted crude oil |
|
Drying and sweetening |
Treatment |
Absorption/thermal |
Remove H2O and sulphur compounds |
Liquid hydrocarbon, LPG, alkylated feedstock |
Sweet and dry hydrocarbons |
|
Furfural extraction |
Solvent extraction |
Absorption |
Upgrade middistillate and lubes |
Cycle oils and lube feedstocks |
High-quality diesel and lube oil |
|
Hydrodesulphurization |
Treatment |
Catalytic |
Remove sulphur, contaminants |
High-sulphur residual/gas oil |
Desulphurized olefins |
|
Hydrotreating |
Hydrogenation |
Catalytic |
Remove impurities/ saturate hydrocarbons |
Residuals, cracked hydrocarbons |
Cracker feed, cistillate, lube |
|
Phenol extraction |
Solvent extraction |
Absorption/thermal |
Improve lube viscosity index, colour |
Lube oil base stocks |
High-quality lube oils |
|
Solvent deasphalting |
Treatment |
Absorption |
Remove asphalt |
Vacuum tower residual, cropane |
Heavy lube oil, csphalt |
|
Solvent dewaxing |
Treatment |
Cool/filter |
Remove wax from lube stocks |
Vacuum tower lube oils |
Dewaxed lube base stock |
|
Solvent extraction |
Solvent extraction |
Absorption/ |
Separate unsaturated aromatics |
Gas oil, ceformate, cistillate |
High-octane gasoline |
|
Sweetening |
Treatment |
Catalytic |
Remove H2S, convert mercaptan |
Untreated distillate/gasoline |
High-quality distillate/gasoline |
A number of hydrocarbon molecules not normally found in crude oil but important to the refining process are created as a result of conversion. Olefins (alkenes, di-olefins and alkynes) are unsaturated chain- or ring-type hydrocarbon molecules with at least one double bond. They are usually formed by thermal and catalytic cracking and rarely occur naturally in unprocessed crude oil.
Alkenes are straight-chain molecules with the formula CnHn containing at least one double bond (unsaturated) linkage in the chain. The simplest alkene molecule is the mono-olefin ethylene, with two carbon atoms, joined by a double bond, and four hydrogen atoms. Di-olefins (containing two double bonds), such as 1,2-butadiene and 1,3-butadiene, and alkynes (containing a triple bond), such as acetylene, occur in C5 and lighter fractions from cracking. Olefins are more reactive than paraffins or naphthenes, and readily combine with other elements such as hydrogen, chlorine and bromine.
Cracking processes
Following distillation, subsequent refinery processes are used to alter the molecular structures of the fractions to create more desirable products. One of these processes, cracking, breaks (or cracks) heavier, higher-boiling-point petroleum fractions into more valuable products such as gaseous hydrocarbons, gasoline blending stocks, gas oil and fuel oil. During the process, some of the molecules combine (polymerize) to form larger molecules. The basic types of cracking are thermal cracking, catalytic cracking and hydro-cracking.
Thermal cracking processes
Thermal cracking processes, developed in 1913, heat distillate fuels and heavy oils under pressure in large drums until they crack (divide) into smaller molecules with better anti-knock characteristics. This early method, which produced large amounts of solid, unwanted coke, has evolved into modern thermal cracking processes including visbreaking, steam cracking and coking.
Visbreaking
Visbreaking is a mild form of thermal cracking which reduces the pour point of waxy residues and significantly lowers the viscosity of feedstock without affecting its boiling-point range. Residual from the atmospheric distillation tower is mildly cracked in a heater at atmospheric pressure. It is then quenched with cool gas oil to control overcracking, and flashed in a distillation tower. The thermally cracked residue tar, which accumulates in the bottom of the fractionation tower, is vacuum flashed in a stripper and the distillate is recycled. (See figure 7.)
Figure 7. Visbreaking process
Steam cracking
Steam cracking produces olefins by thermally cracking large hydrocarbon molecule feedstocks at pressures slightly above atmospheric and at very high temperatures. Residual from steam cracking is blended into heavy fuels. Naphtha produced from steam cracking usually contains benzene, which is extracted prior to hydrotreating.
Coking
Coking is a severe form of thermal cracking used to obtain straight-run gasoline (coker naphtha) and various middle distillate fractions used as catalytic cracking feedstocks. This process so completely reduces hydrogen from the hydrocarbon molecule, that the residue is a form of almost pure carbon called coke. The two most common coking processes are delayed coking and continuous (contact or fluid) coking, which, depending upon the reaction mechanism, time, temperature and the crude feedstock, produce three types of coke—sponge, honeycomb and needle coke. (See figure 8.)
Figure 8. Coking process
- Delayed coking. In delayed coking, the feedstock is first charged to a fractionator to separate lighter hydrocarbons, and then combined with heavy recycle oil. The heavy feedstock is fed to the coker furnace and heated to high temperatures at low pressures to prevent premature coking in the heater tubes, producing partial vaporization and mild cracking. The liquid/vapour mixture is pumped from the heater to one or more coker drums, where the hot material is held approximately 24 hours (delayed) at low pressures until it cracks into lighter products. After the coke reaches a predetermined level in one drum, the flow is diverted to another drum to maintain continuous operation. Vapour from the drums is returned to the fractionator to separate out gas, naphtha and gas oils, and to recycle heavier hydrocarbons through the furnace. The full drum is steamed to strip out uncracked hydrocarbons, cooled by water injection and decoked mechanically by an auger rising from the bottom of the drum, or hydraulically by fracturing the coke bed with high-pressure water ejected from a rotating cutter.
- Continuous coking. Continuous (contact or fluid) coking is a moving bed process which operates at lower pressures and higher temperatures than delayed coking. In continuous coking, thermal cracking occurs by using heat transferred from hot recycled coke particles to feedstock in a radial mixer, called a reactor. Gases and vapours are taken from the reactor, quenched to stop further reaction and fractionated. The reacted coke enters a surge drum and is lifted to a feeder and classifier where the larger coke particles are removed. The remaining coke is dropped into the reactor preheater for recycling with feedstock. The process is automatic in that there is a continuous flow of coke and feedstock, and coking occurs both in the reactor and in the surge drum.
Health and safety considerations
In coking, temperature control should be held within a close range, as high temperatures will produce coke which is too hard to cut out of the drum. Conversely, temperatures which are too low will result in a high asphaltic content slurry. Should coking temperatures get out of control, an exothermic reaction could occur.
In thermal cracking when sour crudes are processed, corrosion can occur where metal temperatures are between 232 °C and 482 °C. It appears that coke forms a protective layer on the metal above 482 °C. However, hydrogen sulphide corrosion occurs when temperatures are not properly controlled above 482 °C. The lower part of the tower, high temperature exchangers, furnace and soaking drums are subject to corrosion. Continuous thermal changes cause coke drum shells to bulge and crack.
Water or steam injection is used to prevent buildup of coke in delayed coker furnace tubes. Water must be completely drained from the coker, so as not to cause an explosion upon recharging with hot coke. In emergencies, alternate means of egress from the working platform on top of coke drums is needed.
Burns may occur when handling hot coke, from steam in the event of a steam line leak, or from hot water, hot coke or hot slurry which may be expelled when opening cokers. The potential exists for exposure to aromatic naphthas containing benzene, hydrogen sulphide and carbon monoxide gases, and to trace amounts of carcinogenic PAHs associated with coking operations. Waste sour water may be highly alkaline, and contain oil, sulphides, ammonia and phenol. When coke is moved as a slurry, oxygen depletion may occur within confined spaces such as storage silos, because wet carbon adsorbs oxygen.
Catalytic cracking processes
Catalytic cracking breaks up complex hydrocarbons into simpler molecules in order to increase the quality and quantity of lighter, more desirable products and decrease the amount of residuals. Heavy hydrocarbons are exposed at high temperature and low pressure to catalysts which promote chemical reactions. This process rearranges the molecular structure, converting heavy hydrocarbon feedstocks into lighter fractions such as kerosene, gasoline, LPG, heating oil and petrochemical feedstocks (see figure 9 and figure 10). Selection of a catalyst depends upon a combination of the greatest possible reactivity and the best resistance to attrition. The catalysts used in refinery cracking units are typically solid materials (zeolite, aluminium hydrosilicate, treated bentonite clay, Fuller’s earth, bauxite and silica-alumina) which are in the form of powders, beads, pellets or shaped materials called extrudites.
Figure 9. Catalytic cracking process
Figure 10. Schematic of catalytic cracking process
There are three basic functions in all catalytic cracking processes:
- Reaction—feedstock reacts with catalyst and cracks into different hydrocarbons.
- Regeneration—catalyst is reactivated by burning off coke.
- Fractionation—cracked hydrocarbon stream is separated into various products.
Catalytic cracking processes are very flexible and operating parameters can be adjusted to meet changing product demand. The three basic types of catalytic cracking processes are:
- fluid catalytic cracking (FCC)
- moving bed catalytic cracking
- thermofor catalytic cracking (TCC).
Fluid catalytic cracking
Fluid-bed catalytic crackers have a catalyst section (riser, reactor and regenerator) and a fractionating section, both operating together as an integrated processing unit. The FCC uses finely powdered catalyst, suspended in oil vapour or gas, which acts as a fluid. Cracking takes place in the feed pipe (riser) in which the mixture of catalyst and hydrocarbons flow through the reactor.
The FCC process mixes a preheated hydrocarbon charge with hot, regenerated catalyst as it enters the riser leading to the reactor. The charge combines with recycle oil within the riser, is vaporized and is raised to reactor temperature by the hot catalyst. As the mixture travels up the reactor, the charge is cracked at low pressure. This cracking continues until the oil vapours are separated from the catalyst in the reactor cyclones. The resultant product stream enters a column where it is separated into fractions, with some of the heavy oil directed back into the riser as recycle oil.
Spent catalyst is regenerated to remove coke which collects on the catalyst during the process. Spent catalyst flows through the catalyst stripper to the regenerator where it mixes with preheated air, burning off most of the coke deposits. Fresh catalyst is added and worn-out catalyst removed to optimize the cracking process.
Moving bed catalytic cracking
Moving-bed catalytic cracking is similar to fluid catalytic cracking; however, the catalyst is in the form of pellets instead of fine powder. The pellets move continuously by conveyor or pneumatic lift tubes to a storage hopper at the top of the unit, and then flow downward by gravity through the reactor to a regenerator. The regenerator and hopper are isolated from the reactor by steam seals. The cracked product is separated into recycle gas, oil, clarified oil, distillate, naphtha and wet gas.
Thermofor catalytic cracking
In thermofor catalytic cracking, the preheated feedstock flows by gravity through the catalytic reactor bed. Vapours are separated from the catalyst and sent to a fractionating tower. The spent catalyst is regenerated, cooled and recycled, and the flue gas from regeneration is sent to a carbon monoxide boiler for heat recovery.
Health and safety considerations
Regular sampling and testing of feedstock, product and recycle streams should be performed to assure that the cracking process is working as intended and that no contaminants have entered the process stream. Corrosives or deposits in feedstock can foul gas compressors. When processing sour crude, corrosion may be expected where temperatures are below
482 °C. Corrosion takes place where both liquid and vapour phases exist and at areas subject to local cooling, such as nozzles and platform supports. When processing high-nitrogen feedstocks, exposure to ammonia and cyanide may subject carbon steel equipment in the FCC overhead system to corrosion, cracking or hydrogen blistering, which can be minimized by water wash or by corrosion inhibitors. Water wash may be used to protect overhead condensers in the main column subjected to fouling from ammonium hydrosulphide.
Critical equipment, including pumps, compressors, furnaces and heat exchangers should be inspected. Inspections should include checking for leaks due to erosion or other malfunctions such as catalyst buildup on the expanders, coking in the overhead feeder lines from feedstock residues, and other unusual operating conditions.
Liquid hydrocarbons in the catalyst or entering the heated combustion air stream can cause exothermic reactions. In some processes, caution must be taken to assure that explosive concentrations of catalyst dust are not present during recharge or disposal. When unloading coked catalyst, the possibility of iron sulphide fires exists. Iron sulphide will ignite spontaneously when exposed to air, and therefore needs to be wetted down with water to prevent it from becoming a source of ignition for vapours. Coked catalyst may either be cooled to below 49 °C before dumping from the reactor, or first dumped into containers purged with inert nitrogen and then cooled before further handling.
The possibility of exposure to extremely hot hydrocarbon liquids or vapours is present during process sampling or if a leak or release occurs. In addition, exposure to carcinogenic PAHs, aromatic naphtha containing benzene, sour gas (fuel gas from processes such as catalytic cracking and hydrotreating, which contains hydrogen sulphide and carbon dioxide), hydrogen sulphide and/or carbon monoxide gas may occur during a release of product or vapour. Inadvertent formation of highly toxic nickel carbonyl may occur in cracking processes that use nickel catalysts with resultant potential for hazardous exposures.
Catalyst regeneration involves steam stripping and decoking, which results in potential exposure to fluid waste streams which may contain varying amounts of sour water, hydrocarbon, phenol, ammonia, hydrogen sulphide, mercaptan and other materials, depending upon the feedstocks, crudes and processes. Safe work practices and the use of appropriate personal protective equipment (PPE) are needed when handling spent catalyst, recharging catalyst, or if leaks or releases occur.
Hydrocracking process
Hydrocracking is a two-stage process combining catalytic cracking and hydrogenation, wherein distillate fractions are cracked in the presence of hydrogen and special catalysts to produce more desirable products. Hydrocracking has an advantage over catalytic cracking in that high-sulphur feedstocks can be processed without previous desulphurization. In the process, heavy aromatic feedstock is converted into lighter products under very high pressures and fairly high temperatures. When the feedstock has a high paraffinic content, the hydrogen prevents the formation of PAHs, reduces tar formation and prevents build-up of coke on the catalyst. Hydrocracking produces relatively large amounts of isobutane for alkylation feedstocks and also causes isomerization for pour point control and smoke point control, both of which are important in high-quality jet fuel.
In the first stage, feedstock is mixed with recycled hydrogen, heated and sent to the primary reactor, where a large amount of the feedstock is converted to middle distillates. Sulphur and nitrogen compounds are converted by a catalyst in the primary stage reactor to hydrogen sulphide and ammonia. The residual is heated and sent to a high-pressure separator, where hydrogen-rich gases are removed and recycled. The remaining hydrocarbons are stripped or purified to remove the hydrogen sulphide, ammonia and light gases, which are collected in an accumulator, where gasoline is separated from sour gas.
The stripped liquid hydrocarbons from the primary reactor are mixed with hydrogen and sent to the second-stage reactor, where they are cracked into high-quality gasoline, jet fuel and distillate blending stocks. These products go through a series of high- and low-pressure separators to remove gases, which are recycled. The liquid hydrocarbons are stabilized, split and stripped, with the light naphtha products from the hydrocracker used to blend gasoline while the heavier naphthas are recycled or sent to a catalytic reformer unit. (See figure 11.)
Figure 11. Hydrocracking process
Health and safety considerations
Inspection and testing of safety relief devices are important due to the very high pressures in this process. Proper process control is needed to protect against plugging reactor beds. Because of the operating temperatures and presence of hydrogen, the hydrogen sulphide content of the feedstock must be strictly kept to a minimum in order to reduce the possibility of severe corrosion. Corrosion by wet carbon dioxide in areas of condensation must also be considered. When processing high-nitrogen feedstocks, the ammonia and hydrogen sulphide form ammonium hydrosulphide, which causes serious corrosion at temperatures below the water dew point. Ammonium hydrosulphide is also present in sour water stripping. Because the hydrocracker operates at very high pressures and temperatures, control of both hydrocarbon leaks and hydrogen releases is important to prevent fires.
Because this is a closed process, exposures are minimal under normal operating conditions. There is a potential for exposure to aliphatic naphtha containing benzene, carcinogenic PAHs, hydrocarbon gas and vapour emissions, hydrogen-rich gas and hydrogen sulphide gas as a result of high-pressure leaks. Large quantities of carbon monoxide may be released during catalyst regeneration and changeover. Catalyst steam stripping and regeneration creates waste streams containing sour water and ammonia. Safe work practices and appropriate personal protective equipment are needed when handling spent catalyst. In some processes, care is needed to assure that explosive concentrations of catalytic dust do not form during recharging. Unloading coked catalyst requires special precautions to prevent iron sulphideinduced fires. The coked catalyst should either be cooled to below 49 °C before dumping, or placed in nitrogen-inerted containers until cooled.
Combining processes
Two combining processes, polymerization and alkylation, are used to join together small hydrogen-deficient molecules, called olefins, recovered from thermal and catalytic cracking, in order to create more desirable gasoline blending stocks.
Polymerization
Polymerization is the process of combining two or more unsaturated organic molecules (olefins) to form a single, heavier molecule with the same elements in the same proportion as the original molecule. It converts gaseous olefins, such as ethylene, propylene and butylene converted by thermal and fluid cracking units, into heavier, more complex, higher-octane molecules, including naphtha and petrochemical feedstocks. The olefin feedstock is pretreated to remove sulphur compounds and other undesirables, and then passed over a phosphorus catalyst, usually a solid catalyst or liquid phosphoric acid, where an exothermic polymeric reaction occurs. This requires the use of cooling water and the injection of cold feedstock into the reactor to control temperatures at various pressures. Acid in the liquids is removed by caustic wash, the liquids are fractionated, and the acid catalyst is recycled. The vapour is fractionated to remove butanes and neutralized to remove traces of acid. (See figure 12.)
Figure 12. Polymerization process
Severe corrosion, leading to equipment failure, will occur should water contact the phosphoric acid, such as during water washing at shutdowns. Corrosion may also occur in piping manifolds, reboilers, exchangers and other locations where acid may settle out. There is a potential for exposure to caustic wash (sodium hydroxide), to phosphoric acid used in the process or washed out during turnarounds, and to catalyst dust. The potential for an uncontrolled exothermic reaction exists should loss of cooling water occur.
Alkylation
Alkylation combines the molecules of olefins produced from catalytic cracking with those of isoparaffins in order to increase the volume and octane of gasoline blends. Olefins will react with isoparaffins in the presence of a highly active catalyst, usually sulphuric acid or hydrofluoric acid (or aluminium chloride) to create a long-branched-chain paraffinic molecule, called alkylate (iso-octane), with exceptional anti-knock quality. The alkylate is then separated and fractionated. The relatively low reaction temperatures of 10°C to 16°C for sulphuric acid, 27°C to 0°C for hydrofluoric acid (HF) and 0°C for aluminium chloride, are controlled and maintained by refrigeration. (See figure 13.)
Figure 13. Alkylation process
Sulphuric acid alkylation. In cascade-type sulphuric acid alkylation units, feedstocks, including propylene, butylene, amylene and fresh isobutane, enter the reactor, where they contact the sulphuric acid catalyst. The reactor is divided into zones, with olefins fed through distributors to each zone, and the sulphuric acid and isobutanes flowing over baffles from zone to zone. Reaction heat is removed by evaporation of isobutane. The isobutane gas is removed from the top of the reactor, cooled and recycled, with a portion directed to the depropanizer tower. Residual from the reactor is settled, and the sulphuric acid is removed from the bottom of the vessel and recirculated. Caustic and/or water scrubbers are used to remove small amounts of acid from the process stream, which then goes to a de-isobutanizer tower. The debutanizer isobutane overhead is recycled, and the remaining hydrocarbons are separated in a rerun tower and/or sent to blending.
Hydrofluoric acid alkylation. There are two types of hydrofluoric acid alkylation processes: Phillips and UOP. In the Phillips process, olefin and isobutane feedstock is dried and fed to a combination reactor/settler unit. The hydrocarbon from the settling zone is charged to the main fractionator. The main fractionator overhead goes to a depropanizer. Propane, with trace amounts of hydrofluoric acid (HF), goes to an HF stripper, and is then catalytically defluorinated, treated and sent to storage. Isobutane is withdrawn from the main fractionator and recycled to the reactor/settler, and alkylate from the bottom of the main fractionator is sent to a splitter.
The UOP process uses two reactors with separate settlers. Half of the dried feedstock is charged to the first reactor, along with recycle and make-up isobutane, and then to its settler, where the acid is recycled and the hydrocarbon charged to the second reactor. The other half of the feedstock goes to the second reactor, with the settler acid being recycled and the hydrocarbons charged to the main fractionator. Subsequent processing is similar to Phillips in that the overhead from the main fractionator goes to a depropanizer, isobutane is recycled and alkylate is sent to a splitter.
Health and safety considerations
Sulphuric acid and hydrofluoric acid are dangerous chemicals, and care during delivery and unloading of acid is essential. There is a need to maintain sulphuric acid concentrations of 85 to 95% for good operation and to minimize corrosion. To prevent corrosion from hydrofluoric acid, acid concentrations inside the process unit must be maintained above 65% and moisture below 4%. Some corrosion and fouling in sulphuric acid units occurs from the breakdown of sulphuric acid esters, or where caustic is added for neutralization. These esters can be removed by fresh-acid treating and hot-water washing.
Upsets can be caused by loss of the coolant water needed to maintain process temperatures. Pressure on the cooling water and steam side of exchangers should be kept below the minimum pressure on the acid service side to prevent water contamination. Vents can be routed to soda ash scrubbers to neutralize hydrogen fluoride gas or hydrofluoric acid vapours before release. Curbs, drainage and isolation may be provided for process unit containment so that effluent can be neutralized before release to the sewer system.
Hydrofluoric acid units should be thoroughly drained and chemically cleaned prior to turnarounds and entry, to remove all traces of iron fluoride and hydrofluoric acid. Following shutdown, where water has been used, the unit should be thoroughly dried before hydrofluoric acid is introduced. Leaks, spills or releases involving hydrofluoric acid, or hydrocarbons containing hydrofluoric acid, are extremely hazardous. Precautions are necessary to assure that equipment and materials which have been in contact with acid are handled carefully and are thoroughly cleaned before they leave the process area or refinery. Immersion wash vats are often provided for neutralization of equipment which has come into contact with hydrofluoric acid.
There is a potential for serious hazardous and toxic exposures should leaks, spills or releases occur. Direct contact with sulphuric or hydrofluoric acid will cause severe skin and eye damage, and inhalation of acid mists or hydrocarbon vapours containing acid will cause severe irritation and damage to the respiratory system. Special precautionary emergency preparedness measures should be used, and protection should be provided that is appropriate to the potential hazard and areas possibly affected. Safe work practices and appropriate skin and respiratory personal protective equipment are needed where potential exposures to hydrofluoric and sulphuric acids during normal operations exist, such as reading gauges, inspecting and process sampling, as well as during emergency response, maintenance and turnaround activities. Procedures should be in place to assure that protective equipment and clothing worn in sulphuric or hydrofluoric acid activities, including chemical protective suits, head and shoe coverings, gloves, face and eye protection and respiratory protective equipment, are thoroughly cleaned and decontaminated before reissue.
Rearranging processes
Catalytic reforming and isomerization are processes which rearrange hydrocarbon molecules to produce products with different characteristics. After cracking, some gasoline streams, although of the correct molecular size, require further processing to improve their performance, because they are deficient in some qualities, such as octane number or sulphur content. Hydrogen (steam) reforming produces additional hydrogen for use in hydrogenation processing.
Catalytic reforming
Catalytic reforming processes convert low-octane heavy naphthas into aromatic hydrocarbons for petrochemical feedstocks and high-octane gasoline components, called reformates, by molecular rearrangement or dehydrogenation. Depending on the feedstock and catalysts, reformates can be produced with very high concentrations of toluene, benzene, xylene and other aromatics useful in gasoline blending and petrochemical processing. Hydrogen, a significant by-product, is separated from the reformate for recycling and use in other processes. The resultant product depends on reactor temperature and pressure, the catalyst used and the hydrogen recycle rate. Some catalytic reformers operate at low pressure and others at high pressure. Some catalytic reforming systems continuously regenerate the catalyst, some facilities regenerate all of the reactors during turnarounds, and others take one reactor at a time off stream for catalyst regeneration.
In catalytic reforming, naphtha feedstock is pretreated with hydrogen to remove contaminants such as chlorine, sulphur and nitrogen compounds, which could poison the catalyst. The product is flashed and fractionated in towers where the remaining contaminants and gases are removed. The desulphurized naphtha feedstock is sent to the catalytic reformer, where it is heated to a vapour and passed through a reactor with a stationary bed of bi-metallic or metallic catalyst containing a small amount of platinum, molybdenum, rhenium or other noble metals. The two primary reactions which occur are production of high-octane aromatics by removing hydrogen from the feedstock molecules, and the conversion of normal paraffins to branched-chain or isoparaffins.
In platforming, another catalytic reforming process, feedstock which has not been hydrodesulphurized is combined with recycle gas and first passed over a less expensive catalyst. Any remaining impurities are converted to hydrogen sulphide and ammonia, and removed before the stream passes over the platinum catalyst. Hydrogen-rich vapour is recirculated to inhibit reactions which may poison the catalyst. The reactor output is separated into liquid reformate, which is sent to a stripping tower, and gas, which is compressed and recycled. (See figure 14.)
Figure 14. Catalytic reforming process
Operating procedures are needed to control hot spots during start-up. Care must be taken not to break or crush the catalyst when loading the beds, as small fines will plug up the reformer screens. Precautions against dust when regenerating or replacing catalyst are needed. Small emissions of carbon monoxide and hydrogen sulphide may occur during regeneration of catalyst.
Water wash should be considered where stabilizer fouling has occurred in reformers due to the formation of ammonium chloride and iron salts. Ammonium chloride may form in pretreater exchangers and cause corrosion and fouling. Hydrogen chloride, from the hydrogenation of chlorine compounds, may form acids or ammonium chloride salt. The potential exists for exposure to aliphatic and aromatic naphthas, hydrogen-rich process gas, hydrogen sulphide and benzene should a leak or release occur.
Isomerization
Isomerization converts n-butane, n-pentane and n-hexane into their respective iso-paraffins. Some of the normal straight-chain paraffin components of light straight-run naphtha are low in octane. These can be converted to high-octane, branched-chain isomers by rearranging the bonds between atoms, without changing the number or kinds of atoms. Isomerization is similar to catalytic reforming in that the hydrocarbon molecules are rearranged, but unlike catalytic reforming, isomerization just converts normal paraffins to iso-paraffins. Isomerization uses a different catalyst than catalytic reforming.
The two distinct isomerization processes are butane (C4) and pentane/hexane. (C5/C6).
Butane (C4) isomerization produces feedstock for alkylation. A lower-temperature process uses highly active aluminium chloride or hydrogen chloride catalyst without fired heaters, to isomerize n-butane. The treated and preheated feedstock is added to the recycle stream, mixed with HCl and passed through the reactor (see figure 15).
Figure 15. C4 isomerization
Pentane/hexane isomerization is used to increase the octane number by converting n-pentane and n-hexane. In a typical pentane/hexane isomerization process, dried and desulphurized feedstock is mixed with a small amount of organic chloride and recycled hydrogen, and heated to reactor temperature. It is then passed over supported-metal catalyst in the first reactor, where benzene and olefins are hydrogenated. The feed next goes to the isomerization reactor, where the paraffins are catalytically isomerized to isoparaffins, cooled and passed to a separator. Separator gas and hydrogen, with make-up hydrogen, is recycled. The liquid is neutralized with alkaline materials and sent to a stripper column, where hydrogen chloride is recovered and recycled. (See figure 16.)
Figure 16. Isomerization process
If the feedstock is not completely dried and desulphurized, the potential exists for acid formation, leading to catalyst poisoning and metal corrosion. Water or steam must not be allowed to enter areas where hydrogen chloride is present. Precautions are needed to prevent HCl from entering sewers and drains. There is a potential for exposure to isopentane and aliphatic naphtha vapours and liquid, as well as to hydrogen-rich process gas, hydrochloric acid and hydrogen chloride, and to dust when solid catalyst is used.
Hydrogen production (steam reforming)
High-purity hydrogen (95 to 99%) is needed for hydrodesulphurization, hydrogenation, hydrocracking and petrochemical processes. If not enough hydrogen is produced as by-products of refinery processes to meet the total refinery demand, the manufacture of additional hydrogen is required.
In hydrogen steam reforming, desulphurized gases are mixed with superheated steam and reformed in tubes containing a nickel base catalyst. The reformed gas, which consists of steam, hydrogen, carbon monoxide and carbon dioxide, is cooled and passed through converters where the carbon monoxide reacts with steam to form hydrogen and carbon dioxide. The carbon dioxide is scrubbed with amine solutions and vented to the atmosphere when the solutions are reactivated by heating. Any carbon monoxide remaining in the product stream is converted to methane. (See figure 17.)
Figure 17. Steam reforming process
Inspections and testing must be conducted where the possibility exists for valve failure due to contaminants in the hydrogen. Carryover from caustic scrubbers to prevent corrosion in preheaters must be controlled and chlorides from the feedstock or steam system prevented from entering reformer tubes and contaminating the catalyst. Exposures can result from contamination of condensate by process materials such as caustics and amine compounds, and from excess hydrogen, carbon monoxide and carbon dioxide. The potential exists for burns from hot gases and superheated steam should a release occur.
Miscellaneous refinery processes
Lubricant base stock and wax processes
Lubricating oils and waxes are refined from various fractions of atmospheric and vacuum distillation. With the invention of vacuum distillation, it was discovered that the waxy residuum made a better lubricant than any of the animal fats that were then in use, which was the beginning of modern hydrocarbon lubricant refining technology, whose primary objective is to remove undesirable products, such as asphalts, sulphonated aromatics and paraffinic and iso-paraffinic waxes from the residual fractions in order to produce high-quality lubricants. This is done by a series of processes including de-asphalting, solvent extraction and separation and treatment processes such as dewaxing and hydrofinishing. (See figure 18)
Figure 18. Lubricating oil & wax manufacturing process
In extraction processing, reduced crude from the vacuum unit is propane de-asphalted and combined with straight-run lubricating-oil feedstock, preheated and solvent extracted to produce a feedstock called raffinate. In a typical extraction process which uses phenol as the solvent, the feedstock is mixed with phenol in the treating section at temperatures below 204 °C. Phenol is then separated from the raffinate and recycled. The raffinate may then be subjected to another extraction process which uses furfural to separate aromatic compounds from non-aromatic hydrocarbons, producing a lighter-coloured raffinate with improved viscosity index and oxidation and thermal stability.
Dewaxed raffinate may also be subject to further processing to improve the qualities of the base stock. Clay adsorbents are used to remove dark-coloured, unstable molecules from lubricating-oil base stocks. An alternate process, lube hydrofinishing, passes hot dewaxed raffinate and hydrogen through a catalyst that slightly changes the molecular structure, resulting in a lighter-coloured oil with improved characteristics. The treated lube oil base stocks are then mixed and/or compounded with additives to meet the required physical and chemical characteristics of motor oils, industrial lubricants and metal-working oils.
The two distinct types of wax derived from crude oil are paraffin wax, produced from distillate stocks, and microcrystalline wax, manufactured from residual stocks. Raffinate from the extraction unit contains a considerable amount of wax, which can be removed by solvent extraction and crystallization. The raffinate is mixed with a solvent, such as propane, methyl ethyl ketone (MEK) and toluene mixture or methyl isobutyl ketone (MIBK), and precooled in heat exchangers. The crystallization temperature is attained by the evaporation of the propane in the chiller and filter feed tanks. The wax is continuously removed by filters and cold solvent washed to recover retained oil. The solvent is recovered from the dewaxed raffinate by flashing and steam stripping, and recycled.
The wax is heated with hot solvent, chilled, filtered and given a final wash to remove all traces of oil. Before the wax is used, it may be hydro-finished to improve its odour and eliminate all traces of aromatics so the wax can be used in food processing. The dewaxed raffinate, which contains small amounts of paraffins, naphthenes and some aromatics, may be further processed for use as lubricating-oil base stocks.
Control of treater temperature is important to prevent corrosion from phenol. Wax can clog sewer or oil drainage systems and interfere with waste water treatment. The potential exists for exposure to process solvents such as phenol, propane, a methyl ethyl ketone and toluene mixture or methyl isobutyl ketone. Inhalation of hydrocarbon gases and vapours, aromatic naphtha containing benzene, hydrogen sulphide and hydrogen-rich process gas is a hazard.
Asphalt processing
After primary distillation operations, asphalt is a portion of residual matter which requires further processing to impart characteristics required by its final use. Asphalt for roofing materials is produced by air blowing. Residual is heated in a pipe still almost up to its flashpoint and charged to a blowing tower where hot air is injected for a predetermined period of time. The dehydrogen ation of the asphalt forms hydrogen sulphide, and the oxidation creates sulphur dioxide. Steam is used to blanket the top of the tower to entrain the contaminants, and is passed through a scrubber to condense the hydrocarbons.
Vacuum distillation is generally used to produce road tar asphalt. The residual is heated and charged to a column where vacuum is applied to prevent cracking.
Condensed steam from the various asphalt processes will contain trace amounts of hydrocarbons. Any disruption of the vacuum can result in the entry of atmospheric air and subsequent fire. In asphalt production, raising the temperature of the vacuum tower bottom to improve efficiency can generate methane by thermal cracking. This creates vapours in asphalt storage tanks which are in the flammable range, but not detectable by flash testing. Air blowing can create some polynuclear aromatics (i.e., PAHs). Condensed steam from the air blowing asphalt process may also contain various contaminants.
Hydrocarbon sweetening and treating processes
Many products, such as thermal naphthas from visbreaking, coking or thermal cracking, and high-sulphur naphthas and distillates from crude-oil distillation, require treating in order to be used in gasoline and fuel oil blends. Distillation products, including kerosene and other distillates, may contain trace amounts of aromatics, and naphthenes and lubricating-oil base stocks may contain wax. These undesirables are removed either at intermediate refining stages or just prior to sending products to blending and storage, by refining processes such as solvent extraction and solvent dewaxing. A variety of intermediate and finished products, including middle distillates, gasoline, kerosene, jet fuel and sour gases need to be dried and sweetened.
Treating is performed either at an intermediate stage in the refining process or just before sending finished products to blending and storage. Treating removes contaminants from oil, such as organic compounds containing sulphur, nitrogen and oxygen, dissolved metals, inorganic salts and soluble salts dissolved in emulsified water. Treating materials include acids, solvents, alkalis and oxidizing and adsorption agents. Acid treatments are used to improve the odour, colour and other properties of lube base stocks, to prevent corrosion and catalyst contamination, and to improve product stability. Hydrogen sulphide which is removed from “dry” sour gas by an absorbing agent (diethanolamine) is flared, used as a fuel or converted to sulphur. The type of treatment and agents depends on the crude feedstock, intermediate processes and end-product specifications.
Solvent treatment processes
Solvent extraction separates aromatics, naphthenes and impurities from product streams by dissolving or precipitation. Solvent extraction prevents corrosion, protects catalyst in subsequent processes and improves finished products by removing unsaturated, aromatic hydrocarbons from lubricant and grease base stocks.
The feedstock is dried and subjected to continuous countercurrent solvent treatment. In one process, feedstock is washed with a liquid in which the substances to be removed are more soluble than in the desired resultant product. In another process, selected solvents are added, causing impurities to precipitate out of the product. The solvent is separated from the product stream by heating, evaporation or fractionation, with residual trace amounts subsequently removed from the raffinate by steam stripping or vacuum flashing. Electric precipitation may be used for separation of inorganic compounds. The solvent is then regenerated to be used again in the process.
Typical chemicals used in the extraction process include a wide variety of acids, alkalis and solvents, including phenol and furfural, as well as oxidizing agents and adsorption agents. In the adsorption process, highly porous solid materials collect liquid molecules on their surfaces. The selection of specific processes and chemical agents depends on the nature of the feedstock being treated, the contaminants present and the finished product requirements. (See figure 19.)
Figure 19. Solvent extraction process
Solvent dewaxing removes wax from either distillate or residual base stocks, and may be applied at any stage in the refining process. In solvent dewaxing, waxy feedstocks are chilled by heat exchanger and refrigeration, and solvent is added to help develop crystals that are removed by vacuum filtration. The dewaxed oil and solvent are flashed and stripped, and the wax passes through a water settler, solvent fractionator and flash tower. (See figure 20.)
Figure 20. Solvent dewaxing process
Solvent de-asphalting separates heavy oil fractions to produce heavy lubricating oil, catalytic cracking feedstock and asphalt. Feedstock and liquid propane (or hexane) are pumped to an extraction tower at precisely controlled mixtures, temperatures and pressures. Separation occurs in a rotating-disc contactor, based on differences in solubility. The products are then evaporated and steam stripped to recover propane for recycle. Solvent de-asphalting also removes sulphur and nitrogen compounds, metals, carbon residues and paraffins from feedstock. (See figure 21.)
Figure 21. Solvent de-asphalting process
Health and safety considerations.
In solvent dewaxing, disruption of the vacuum will create a potential fire hazard by allowing air to enter the unit. The potential exists for exposure to dewaxing solvent vapours, a mixture of MEK and toluene. Although solvent extraction is a closed process, there is potential exposure to carcinogenic PAHs in the process oils and to extraction solvents such as phenol, furfural, glycol, MEK, amines and other process chemicals during handling and operations.
De-asphalting requires exact temperature and pressure control to avoid upset. In addition, moisture, excess solvent or a drop in operating temperature may cause foaming which affects the product temperature control and may create an upset. Contact with hot oil streams will cause skin burns. The potential exists for exposure to hot oil streams containing carcinogenic polycyclic aromatic compounds, liquefied propane and propane vapours, hydrogen sulphide and sulphur dioxide.
Hydrotreating processes
Hydrotreating is used to remove about 90% of contaminants, including nitrogen, sulphur, metals and unsaturated hydrocarbons (olefins), from liquid petroleum fractions such as straight-run gasoline. Hydrotreating is similar to hydrocracking in that both the hydrogen and the catalyst are used to enrich the hydrogen content of the olefin feedstock. However, the degree of saturation is not as great as that achieved in hydrocracking. Typically, hydrotreating is done prior to processes such as catalytic reforming, so that the catalyst is not contaminated by untreated feedstock. Hydrotreating is also used before catalytic cracking to reduce sulphur and improve product yields, and to upgrade middle distillate petroleum fractions into finished kerosene, diesel fuel and heating fuel oils.
Hydrotreating processes differ depending upon the feedstocks and catalysts. Hydrodesulphurization removes sulphur from kerosene, reduces aromatics and gum-forming characteristics, and saturates any olefins. Hydroforming is a dehydrogenation process used to recover excess hydrogen and produce high-octane gasoline. Hydrotreated products are blended or used as catalytic reforming feedstock.
In catalytic hydrodesulphurization, the feedstock is de-aerated, mixed with hydrogen, preheated and charged under high pressure through a fixed-bed catalytic reactor. The hydrogen is separated and recycled and the product stabilized in a stripper column where the light ends are removed.
During this process, sulphur and nitrogen compounds present in the feedstock are converted to hydrogen sulphide (H2S) and ammonia (NH3). Residual hydrogen sulphide and ammonia are removed either by steam stripping, by a combination high- and low-pressure separator or by amine wash which recovers hydrogen sulphide in a highly concentrated stream suitable for conversion into elemental sulphur. (See figure 22 and figure 23.)
Figure 22. Hydrodesulphurization process
Figure 23. Schematic of hydrodesulphurization process
In hydrotreating, the hydrogen sulphide content of the feedstock must be strictly controlled to a minimum to reduce corrosion. Hydrogen chloride may form and condense as hydrochloric acid in the lower-temperature portions of the unit. Ammonium hydrosulphide may form in high-temperature, high-pressure units. In the event of a release, there is a potential for exposure to aromatic naphtha vapours which contain benzene, hydrogen sulphide or hydrogen gas, or to ammonia should a sour water leak or spill occur. Phenol may also be present if high-boiling-point feedstocks are processed.
Excessive contact time and/or temperature will create coking in the unit. Precautions need to be taken when unloading coked catalyst from the unit to prevent iron sulphide fires. The coked catalyst should be cooled to below 49 °C before removal, or dumped into nitrogen-inerted bins where it can be cooled before further handling. Special anti-foam additives may be used to prevent catalyst poisoning from silicone carryover in coker feedstock.
Other sweetening and treating processes
Treatment, drying and sweetening processes are used to remove impurities from blending stocks. (See figure 24.)
Figure 24. Sweetening & treating processes
Sweetening processes use air or oxygen. If excess oxygen enters these processes, it is possible for a fire to occur in the settler due to the generation of static electricity. There is a potential for exposure to hydrogen sulphide, sulphur dioxide, caustic (sodium hydroxide), spent caustic, spent catalyst (Merox), catalyst dust and sweetening agents (sodium carbonate and sodium bicarbonate).
Amine (acid gas treatment) plants
Sour gas (fuel gas from processes such as catalytic cracking and hydrotreating, which contains hydrogen sulphide and carbon dioxide) must be treated before it can be used as refinery fuel. Amine plants remove acid contaminants from sour gas and hydrocarbon streams. In amine plants, gas and liquid hydrocarbon streams containing carbon dioxide and/or hydrogen sulphide are charged to a gas absorption tower or liquid contactor, where the acid contaminants are absorbed by counterflowing amine solutions—monoethanolamine (MEA), diethanolamine (DEA) or methyldiethanolamine (MDEA). The stripped gas or liquid is removed overhead, and the amine is sent to a regenerator. In the regenerator, the acidic components are stripped by heat and reboiling action, and disposed of, while the amine is recycled.
In order to minimize corrosion, proper operating practices should be established, and regenerator bottom and reboiler temperatures need to be controlled. Oxygen should be kept out of the system to prevent amine oxidation. There is potential for exposure to amine compounds (i.e., MEA, DEA, MDEA), hydrogen sulphide and carbon dioxide.
Sweetening and drying
Sweetening (mercaptan removal) treats sulphur compounds (hydrogen sulphide, thiophene and mercaptan) to improve colour, odour and oxidation stability, and reduces concentrations of carbon dioxide in gasoline. Some mercaptans are removed by having the product make contact with water-soluble chemicals (e.g., sulphuric acid) that react with the mercaptans. Caustic liquid (sodium hydroxide), amine compounds (diethanolamine) or fixed-bed catalyst sweetening may be used to convert mercaptans to less objectionable disulphides.
Product drying (water removal) is accomplished by water absorption, with or without adsorption agents. Some processes simultaneously dry and sweeten by adsorption on molecular sieves.
Sulphur recovery
Sulphur recovery removes hydrogen sulphide from sour gases and hydrocarbon streams. The Clause process converts hydrogen sulphide to elemental sulphur through the use of thermal and catalytic reactions. After burning hydrogen sulphide under controlled conditions, knockout pots remove water and hydrocarbons from feed-gas streams, which are then exposed to a catalyst to recover additional sulphur. The sulphur vapour from burning and conversion is condensed and recovered.
Tail gas treatment
Both oxidation and reduction are used to treat tail gas from sulphur recovery units, depending on the composition of the gas and on refinery economics. Oxidation processes burn tail gas to convert all sulphur compounds to sulphur dioxide, and reduction processes convert sulphur compounds to hydrogen sulphide.
Hydrogen sulphide scrubbing
Hydrogen sulphide scrubbing is a primary hydrocarbon feedstock treating process used to prevent catalyst poisoning. Depending on the feedstock and the nature of the contaminants, desulphurization methods will vary from ambient-temperature-activated charcoal absorption to high-temperature catalytic hydrogenation followed by zinc oxide treating.
Sat and unsat gas plants
Feedstocks from various refinery units are sent to gas treating plants, where butanes and butenes are removed for use as alkylation feedstock, heavier components are sent to gasoline blending, propane is recovered for LPG and propylene is removed for use in petrochemicals.
Sat gas plants separate components from refinery gases, including butanes for alkylation, pentanes for gasoline blending, LPGs for fuel and ethane for petrochemicals. There are two different sat gas processes: absorption-fractionation or straight fractionation. In absorption-fractionation, gases and liquids from various units are fed to an absorber/de-ethanizer where C2 and lighter fractions are separated by lean-oil absorption and removed for use as fuel gas or petrochemical feed. The remaining heavier fractions are stripped and sent to a debutanizer, and the lean oil is recycled back to the absorber/de-ethanizer. C3/C4 is separated from pentanes in the debutanizer, scrubbed to remove hydrogen sulphide, and fed to a splitter to separate propane and butane. The absorption stage is eliminated in fractionation plants. Sat gas processes depend on feedstock and product demand.
Corrosion occurs from the presence of hydrogen sulphide, carbon dioxide and other compounds as a result of prior treating. Streams containing ammonia should be dried before processing. Anti-fouling additives are used in absorption oil to protect heat exchangers. Corrosion inhibitors are used to control corrosion in overhead systems. The potential exists for exposure to hydrogen sulphide, carbon dioxide, sodium hydroxide, MEA, DEA and MDEA to be carried over from prior treating.
Unsat gas plants recover light hydrocarbons from wet gas streams from catalytic crackers and delayed coker overhead accumulators or fractionation receivers. In a typical process, wet gases are compressed and treated with amine to remove hydrogen sulphide either before or after entering a fractionating absorber, where they mix into a concurrent flow of debutanized gasoline. The light fractions are separated by heat in a reboiler, with the offgas sent to a sponge absorber and the bottoms sent to a debutanizer. A portion of the debutanized hydrocarbon is recycled, and the balance goes to a splitter for separation. Overhead gases go to a depropanizer for use as alkylation unit feedstock. (See figure 25.)
Figure 25. Unsat gas plant process
Corrosion can occur from moist hydrogen sulphide and cyanides in unsat gas plants which handle FCC feedstocks. Corrosion from hydrogen sulphide and deposits in the high-pressure sections of gas compressors from ammonium compounds is possible when feedstocks are from the delayed coker or the TCC. The potential exists for exposure to hydrogen sulphide and to amine compounds such as MEA, DEA and MDEA.
Gasoline, distillate fuel and lubricant base stock blending processes
Blending is the physical mixture of a number of different liquid hydrocarbon fractions to produce finished products with specific desired characteristics. Products can be blended in-line through a manifold system or batch blended in tanks and vessels. In-line blending of gasoline, distillates, jet fuel and lubricant base stocks is accomplished by injecting proportionate amounts of each component into the main stream where turbulence promotes thorough mixing.
- Gasolines are blends of reformates, alkylates, straight-run gasoline, thermal and catalytically cracked gasolines, coker gasoline, butane and appropriate additives.
- Fuel oil and diesel fuel are blends of distillates and cycle oils, and jet fuel may be straight-run distillate or blended with naphtha.
- Lubricating oils are blends of refined base stocks
- Asphalt is blended from various residual stocks depending on its intended use.
Additives are often mixed into gasoline and motor fuels during or after blending to provide specific properties not inherent in petroleum hydrocarbons. These additives include octane enhancers, anti-knock agents, anti-oxidants, gum inhibitors, foam inhibitors, rust inhibitors, carburettor (carbon) cleaners, detergents for injector cleaning, diesel odourizers, colour dyes, distillate anti-static, gasoline oxidizers such as methanol, ethanol and methyl tertiary butyl ether, metal deactivators and others.
Batch and in-line blending operations require strict controls to maintain desired product quality. Spills should be cleaned and leaks repaired to avoid slips and falls. Additives in drums and bags need to be handled properly to avoid strain and exposure. The potential for contacting hazardous additives, chemicals, benzene and other materials exists during blending, and appropriate engineering controls, personal protective equipment and proper hygiene are needed to minimize exposures.
Auxiliary Refinery Operations
Auxiliary operations supporting refinery processes include those which provide process heat and cooling; provide pressure relief; control air emissions; collect and treat waste water; provide utilities such as power, steam, air and plant gases; and pump, store, treat and cool process water.
Waste water treatment
Refinery waste water includes condensed steam, stripping water, spent caustic solutions, cooling tower and boiler blowdown, wash water, alkaline and acid waste neutralization water and other process-associated water. Waste water typically contains hydrocarbons, dissolved materials, suspended solids, phenols, ammonia, sulphides and other compounds. Waste water treatment is used for process water, runoff water and sewerage water prior to their discharge. These treatments may require permits, or there must be recycling.
The potential exists for fire should vapours from waste water containing hydrocarbons reach a source of ignition during the treatment process. The potential exists for exposure to the various chemicals and waste products during process sampling, inspection, maintenance and turnarounds.
Pretreatment
Pretreatment is the initial separation of hydrocarbons and solids from waste water. API separators, interceptor plates and settling ponds are used to remove suspended hydrocarbons, oily sludge and solids by gravity separation, skimming and filtration. Acidic waste water is neutralized with ammonia, lime or soda ash. Alkaline waste water is treated with sulphuric acid, hydrochloric acid, carbon dioxide-rich flue gas or sulphur. Some oil-in-water emulsions are first heated to help separate the oil and the water. Gravity separation depends on the different specific gravities of water and immiscible oil globules, which allows free oil to be skimmed off the surface of the waste water.
Sour water stripping
Water containing sulphides, called sour water, is produced in catalytic cracking and hydro-treating processes, and whenever steam is condensed in the presence of gases containing hydrogen sulphide.
Stripping is used on waste water containing sulphides and/or ammonia, and solvent extraction is used to remove phenols from waste water. Waste water which is to be recycled may require cooling to remove heat and/or oxidation by spraying or air stripping to remove any remaining phenols, nitrates and ammonia.
Secondary treatment
Following pretreatment, suspended solids are removed by sedimentation or air flotation. Waste water with low levels of solids is screened or filtered, and flocculation agents may be added to help separation. Materials with high adsorption characteristics are used in fixed-bed filters or added to the waste water to form a slurry which is removed by sedimentation or filtration. Secondary treatment processes biologically degrade and oxidize soluble organic matter by the use of activated sludge, unaerated or aerated lagoons, trickling filter methods or anaerobic treatments. Additional treatment methods are used to remove oils and chemicals from waste water.
Tertiary treatment
Tertiary treatments remove specific pollutants in order to meet regulatory discharge requirements. These treatments include chlorination, ozonation, ion exchange, reverse osmosis, activated carbon adsorption, and others. Compressed oxygen may be diffused into waste water streams to oxidize certain chemicals or to satisfy regulatory oxygen content requirements.
Cooling towers
Cooling towers remove heat from process water by evaporation and latent heat transfer between hot water and air. The two types of towers are counterflow and crossflow.
- In counterflow cooling, hot process water is pumped to the uppermost plenum and allowed to fall through the tower. Numerous slats, or spray nozzles, are located throughout the length of the tower to disperse the water flow and help in cooling. Simultaneously, air enters at the tower bottom, creating a concurrent flow of air against the water. Induced draft towers have the fans at the air outlet. Forced draft towers have the fans or blowers at the air inlet.
- Crossflow towers introduce airflow at right angles to the water flow throughout the structure.
Recirculated cooling water must be treated to remove impurities and any dissolved hydrocarbons. Impurities in cooling water can corrode and foul piping and heat exchangers, scale from dissolved salts can deposit on pipes, and wooden cooling towers can be damaged by micro-organisms.
Cooling tower water can be contaminated by process materials and by-products, including sulphur dioxide, hydrogen sulphide and carbon dioxide, with resultant exposures. There is potential for exposure to water treatment chemicals or to hydrogen sulphide when waste water is treated in conjunction with cooling towers. Because the water is saturated with oxygen from being cooled with air, the chances for corrosion are intensified. One means of corrosion prevention is the addition of a material to the cooling water which forms a protective film on pipes and other metal surfaces.
When cooling water is contaminated by hydrocarbons, flammable vapours can evaporate into the discharge air. If a source of ignition or lightning is present, fires may start. Fire hazards exist when there are relatively dry areas in induced-draft cooling towers of combustible construction. Loss of power to cooling tower fans or water pumps can create serious consequences in process operations.
Steam generation
Steam is produced through heater and boiler operations in central steam generation plants and at various process units, using heat from flue gas or other sources. Steam generation systems include:
- heaters (furnaces), with their burners and a combustion air system
- draft or pressure systems to remove flue gas from the furnace, soot blowers, and compressed air systems which seal openings to prevent flue gas from escaping
- boilers, consisting of a number of tubes which carry the water/steam mixture through the furnace providing for maximum heat transfer (these tubes run between steam distribution drums at the top of the boiler, and water collecting drums at the bottom of the boiler)
- steam drums to collect steam and direct it to the superheater before it enters the steam distribution system.
The most potentially hazardous operation in steam generation is heater start-up. A flammable mixture of gas and air can build up as a result of loss of flame at one or more burners during light-off. Specific start-up procedures are required for each different type of unit, including purging before light-off and emergency procedures in the event of misfire or loss of burner flame. If feedwater runs low and boilers are dry, the tubes will overheat and fail. Excess water will be carried over into the steam distribution system, causing damage to the turbines. Boilers should have continuous or intermittent blowdown systems to remove water from steam drums and to limit build-up of scale on turbine blades and superheater tubes. Care must be taken not to overheat the superheater during start-up and shut down. Alternate fuel sources should be provided in event of loss of fuel gas due to refinery unit shutdown or emergency.
Heater fuel
Any one or any combination of fuels, including refinery gas, natural gas, fuel oil and powdered coal may be used in heaters. Refinery off-gas is collected from process units and combined with natural gas and LPG in a fuel gas balance drum. The balance drum provides constant system pressure, fairly stable BTU (energy) content fuel and automatic separation of suspended liquids in gas vapours, and prevents carryover of large slugs of condensate into the distribution system.
Fuel oil is typically a mix of refinery crude oil and straight-run and cracked residues, blended with other products. The fuel oil system delivers fuel to process unit heaters and steam generators at required temperatures and pressures. The fuel oil is heated to pumping temperature, sucked through a coarse suction strainer, pumped to a temperature-control heater and then through a fine mesh strainer before being burned. Knockout pots, provided at process units, are used to remove liquids from fuel gas before burning.
In one example of process unit heat generation, carbon monoxide (CO) boilers recover heat in catalytic cracking units as carbon monoxide in flue gas is burned to complete combustion. In other processes, waste heat recovery units use heat from the flue gas to make steam.
Steam distribution
Steam typically is generated by heaters and boilers combined into one unit. Steam leaves the boilers at the highest pressure required by the process units or the electrical generator. The steam pressure is then reduced in turbines which drive process pumps and compressors. When refinery steam is also used to drive steam turbine generators to produce electricity, the steam must be produced at much higher pressure than required for process steam. The steam distribution system consists of valves, fittings, piping and connections which are suitable for the pressure of the steam transported. Most steam used in the refinery is condensed to water in heat exchangers and reused as boiler feedwater, or discharged to waste water treatment.
Steam feedwater
Feedwater supply is an important part of steam generation. There must always be as many pounds of water entering the steam generation system as there are pounds of steam leaving it. Water used in steam generation must be free of contaminants, including minerals and dissolved impurities, which can damage the system or affect the operation. Suspended materials such as silt, sewage and oil, which form scale and sludge, are coagulated or filtered out of the water. Dissolved gases, particularly carbon dioxide and oxygen which cause boiler corrosion, are removed by de-aeration and treatment. Dissolved minerals such as metallic salts, calcium and carbonates, which cause scale, corrosion and turbine blade deposits, are treated with lime or soda ash to precipitate them out of the water. Depending on its characteristics, raw boiler feedwater may be treated by clarification, sedimentation, filtration, ion exchange, de-aeration and internal treatment. Recirculated cooling water must also be treated to remove hydrocarbons and other contaminants.
Process heaters, heat exchangers and coolers
Process heaters and heat exchangers preheat feedstocks in distillation towers and in refinery processes to reaction temperatures. The major portion of heat provided to process units comes from fired heaters found on crude and reformer preheater units, coker heaters and large-column reboilers, which are fueled by refinery or natural gas, distillate and residual oils. Heaters are usually designed for specific process operations, and most are either cylindrical vertical or box-type designs. Heat exchangers use either steam or hot hydrocarbon, transferred from some other section of the process, for heat input.
Heat is also removed from some processes by air and water exchangers, fin fans, gas and liquid coolers and overhead condensers, or by transferring the heat to other systems. The basic mechanical vapour compression refrigeration system is designed to serve one or more process units, and includes an evaporator, compressor, condenser, controls and piping. Common coolants are water, alcohol/water mixture or various glycol solutions.
A means of providing adequate draft or steam purging is required to reduce the chance of explosions when lighting fires in heater furnaces. Specific start-up and emergency procedures are required for each type of unit. If fire impinges on fin fans, failure could occur due to overheating. If flammable product escapes from a heat exchanger or cooler due to a leak, a fire could occur.
Care must be taken to assure that all pressure is removed from heater tubes before removing any header or fitting plugs. Consideration should be given to providing for pressure relief in heat exchanger piping systems in the event they are blocked off while full of liquid. If controls fail, variations of temperature and pressure could occur on either side of the heat exchanger. If heat exchanger tubes fail and process pressure is greater than heater pressure, product could enter the heater with downstream consequences. If the pressure is less, the heater stream could enter into the process fluid stream. If loss of circulation occurs in liquid or gas coolers, increased product temperature could affect downstream operations, requiring pressure relief.
Depending on the fuel, process operation and unit design, there is a potential for exposure to hydrogen sulphide, carbon monoxide, hydrocarbons, steam boiler feedwater sludge and water treatment chemicals. Skin contact with boiler blowdown which may contain phenolic compounds should be avoided. Exposure to radiant heat, superheated steam and hot hydrocarbons is possible.
Pressure relief and flare systems
Engineering controls which are incorporated into processes include reducing flammable vapour concentrations by ventilation, dilution and inerting. Pressurization is used to maintain control rooms above atmospheric pressure in order to reduce the possibility of vapours entering. Pressure relief systems are provided to control vapours and liquids which are released by pressure-relieving devices and blowdowns. Pressure relief is an automatic, planned release when operating pressure reaches a predetermined level. Blowdown usually refers to the intentional release of material, such as blowdowns from process unit start-ups, furnace blowdowns, shutdowns and emergencies. Vapour depressuring is the rapid removal of vapours from pressure vessels in case of emergency. This may be accomplished by the use of a rupture disc, usually set at a higher pressure than the relief valve.
Safety relief valves
Safety relief valves, used to control air, steam, gas and hydrocarbon vapour and liquid pressures, open in proportion to the increase in pressure over the normal operating pressure. Safety valves, designed primarily to release high volumes of steam, usually pop open to full capacity. The overpressure needed to open liquid relief valves, where large-volume discharge is not required, increases as the valve lifts due to increased spring resistance. Pilot-operated safety release valves, with up to six times the capacity of normal relief valves, are used where tighter sealing and larger-volume discharges are required. Non-volatile liquids are usually pumped to oil/water separation and recovery systems, and volatile liquids are sent to units operating at a lower pressure.
Flares
A typical closed pressure-release and flare system includes relief valves and lines from process units for collection of discharges, knockout drums to separate vapours and liquids, seals and/or purge gas for flashback protection and a flare and igniter system, which combusts vapours if discharge direct to the atmosphere is not permitted. Steam may be injected into the flare tip to reduce visible smoke.
Liquids should not be allowed to discharge to a vapour disposal system. Flare knockout drums and flares need to be large enough to handle emergency blowdowns, and drums require relief in event of overpressure. Provide pressure relief valves where the potential exists for overpressure in refinery processes, such as due to the following causes:
- loss of cooling water, possibly resulting in a greatly increased pressure drop in condensers, in turn increasing the pressure in the process unit
- rapid vaporization and pressure increase from injection of a lower-boiling-point liquid, including water, into a process vessel operating at higher temperatures
- expansion of vapour and resultant overpressure due to overheated process steam, malfunctioning heaters or fire
- failure of automatic controls, closed outlets, heat exchanger failure, etc.
- internal explosion, chemical reaction, thermal expansion, accumulated gases, etc.
- loss of reflux, causing a pressure rise in distillation towers.
Because the quantity of reflux affects the volume of vapours leaving the distillation tower, loss of volume causes a pressure drop in condensers and a pressure rise in distillation towers.
Maintenance is important because valves are required to function properly. Common valve operating problems include:
- failure to open at set pressure due to plugging of the valve inlet or outlet or by corrosion, preventing proper operation of the disc holder and guides
- failure to reseat after popping open due to fouling, corrosion or deposits on the seat or moving parts, or by solids in the gas stream cutting the valve disc
- chattering and premature opening, due to operating pressure being too close to the valve set point.
Utilities
Water. Depending on location and community resources, refineries may draw upon public water supplies for drinking and process water or may have to pump and treat their own potable water. Treatment may include a wide range of requirements, from desalting to filtration, chlorination and testing.
Sewage. Also, depending on availability of community or private offsite treatment plants, refineries may have to provide for the permitting, collection, treatment and discharge of their sanitary waste.
Electric power. Refineries either receive electricity from outside sources or produce their own, using electric generators driven by steam turbines or gas engines. Areas are classified with regard to the type of electrical protection required to prevent a spark from igniting vapours or contain an explosion within electrical equipment. Electrical substations, which are normally located in non-classified areas, away from sources of flammable hydrocarbon vapour or cooling tower water spray, contain transformers, circuit breakers and feed circuit switches. Substations feed power to distribution stations within the process unit areas. Distribution stations can be located in classified areas, provided that electrical classification requirements are met. Distribution stations typically use a liquid-filled transformer provided with an oil-filled or air-break disconnect device.
Normal electrical safety precautions, including dry footing, “high voltage” warning signs and guarding should be implemented to protect against electrocution. Employees should be familiar with refinery electrical safe work procedures. Lockout/tagout and other appropriate safe work practices should be implemented to prevent energizing while work is being performed on high-voltage electrical equipment. Hazardous exposures may occur when working around transformers and switches which contain a dielectric fluid requiring special handling precautions. These subjects are discussed more fully elsewhere in this Encyclopaedia.
Turbine, gas and air compressor operations
Air and gas compressors
Refinery exhaust ventilation and air supply systems are designed to capture or dilute gases, fumes, dusts and vapours which may contaminate working spaces or the outside atmosphere. Captured contaminants are reclaimed if feasible, or directed to disposal systems after being cleaned or burned. Air supply systems include compressors, coolers, air receivers, air dryers, controls and distribution piping. Blowers are also used to provide air to certain processes. Plant air is provided for the operation of air-powered tools, catalyst regeneration, process heaters, steam-air decoking, sour water oxidation, gasoline sweetening, asphalt blowing and other uses. Instrument air is provided for use in pneumatic instruments and controls, air motors and purge connections. Plant gas, such as nitrogen, is provided for inerting vessels and other uses. Both reciprocating and centrifugal compressors are used for gas and compressed air.
Air compressors should be located so that the suction does not take in flammable vapours or corrosive gases. There is a potential for fire should a leak occur in gas compressors. Knockout drums are needed to prevent liquid surges from entering gas compressors. If gases are contaminated with solid materials, strainers are needed. Failure of automatic compressor controls will affect processes. If maximum pressure could potentially be greater than compressor or process equipment design pressure, pressure relief should be provided. Guarding is needed for exposed moving parts on compressors. Compressor buildings should be properly electrically classified, and provisions made for proper ventilation.
Where plant air is used as back-up to instrument air, interconnections must be upstream of the instrument air drying system to prevent contamination of instruments with moisture. Alternate sources of instrument air supply, such as use of nitrogen, may be needed in the event of power outages or compressor failure. Apply appropriate safeguards so that gas, plant air and instrument air are not used as the source for breathing or for pressuring potable water systems.
Turbines
Turbines are usually gas or steam powered and are used to drive pumps, compressors, blowers and other refinery process equipment. Steam enters turbines at high temperatures and pressures, expanding across and driving rotating blades while directed by fixed blades.
Steam turbines used for exhaust operating under vacuum need a safety relief valve on the discharge side for protection and to maintain steam in event of vacuum failure. Where maximum operating pressure could be greater than design pressure, steam turbines need relief devices. Consideration should be given to providing governors and overspeed-control devices on turbines.
Pumps, Piping and Valves
Centrifugal and positive displacement (reciprocating) pumps are used to move hydrocarbons, process water, fire water and waste water throughout the refinery. Pumps are driven by electric motors, steam turbines or internal combustion engines.
Process and utility piping systems distribute hydrocarbons, steam, water and other products throughout the facility. They are sized and constructed of materials dependent on the type of service, pressure, temperature and nature of the products. There are vent, drain and sample connections on piping, as well as provisions for blanking. Different types of valves, including gate valves, bypass valves, globe and ball valves, plug valves, block and bleed valves and check valves are used, depending on their operating purpose. These valves can be operated manually or automatically.
Valves and instrumentation which require servicing or other work should be accessible at grade level or from an operating platform. Remote-controlled valves, fire valves and isolation valves may be used to limit the loss of product at pump suction lines in the event of leakage or fire. Operating vent and drain connections may be provided with double block valves, or a block valve and plug or blind flange for protection against releases. Depending on the product and service, backflow prevention from the discharge line may be needed. Provisions may be made for pipeline expansion, movement and temperature changes to avoid rupture. Pumps operated with reduced or no flow can overheat and rupture. The failure of automatic pump controls could cause a deviation in process pressure and temperature. Pressure relief in the discharge piping should be provided where pumps can be overpressured.
Tank storage
Atmospheric storage tanks and pressure storage tanks are used throughout the refinery for storage of crudes, intermediate hydrocarbons (those used for processing) and finished products, both liquids and gases. Tanks are also provided for fire water, process and treatment water, acids, air and hydrogen, additives and other chemicals. The type, construction, capacity and location of tanks depends on their use and the nature, vapour pressure, flashpoints and pour points of the materials stored. Many types of tanks are used in refineries, the simplest being above-ground, cone-roof tanks for storage of combustible (non-volatile) liquids such as diesel fuels, fuel oils and lubricating oils. Open-top and covered (internal) floating-roof tanks, which store flammable (volatile) liquids such as gasoline and crude oil, restrict the amount of space between the top of the product and the tank roof in order to maintain a vapour-rich atmosphere to preclude ignition.
The potential for fire exists if hydrocarbon storage tanks are overfilled or develop leaks which allow liquid and vapours to escape and reach sources of ignition. Refineries should establish manual gauging and product receipt procedures to control overfills or provide automatic overflow control and signaling systems on tanks. Tanks may be equipped with fixed or semi-fixed foam-water fire protection systems. Remote-controlled valves, isolation valves and fire valves may be provided at tanks for pump-out or closure in the event of a fire inside the tank or in the tank dike or storage area. Tank venting, cleaning and confined-space entry programmes are used to control work inside tanks, and hot work permit systems are used to control sources of ignition in and around storage tanks.
Handling, shipping and transportation
Loading gases and liquid hydrocarbons into pipelines, tank cars, tank trucks and marine vessels and barges for transport to terminals and consumers is the final refinery operation. Product characteristics, distribution needs, shipping requirements, fire prevention, and environmental protection and operating criteria are important when designing marine docks, loading racks and pipeline manifolds. Operating procedures need to be established and agreed to by the shipper and receiver, and communications maintained during product transfer. Tank trucks and rail tank cars may be either top or bottom loaded. Loading and unloading liquefied petroleum gas (LPG) requires special considerations over and above those for liquid hydrocarbons. Where required, vapour recovery systems should be provided at loading racks and marine docks.
Safe work practices and appropriate personal protective equipment may be needed when loading or unloading, cleaning up spills or leaks, or when gauging, inspecting, sampling or performing maintenance activities on loading facilities or vapour recovery systems. Delivery should be stopped or diverted in the event of an emergency such as a tank truck or tank car compartment overfill.
A number of different hazardous and toxic chemicals are used in refineries, varying from small amounts of test reagents used in laboratories to large quantities of sulphuric acid and hydrofluoric acids used in alkaline processing. These chemicals need to be received, stored and handled properly. Chemical manufacturers provide material safety information which can be used by refineries to develop safety procedures, engineering controls, personal protection requirements and emergency response procedures for handling chemicals.
The nature of the hazard at loading and unloading facilities depends upon the products being loaded and the products previously transported in the tank car, tank truck or marine vessel. Bonding equalizes the electrical charge between the loading rack and the tank truck or tank car. Grounding prevents the flow of stray currents at truck and rail loading facilities. Insulating flanges are used on marine dock piping connections to prevent static electricity build-up and discharges. Flame arrestors are installed in loading rack and marine vapour recovery lines to prevent flashback. Where switch loading is permitted, safe procedures should be established and followed.
Automatic or manual shutoff systems at supply headers should be provided at top- and bottom-loading racks and marine docks in the event of leaks or overfills. Anti-fall protection, such as hand rails, may be needed for docks and top-loading racks. Drainage and recovery systems may be provided at loading racks for storm drainage, at docks and to handle spills and leaks. Precautions are needed at LPG-loading facilities so as not to overload or overpressurize tank cars and trucks.
Refinery Support Activities and Facilities
A number of different facilities, activities and programmes, each of which has its own specific safety and health requirements, are needed to support refinery processes depending on the refinery’s location and available resources.
Administrative activities
A wide variety of administrative support activities, depending on the refining company’s philosophy and the availability of community services, are required to assure continued operation of a refinery. The function which controls oil movements into, within and out from the refinery is unique to refineries. The administrative functions can be broken down as follows. The day-to-day operation of the process units is the operations function. Another function is responsible for assuring that arrangements have been made for a continuous supply of crude oil. Other functional activities include medical services (both emergency and continuing health care), food service, engineering services, janitorial services and routine administrative and management functions common to most industries, such as accounting, purchasing, human relations and so on. The refinery training function is responsible for supervisor and employee skills and crafts training including initial, refresher and remedial training, and for employee and contractor orientation and training in emergency response and safe work practices and procedures.
Construction and maintenance
The continued safe operation of refineries depends upon the establishment and implementation of programmes and procedures for regular maintenance and preventive maintenance, and assuring replacement when necessary. Turnarounds, wherein the entire refinery or entire process units will be shut down for total equipment overall and replacement at one time, is a type of preventive maintenance programme unique to the process industry. Mechanical integrity activities, such as inspection, repair, testing and certification of valves and relief devices, which are part of the process safety management programme, are important to the continued safe operation of a refinery, as are maintenance work orders for the continued effectiveness of the refinery “management of change” programme. Work permit programmes control hot work and safe work, such as isolation and lockout, and entry into confined spaces. Maintenance and instrumentation shops have purposes which include:
- delicate and precise work to test, maintain and calibrate refinery process controls, instruments and computers
- welding
- equipment repair and overhaul
- vehicle maintenance
- carpentry and so on.
Construction and maintenance safety and health relies on some of the following programmes.
Isolation
The safe maintenance, repair and replacement of equipment within process units often requires the isolation of tanks, vessels and lines in order to preclude the possibility of flammable liquids or vapours entering an area where hot work is being performed. Isolation is normally attained by disconnecting and closing off all of the piping leading to or from a vessel; blinding or blanking the pipe at a connection near the tank or vessel; or closing a double set of block valves on the piping, if provided, and opening a bleeder valve between the two closed valves.
Lockout/tagout
Lockout and tagout programmes prevent the inadvertent activation of electrical, mechanical, hydraulic or pneumatically energized equipment during repair or maintenance. All electrically powered equipment should have its circuit breaker or main switch locked or tagged out and tested to assure non-operability, prior to starting work. Mechanical hydraulic and pneumatic equipment should be de-energized and have its power source locked or tagged out prior to starting work. Valve closing lines which are being worked on, or which are isolated, should also be locked out or tagged to prevent unauthorized opening.
Metallurgy
Metallurgy is used to assure the continued strength and integrity of lines, vessels, tanks and reactors which are subject to corrosion from the acids, corrosives, sour water, and gases and other chemicals created by and used in processing crude oil. Non-destructive testing methods are employed throughout the refinery to detect excessive corrosion and wear before failure occurs. Proper safety precautions are required to prevent excessive exposures to workers who are handling or are exposed to radioactive testing equipment, dyes and chemicals.
Warehouses
Warehouses store not only the parts, materials and equipment needed for continued refinery operations, but also store packaged chemicals and additives that are used in maintenance, processing and blending. Warehouses may also maintain supplies of required personal protective clothing and equipment including hard hats, gloves, aprons, eye and face protection, respiratory protection, safety and impervious footwear, flame-resistant clothing and acid-protective clothing. Proper storage and separation of flammable and combustible liquids and hazardous chemicals is needed to prevent spills, fires and mixing of incompatible products.
Laboratories
Laboratories are responsible for determining the values and consistency of the crude oils prior to processing, as well as performing the testing required for finished product quality control. Laboratory personnel should be trained to recognize the hazards inherent in the handling and mixing of toxic chemicals and flammable liquids, and provide protection for themselves and others.
Safety and environmental and occupational hygiene
Other important refinery support activities are safety, fire prevention and protection, environmental protection and industrial hygiene. These may be provided as separate functions or integrated into the refinery operations. Safety, emergency preparedness and response, and fire prevention and protection activities are often the responsibility of the same function within a refinery.
The safety function participates in process safety management programmes as part of the design review, pre-construction and construction review and pre-start-up review teams. Safety often assists in the contractor qualification process, reviews contractor activities and investigates incidents involving employees and contractors. Safety personnel may be responsible for overseeing permit-required activities such as confined space entry and hot work, and for checking the availability and readiness of portable fire extinguishers, decontamination facilities, safety showers, eye wash stations, fixed detection devices and alarms, and emergency self-contained breathing apparatus placed at strategic locations in event of a toxic gas release.
Safety programmes. The refinery safety function usually has responsibility for the development and administration of various safety and incident prevention programmes, including, but not limited to, the following:
- design construction and pre-start-up safety reviews
- accident, incident and near miss investigation and reporting
- emergency preparedness plans and response programmes
- contractor safety programme
- safe work practices and procedures
- lockout/tagout
- confined and inert space entry
- scaffolding
- electrical safety, equipment grounding and fault protection programme
- machine guarding
- safety signs and notices
- hot work, safe work and entry permit systems.
Fire brigades. Refinery fire brigades and emergency responders may be full-time brigade members; designated refinery employees, such as operators and maintenance personnel who are trained and assigned to respond in addition to their regular duties; or a combination of both. Besides fires, brigades traditionally respond to other refinery incidents such as acid or gas releases, rescue from vessels or tanks, spills and so on. The fire protection function may be responsible for the inspection and testing of fire detectors and signals, and fixed and portable fire protection systems and equipment, including fire trucks, fire pumps, fire water lines, hydrants, hoses and nozzles.
Refinery firefighting differs from normal firefighting because rather than extinguishment, it is often preferable to allow certain fires to continue to burn. In addition, each type of hydrocarbon liquid, gas and vapour has unique fire chemistry characteristics which must be thoroughly understood in order to best control their fires. For example, extinguishment of a hydrocarbon vapour fire without first stopping the vapour release, would only create a continued vapour gas cloud with the probability of re-ignition and explosion. Fires in tanks containing crude oil and heavy residuals need to be handled with specific firefighting techniques to avoid the possibility of an explosion or tank boil-over.
Hydrocarbon fires are often extinguished by stopping the flow of product and allowing the fire to burn out while applying cooling water to protect adjacent equipment, tanks and vessels from heat exposures. Many fixed fire protection systems are designed with this specific purpose. Fighting fires in process units under pressure requires special consideration and training, particularly when catalysts such as hydrofluoric acid are involved. Special firefighting chemicals, such as dry powder and foam-water solutions, may be used to extinguish hydrocarbon fires and control vapour emissions.
Emergency preparedness. Refineries need to develop and implement emergency response plans for a number of different potential situations, including explosions, fires, releases and rescues. The emergency plans should include the use of outside assistance, including contractors, governmental and mutual aid as well as availability of special supplies and equipment, such as firefighting foam and spill containment and adsorption materials.
Gas and vapour testing
Gas, particulate and vapour monitoring, sampling and testing in refineries is conducted to assure that work can be performed safely and processes can be operated without toxic or hazardous exposures, explosions or fires. Atmospheric testing is conducted using a variety of instruments and techniques to measure oxygen content, hydrocarbon vapours and gases, and to determine hazardous and toxic exposure levels. Instruments must be properly calibrated and adjusted prior to use, by qualified persons, to assure dependable and accurate measurements. Depending on the work location, potential hazards and type of work being performed, testing, sampling and monitoring may be conducted prior to the start of work, or at specified intervals during work, or continuously throughout the course of work.
When establishing refinery procedures for sampling and testing flammable, inert and toxic atmospheres, the use of personal protective equipment, including appropriate respiratory protection, should be considered. It should be noted that canister-type respirators are unsuitable for oxygen-deficient atmospheres. Testing requirements should depend upon the degree of hazard which would be present in the event of instrument failure.
Testing of the following substances may be performed using portable equipment or fixed instrumentation:
Oxygen. Combustible gas meters work by burning a minute sample of the atmosphere being tested. In order to obtain an accurate combustible gas reading, a minimum of 10% and a maximum of 25% oxygen must be present in the atmosphere. The amount of oxygen present in the atmosphere is determined by using an oxygen meter prior to, or simultaneously with, using the combustible gas meter. Testing for oxygen is essential when working in confined or enclosed spaces, as entry without respiratory protection (provided that there are no toxic exposures) requires normal breathing-air oxygen concentrations of approximately 21%. Oxygen meters are also used to measure the amount of oxygen present in inerted spaces, to assure that there is not enough present to support combustion during hot work or other operations.
Hydrocarbon vapours and gases. “Hot work” is work which creates a source of ignition, such as welding, cutting, grinding, blast cleaning, operating an internal combustion engine and so on, in an area where the potential for exposure to flammable vapours and gases exists. In order to conduct hot work safely, instruments known as combustible gas meters are used to test the atmosphere for hydrocarbon vapours. Hydrocarbon vapours or gases will burn only when mixed with air (oxygen) in certain proportions and ignited. If there is not enough vapour in the air, the mixture is said to be “too lean to burn”, and if there is too much vapour (too little oxygen), the mixture is “too rich to burn”. The limiting proportions are called the “upper and lower flammable limits” and are expressed as a percentage of volume of vapour in air. Each hydrocarbon molecule or mixture has different flammability limits, typically ranging from about 1 to 10% vapour in air. Gasoline vapour, for example, has a lower flammable limit of 1.4% and an upper flammable limit of 7.6 per cent.
Toxic atmospheres. Special instruments are used to measure the levels of toxic and hazardous gases, vapours and particulates which may be present in the atmosphere where people are working. These measurements are used to determine the level and type of protection needed, which may vary from complete ventilation and replacement of the atmosphere to the use of respiratory and personal protective equipment by people working in the area. Examples of hazardous and toxic exposures which may be found in refineries include asbestos, benzene, hydrogen sulphide, chlorine, carbon dioxide, sulphuric and hydrofluoric acids, amines, phenol and others.
Health and safety programmes
The basis for refinery industrial hygiene is an administrative and engineering controls programme covering facility exposures to toxic and hazardous chemicals, laboratory safety and hygiene, ergonomics and medical surveillance.
Regulatory agencies and companies establish exposure limitations for various toxic and hazardous chemicals. The occupational hygiene function conducts monitoring and sampling to measure employee exposure to hazardous and toxic chemicals and substances. Industrial hygienists may develop or recommend engineering controls, preventive work practices, product substitution, personal protective clothing and equipment or alternate measures of protection or reducing exposure.
Medical programmes. Refineries typically require preplacement and periodic medical examinations to determine the employee’s ability to initially and subsequently perform the work, and assure that the continued work requirements and exposures will not endanger the employee’s health or safety.
Personal protection. Personal protection programmes should cover typical refinery exposures, such as noise, asbestos, insulation, hazardous waste, hydrogen sulphide, benzene and process chemicals including caustics, hydrogen fluoride, sulphuric acid and so on. Industrial hygiene may designate the appropriate personal protective equipment to be used for various exposures, including negative pressure and air-supplied respirators and hearing, eye and skin protection.
Product safety. Product safety awareness covers knowing about the hazards of chemicals and materials to which the potential for exposure exists in the workplace, and what actions to take in the event exposure by ingestion, inhalation or skin contact occurs. Toxicological studies of crude oil, refinery streams, process chemicals, finished products and proposed new products are conducted to determine the potential effects of exposure on both employees and consumers. The data are used to develop health information concerning permissible limits of exposure or acceptable amounts of hazardous materials in products. This information is typically distributed by material safety data sheets (MSDSs) or similar documents, and employees are trained or educated in the hazards of the materials in the workplace.
Environmental Protection
Environmental protection is an important consideration in refinery operations because of regulatory compliance requirements and a need for conservation as oil prices and costs escalate. Oil refineries produce a wide range of air and water emissions that can be hazardous to the environment. Some of these are contaminants in the original crude oil, while others are a result of refinery processes and operations. Air emissions include hydrogen sulphide, sulphur dioxide, nitrogen oxides and carbon monoxide (see table 2). Waste water typically contains hydrocarbons, dissolved materials, suspended solids, phenols, ammonia, sulphides, acids, alkalis and other contaminants. There is also the risk of accidental spills and leaks of a wide range of flammable and/or toxic chemicals.
Controls established to contain liquid and vapour releases and reduce operating costs include the following:
- Energy conservation. Controls include steam leak control and condensate recovery programmes to conserve energy and increase efficiency.
- Water pollution. Controls include waste water treatment in API separators and subsequent treatment facilities, storm water collection, retainment and treatment and spill prevention containment and control programmes.
- Air pollution. Since refineries operate continuously, leak detection, particularly at valves and pipe connections, is important. Controls include reducing hydrocarbon vapour emissions and releases to the atmosphere, refinery valve and fitting tightness programmes, floating roof tank seals and vapour containment programmes, and vapour recovery for loading and unloading facilities and for venting tanks and vessels.
- Ground pollution. Preventing oil spillage from polluting soil and contaminating ground water is accomplished by the use of dikes and the providing of drainage to specified, protected containment areas. Contamination from spillage inside dike areas may be prevented by the use of secondary containment measures, such as impervious plastic or clay dike liners.
- Spill response. Refineries should develop and implement programmes to respond to spills of crude oil, chemicals and finished products, on both land and water. These programmes may rely on trained employees or outside agencies and contractors to respond to the emergency. The type, amount needed and availability of spill clean-up and restoration supplies and equipment, either on site or on call, should be included in the preparedness plan.
Developing a Process Safety Management Programme
Whenever there are processes that use temperature and pressure to change the molecular structure or create new products from chemicals, the possibility exists for fires, explosions or releases of flammable or toxic liquids, vapours, gases or process chemicals. The control of these undesired events requires a special science called process safety management. The terms process safety and process safety management are most commonly used to describe the protection of employees, the public and the environment from the consequences of undesirable major incidents involving flammable liquids and highly hazardous materials. According to the United States Chemical Manufacturers’ Association (CMA), “process safety is the control of hazards which are caused by maloperation or malfunction of the processes used to convert raw materials into finished products, which may lead to the unplanned release of hazardous material” (CMA 1985).
Industry and labour process safety involvement
Process safety technology has played an important role in the chemical processing industries so that handling flammable and combustible liquids and gases could proceed without undesirable consequences. During the 1980s, the oil and gas industries, for example, recognized that process safety technology alone, without process safety management, would not prevent catastrophic incidents. With this in mind, a number of industry associations, such as, in the United States, the Center for Chemical Process Safety (CCPS), the American Petroleum Institute (API) and the Chemical Manufacturers' Association (CMA), initiated programmes to develop and provide process safety management guidelines for use by their members. As stated by the CCPS, "The evolution of process safety from a purely technical issue to one that demanded management approaches was essential to continued process safety improvement".
The CCPS was formed in 1985 to promote the improvement of process safety management techniques among those who store, handle, process and use hazardous chemicals and materials. In 1988, the Chemical Manufacturer's Association (CMA) initiated its Responsible Care® programme outlining each member company's commitment to environmental, health and safety responsibility in managing chemicals.
In 1990, the API initiated an industry-wide programme entitled, STEP-Strategies for Today's Environmental Partnership, with the intention of improving the oil and gas industry's environmental, health and safety performance. One of the seven strategic elements of the STEP programme covers petroleum operating and process safety. The following documents are examples of some of the materials developed as a result of the STEP programme which provide guidance to the oil and gas industry to help prevent the occurrence or minimize the consequences of catastrophic releases of flammable liquids and vapours or hazardous process materials:
- Management of Process Hazards (RP 750)
RP 750 covers the management of hydrocarbon process hazards in design, construction, start-up, operations, inspection, maintenance and facility modifications. It applies specifically to refineries, petro-chemical plants and major processing facilities that use, produce, process or store flammable liquids and toxic processing chemicals in quantities above certain hazardous amounts (as defined therein).
- Management of Hazards Associated with Location of Process Plant Buildings (RP 752)
RP 752, co-developed by API and CMA, is intended to help identify process plant buildings of concern, understand the potential hazards related to their location within the process facility and manage the risk of fire, explosion and toxic releases.
- Management Practices, Self-assessment Process, and Resource Materials (RP 9000)
RP 9000 provides resource materials and self assessment methodology to measure progress in implementing process safety management elements.
Examples of other organizations which have developed materials and programmes providing guidance covering chemical process safety management include, but are not limited to, the following:
- Organizations Resource Counselors' (ORC) report, Process Hazards Management of Substances with Catastrophic Potential
- National Petroleum Refiners Association (NPRA), BEST (Building Environmental Stewardship Tools) programme
- International Labour Organization (ILO), Code of Practice on the Prevention of Major Accident Hazards
- International Chamber of Commerce (ICC), Charter for Sustainable Development.cmp01ce.doc
The process design and technology, changes in the process, materials and changes in materials, operations and maintenance practices and procedures, training, emergency preparedness and other elements affecting the process must all be considered in the systematic identification and evaluation of hazards so as to determine whether or not they have the potential to lead to a catastrophe in the workplace and surrounding community.
Beginning in the early 1980s, a number of serious major incidents occurred in the petroleum and chemical industries involving highly hazardous materials, which resulted in considerable numbers of fatalities and injuries and significant property losses. These incidents provided the impetus for government agencies, labour organizations and industry associations throughout the world to develop and implement codes, regulations, procedures and safe work practices directed toward the elimination or mitigation of these undesirable events, through the application of the principles of process safety management. They are discussed more fully in the Disasters, natural and technological chapter and elsewhere in this Encyclopaedia.
In response to public concern over the potential hazards of chemicals, governments and regulatory agencies throughout the world initiated programmes which required manufacturers and users to identify hazardous materials in the workplace and inform employees and consumers of the hazards presented by their manufacture, use, storage and handling. These programmes, which covered emergency preparedness and response, hazard recognition, product knowledge, control of hazardous chemicals and reporting of toxic releases, included hydrocarbon processing.
Process Safety Management Requirements
Process safety management is an integral part of the overall chemical processing facility safety programme. An effective process safety management programme requires the leadership, support and involvement of top management, facility management, supervisors, employees, contractors and contractor employees.
Components to be considered when developing a process safety management programme include:
- Interdependent continuity of operations, systems and organization
- Management of information. The process safety management programme relies upon providing availability and access to good records and documentation.
- Control of process quality, deviations and exceptions and alternate methods
- Management and supervisory accessibility and communications. Because process safety management is the basis for all safety efforts within the facility, managerial, supervisory and employee responsibility and accountability should be clearly delineated, communicated and understood in order for the programme to work.
- Goals and objectives, compliance audits and performance measurement. Prior to implementation, it is important to establish both long-term and short-term goals and objectives for each of the elements of the process safety management programme.
Elements of the Process Safety Management Programme
All chemical facility process safety management programmes cover the same basic requirements, although the number of programme elements may vary depending on the criteria used. Regardless which government, company or association source document is used as a guide, there are a number of basic requirements which should be included in every chemical process safety management programme:
- process safety information
- employee involvement
- process hazard analysis
- management of change
- operating procedures
- safe work practices and permits
- employee information and training
- contractor personnel
- pre-startup safety reviews
- design quality assurance
- maintenance and mechanical integrity
- emergency response
- periodic safety audits
- process incident investigation
- standards and regulations
- trade secrets.
Process safety information
Process safety information is used by the process industry to define critical processes, materials and equipment. Process safety information includes all available written information concerning process technology, process equipment, raw materials and products and chemical hazards before conducting a process hazard analysis. Other critical process safety information is documentation relating to capital project reviews and design basis criteria.
Chemical information includes not only the chemical and physical properties, reactivity and corrosive data and thermal and chemical stability of chemicals such as hydrocarbons and highly hazardous materials in the process, but also the hazardous effects of inadvertently mixing different incompatible materials. Chemical information also includes that which may be needed to conduct environmental hazard assessments of toxic and flammable releases and permissible exposure limits.
Process technology information includes block flow diagrams and/ or simple process flow diagrams as well as descriptions of the chemistry of each specific process with the safe upper and lower limits for temperatures, pressures, flows, compositions and, where available, process design material and energy balances. The consequences of deviations in the process and materials, including their effect on employee safety and health, are also determined. Whenever processes or materials are changed, the information is updated and re-evaluated in accordance with the facility’s management of change system.
Process equipment and mechanical design information includes documentation covering the design codes employed and whether or not equipment complies with recognized engineering practices. A determination is made as to whether existing equipment which was designed and constructed in accordance with codes, standards and practices no longer in general use is maintained, operated, inspected and tested to assure continued safe operation. Information on materials of construction, piping and instrument diagrams, relief system design, electrical classification, ventilation design and safety systems is updated and re-evaluated when changes occur.
Employee involvement
Process safety management programmes should include employee participation in the development and conduct of process safety analyses and other elements of the programme. Access to process safety information, incident investigation reports and process hazard analyses is usually provided to all employees and contractor employees working in the area. Most industrialized nations require that workers be systematically instructed in the identification, nature and safe-handling of all chemicals to which they may be exposed.
Process hazard analysis
After the process safety information is compiled, a thorough and systematic multi-disciplinary process hazard analysis, appropriate to the complexity of the process, is conducted in order to identify, evaluate and control the hazards of the process. Persons performing the process hazard analysis should be knowledgeable and experienced in relevant chemistry, engineering and process operations. Each analysis team normally includes at least one person who is thoroughly familiar with the process being analysed and one person who is competent in the hazard analysis methodology being used.
The priority order used to determine where within the facility to begin conducting process hazard analyses is based on the following criteria:
- extent and nature of the process hazards
- number of potentially affected workers
- operating and incident history of the process
- age of the process.
A number of methods for conducting process safety analyses are used in the chemical industry.
The “what if?” method asks a series of questions to review potential hazard scenarios and possible consequences and is most often used when examining proposed modifications or changes to the process, materials, equipment or facility.
The “checklist” method is similar to the “what if?” method, except that a previously developed checklist is used which is specific to the operation, materials, process and equipment. This method is useful when conducting pre-startup reviews upon completion of initial construction or following major turnarounds or additions to the process unit. A combination of the “what if?” and “checklist” methods is often used when analysing units that are identical in construction, materials, equipment and process.
The hazard and operability (HAZOP) study method is commonly used in the chemical and petroleum industries. It involves a multi-disciplinary team, guided by an experienced leader. The team uses specific guide words, such as “no”, “increase”, “decrease” and “reverse”, which are systematically applied to identify the consequences of deviations from design intent for the processes, equipment and operations being analysed.
Fault tree/event tree analyses are similar, formal deductive techniques used to estimate the quantitative likelihood of an event occurring. Fault tree analysis works backward from a defined incident to identify and display the combination of operational errors and/ or equipment failures which were involved in the incident. Event tree analysis, which is the reverse of fault tree analysis, works forwards from specific events, or sequences of events, in order to pinpoint those that could result in hazards, and thereby calculate the likelihood of an event’s sequence occurring.
The failure mode and effects analysis method tabulates each process system or unit of equipment with its failure modes, the effect of each potential failure on the system or unit and how critical each failure could be to the integrity of the system. The failure modes are then ranked in importance to determine which is most likely to cause a serious incident.
No matter which method is used, all chemical process hazard analyses consider the following:
- process location, siting and hazards of the process
- identification of any prior incident or near miss with potential catastrophic consequences
- engineering and administrative controls applicable to the hazards
- interrelationships of controls and appropriate application of detection methodology to provide early warnings
- consequences of human factors, facility siting and failure of the controls
- consequences of safety and health effects on workers within areas of potential failure.
Management of change
Chemical process facilities should develop and implement programmes which provide for the revision of process safety information, procedures and practices as changes occur. Such programmes include a system of management authorization and written documentation for changes to materials, chemicals, technology, equipment, procedures, personnel and facilities that affect each process.
Management of change programmes in the chemical industry, for example, include the following areas:
- change of hydrocarbon process technology
- changes in facility, equipment or materials (e.g., catalysts or additives)
- management of change personnel and organizational and personnel changes
- temporary changes, variances and permanent changes
- enhancement of process safety knowledge, including:
- technical basis for proposed change
- impact of change on safety, health and environment
- modifications to operating procedures and safe work practices
- modifications required to other processes
- time required for the change
- authorization requirements for the proposed change
- updating documentation relating to process information, operating procedures and safety practices
- required training or education due to change
- management of subtle change (anything which is not replacement in kind)
- non-routine changes.
The management of change system includes informing employees involved in the process and maintenance and contractor personnel whose tasks would be affected by any changes of the changes and providing updated operating procedures, process safety information, safe work practices and training as needed, prior to the startup of the process or affected part of the process.
Operating procedures
Chemical processing facilities must develop and provide operating instructions and detailed procedures to workers. Operating instructions should be regularly reviewed for completeness and accuracy (and updated or amended as changes occur) and cover the process unit’s operating limits, including the following three areas:
- consequences of deviation
- steps to avoid or correct deviation
- functions of safety systems related to operating limits.
Workers involved in the process have access to operating instructions covering the following areas:
- initial startup (startup after turnarounds, emergencies and temporary operations)
- normal startup (normal and temporary operations and normal shutdown)
- emergency operations and emergency shutdown
- conditions under which emergency shutdown is required and assignment of shutdown responsibilities to qualified operators
- non-routine work
- operator-process and operator-equipment interface
- administrative controls vs. automated controls.
Safe work practices
Chemical process facilities should implement hot-work and safe work permit and work order programmes to control work conducted in or near process areas. Supervisors, employees and contractor personnel must be familiar with the requirements of the various permit programmes, including permit issuance and expiration and appropriate safety, materials handling and fire protection and prevention measures.
The types of work included in typical chemical facility permit programmes include the following:
- hot work (welding, hot tapping, internal combustion engines, etc.)
- lockout/tagout of electrical, mechanical, pneumatic energy and pressure
- confined-space entry and use of inert gas
- venting, opening and cleaning process vessels, tanks, equipment and lines
- control of entry into process areas by non-assigned personnel.
Chemical facilities should develop and implement safe work practices to control potential hazards during process operations, covering the following areas of concern:
- properties and hazards of materials, catalysts and chemicals used in the process
- engineering, administrative and personal protection controls to prevent exposures
- measures to be taken in event of physical contact or exposure with hazardous chemical
- quality control of raw materials, catalysts and inventory control of hazardous chemicals
- safety and protection system (interlock, suppression, detection, etc.) functions
- special or unique hazards in the workplace.
Employee information and training
Chemical process facilities should use formal process safety training programmes to train and educate incumbent, reassigned and new supervisors and workers. The training provided for chemical process operating and maintenance supervisors and workers should cover the following areas:
- required skills, knowledge and qualifications of process employees
- selection and development of process related training programmes
- measuring and documenting employee performance and effectiveness
- design of process operating and maintenance procedures
- overview of process operations and process hazards
- availability and suitability of materials and spare parts for the processes in which they are to be used
- process start-up, operating, shut-down and emergency procedures
- safety and health hazards related to the process, catalysts and materials
- facility and process area safe work practices and procedures.
Contractor personnel
Contractors are often employed in chemical processing facilities. The facilities must institute procedures to assure that contractor personnel performing maintenance, repair, turnaround, major renovation or specialty work are fully aware of the hazards, materials, processes, operating and safety procedures and equipment in the area. Periodic evaluations of performance are made to assure that contractor personnel are trained, qualified, follow all safety rules and procedures and are informed and aware of the following:
- potential fire, explosion and toxic release hazards related to their work
- plant safety procedures and contractor safe work practices
- emergency plan and contractor personnel actions
- controls for contractor personnel entry, exit and presence in process areas.
Pre-startup safety reviews
Pre-startup process safety reviews are conducted in chemical plants prior to startup of new process facilities and introduction of new hazardous materials or chemicals into facilities, following a major turnaround and where facilities have had significant process modifications.
The pre-startup safety reviews assure the following have been accomplished:
- construction, materials and equipment are verified as in accordance with design criteria
- process systems and hardware, including computer control logic, have been inspected, tested and certified
- alarms and instruments are inspected, tested and certified
- relief and safety devices and signal systems are inspected, tested and certified
- fire protection and prevention systems are inspected, tested and certified
- safety, fire prevention and emergency response procedures are developed, reviewed, in place and are appropriate and adequate
- startup procedures are in place and proper actions have been taken
- a process hazard analysis has been performed and all recommendations addressed, implemented or resolved and actions documented
- all required initial and/ or refresher operator and maintenance personnel training, including emergency response, process hazards and health hazards, is completed
- all operating procedures (normal and upset), operating manuals, equipment procedures and maintenance procedures are completed and in place
- management of change requirements for new processes and modifications to existing processes have been met.
Design Quality Assurances
When new processes or major changes to existing processes are undertaken, a series of process safety design reviews are normally conducted before and during construction (prior to the pre-startup review). The design control review, conducted just before plans and specifications are issued as “final design drawings”, covers the following areas:
- plot plan, siting, spacing, electrical classification and drainage
- hazards analysis and process chemistry design
- project management requirements and qualifications
- process equipment and mechanical equipment design and integrity
- piping and instrument drawings
- reliability engineering, alarms, interlocks, reliefs and safety devices
- materials of construction and compatibility.
Another review is normally conducted just prior to the start of construction covering the following:
- demolition and excavation procedures
- control of raw materials
- control of construction personnel and equipment on facility and site
- fabrication, construction and installation procedures and inspection.
One or more reviews are usually conducted during the course of construction or modification to assure the following areas are in accordance with design specifications and facility requirements:
- materials of construction provided and used as specified
- proper assembly and welding techniques, inspections, verifications and certifications
- chemical and occupational health hazards considered during construction
- physical, mechanical and operational safety hazards considered during construction and facility permit and safety practices followed
- interim protective and emergency response systems provided and working.
Maintenance and mechanical integrity
Process facilities have programmes to maintain ongoing integrity of process-related equipment, including periodic inspection, testing, performance maintenance, corrective action and quality assurance. The programmes are intended to assure that mechanical integrity of equipment and materials is reviewed and certified and deficiencies corrected prior to startup, or provisions made for appropriate safety measures.
Mechanical integrity programmes cover the following equipment and systems:
- pressure vessels and storage tanks
- emergency shutdown and fire protection systems
- process safeguards such as relief and vent systems and devices, controls, interlocks, sensors and alarms
- pumps and piping systems (including components such as valves)
- quality assurance, materials of construction and reliability engineering
- maintenance and preventive maintenance programmes.
Mechanical integrity programmes also cover inspection and testing of maintenance materials, spare parts and equipment to assure proper installation and adequacy for the process application involved. The acceptance criteria and frequency of inspections and tests should conform with manufacturers’ recommendations, good engineering practices, regulatory requirements, industry practices, facility policies or prior experience.
Emergency Response
Emergency preparedness and response programmes are developed to cover an entire process facility and to provide for hazard identification and assessment of potential process hazards. These programmes include training and educating employees and contractor employees in emergency notification, response and evacuation procedures.
A typical process facility emergency preparedness programme complies with applicable company and regulatory requirements and includes the following:
- distinctive employee and/ or community alarm or notification system
- preferred method of internal reporting of fires, spills, releases and emergencies
- requirements for reporting process-related incidents to appropriate government agencies
- emergency shutdown, evacuation, procedures to account for personnel, emergency escape procedures, vehicle and equipment removal and route assignments
- emergency response and rescue procedures, duties and capabilities including employees, public safety, contractors and mutual aid organizations
- procedures for handling small spills or releases of hazardous chemicals
- procedures for providing and safeguarding emergency power and utilities
- business continuation plans, personnel and equipment sources
- document and record preservation, site security, cleanup, salvage and restoration.
Periodic safety audits
Many process facilities use self-evaluation process safety management audits to measure facility performance and assure compliance with internal and external (regulatory, company and industry) process safety requirements. The two basic principles of conducting self evaluation audits are: gathering all of the relevant documentation covering process safety management requirements at a specific facility and determining the programme’s implementation and effectiveness by following up on their application in one or more selected processes. A report of the audit findings and recommendations is developed and facility management maintains documentation which notes how deficiencies had been corrected or mitigated, and if not, reasons why no corrective action had been taken.
Compliance audit programmes in hydrocarbon process facilities cover the following areas:
- establishment of goals, schedule and methods of verification of findings prior to the audit
- determination of the methodology (or format) to be used in conducting the audit, and develop appropriate checklists or audit report forms
- readiness to certify compliance with government, industry and company requirements
- assignment of knowledgeable audit teams (internal and/ or external expertise)
- prompt responses to all findings and recommendations and documentation of actions taken
- maintenance of a copy of at least the most recent compliance audit report on file.
Facility and process unit specific checklists are often developed for use when conducting process safety audits which cover the following items:
- orientation and process safety management programme overview
- preliminary walk-around through the refinery or gas processing facility
- process facility documentation review
- “prior incidents” and near misses (in the process facility or specific unit)
- determination and review of selected process units to be audited
- process unit construction (initial and subsequent modifications)
- process unit chemistry hazards (feedstocks, catalysts, process chemicals, etc.)
- process unit operations
- process unit controls, reliefs and safety systems
- process unit maintenance, repair, testing and inspection
- process unit-related training and employee involvement
- process facility management of change programme, implementation and effectiveness
- process fire protection and emergency notification and response procedures.
Because the objectives and scope of audits can vary, the compliance audit team should include at least one person knowledgeable in the process being audited, one person with applicable regulatory and standards expertise and other persons with the skills and qualifications necessary for conducting the audit. Management may decide to include one or more outside experts on the audit team due to lack of facility personnel or expertise, or because of regulatory requirements.
Process incident investigation
Process facilities have established programmes to thoroughly investigate and analyse process-related incidents and near misses, promptly address and resolve findings and recommendations and review the results with workers and contractors whose jobs are relevant to the incident findings. Incidents (or near misses) are thoroughly investigated as soon as possible by a team which includes at least one person knowledgeable in the process operation involved and others with appropriate knowledge and experience.
Standards and Regulations
Process facilities are subject to two distinct and separate forms of standards and regulations.
- External codes, standards and regulations applicable to the design, operation and protection of process facilities and employees typically include government regulations and association and industry standards and practices.
- Internal policies, guidelines and procedures, developed or adopted by the company or facility to complement external requirements and to cover processes which are distinct or unique, are reviewed periodically and changed when necessary, in accordance with the facility’s management of change system.
Trade Secrets
Process facility management should provide process information, without regard to possible trade secrets or confidentiality agreements, to persons who are:
- responsible for gathering and compiling process safety information
- conducting process hazard analyses and compliance audits
- developing maintenance, operating and safe work procedures
- involved in incident (near miss) investigations
- responsible for emergency planning and response.
Facilities typically require that persons to whom process information is made available enter into agreements not to disclose the information.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."