The intervertebral discs occupy about one-third of the spine. Since they not only provide the spinal column with flexibility but also transmit load, their mechanical behaviour has a great influence on the mechanics of the whole spine. A high proportion of cases of low-back pain are associated with the disc, either directly through disc herniation, or indirectly, because degenerated discs place other spinal structures under abnormal stress. In this article, we review the structure and composition of the disc in relation to its mechanical function and discuss changes to the disc in disease.
Anatomy
There are 24 intervertebral discs in the human spine, interspersed between the vertebral bodies. Together these make up the anterior (front) component of the spinal column, with the articulating facet joints and the transverse and spinous processes making up the posterior (rear) elements. The discs increase in size down the spine, to approximately 45 mm antero-posteriorly, 64 mm laterally and 11 mm height in the lower back region.
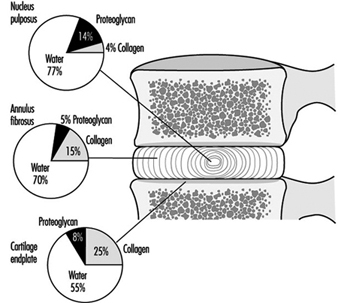
The disc is made of cartilage-like tissue and consists of three distinct regions, (see figure 1). The inner region (nucleus pulposus) is a gelatinous mass, particularly in the young person. The outer region of the disc (annulus fibrosus) is firm and banded. The fibres of the annulus are criss-crossed in an arrangement which allows it to withstand high bending and twisting loads. With increasing age the nucleus loses water, becomes firmer and the distinction between the two regions is less clear than early in life. The disc is separated from the bone by a thin layer of hyaline cartilage, the third region. In adulthood the cartilage endplate and the disc itself normally have no blood vessels of their own but rely on the blood supply of adjacent tissues, such as ligaments and vertebral body, to transport nutrients and remove waste products. Only the outer portion of the disc is innervated.
Figure 1. The relative proportions of the three main components of the normal adult human intervertebral disc and cartilage endplate.
Composition
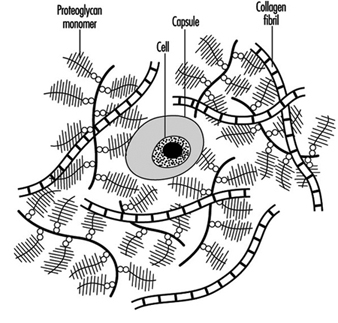
The disc, like other cartilage, consists mainly of a matrix of collagen fibres (which are embedded in a gel of proteoglycan) and of water. These together make up 90 to 95% of the total tissue mass, although the proportions vary with location within the disc and with age and degeneration. There are cells interspersed throughout the matrix that are responsible for synthesizing and maintaining the different components within it (figure 2). A review of the biochemistry of the disc can be found in Urban and Roberts 1994.
Figure 2. Schematic representation of disc structure, showing banded collagen fibres interspersed with numerous bottle-brush-like proteoglycan molecules and few cells.
Proteoglycans: The major proteoglycan of the disc, aggrecan, is a large molecule consisting of a central protein core to which many glycosaminoglycans (repeating chains of disaccharides) are attached (see figure 3). These side chains have a high density of negative charges associated with them, thus making them attractive to water molecules (hydrophilic), a property described as swelling pressure. It is very important to the functioning of the disc.
Figure 3. Diagram of part of a disc proteoglycan aggregate. G1, G2 and G3 are globular, folded regions of the central core protein.
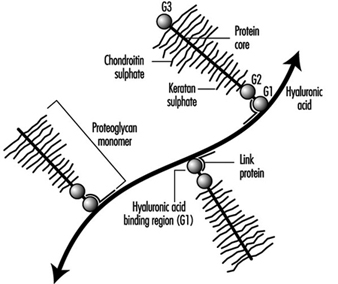 Huge aggregates of proteoglycans can form when individual molecules link onto a chain of another chemical, hyaluronic acid. The size of aggrecans varies (ranging in molecular weight from 300,000 to 7 million dalton) depending on how many molecules make up the aggregate. Other smaller types of proteoglycans have recently also been found in the disc and cartilage endplate—for example, decorin, biglycan, fibromodulin and lumican. Their function is generally unknown but fibromodulin and decorin may be involved in regulating collagen network formation.
Huge aggregates of proteoglycans can form when individual molecules link onto a chain of another chemical, hyaluronic acid. The size of aggrecans varies (ranging in molecular weight from 300,000 to 7 million dalton) depending on how many molecules make up the aggregate. Other smaller types of proteoglycans have recently also been found in the disc and cartilage endplate—for example, decorin, biglycan, fibromodulin and lumican. Their function is generally unknown but fibromodulin and decorin may be involved in regulating collagen network formation.
Water: Water is the main constituent in the disc, making up 65 to 90% of the tissue volume, depending on age and region of the disc. There is a correlation between the quantity of proteoglycan and the water content of the matrix. The amount of water also varies depending on the load applied to the disc, hence water content differs night and day since load will be very different when sleeping. Water is important both to the mechanical functioning of the disc and for providing the medium for transport of dissolved substances within the matrix.
Collagen: Collagen is the main structural protein in the body, and consists of a family of at least 17 distinct proteins. All collagens have helical regions and are stabilized by a series of intra- and inter- molecular crosslinks, which make the molecules very strong in resisting mechanical stresses and enzymatic degradation. The length and shape of different types of collagen molecules and the proportion which is helical, vary. The disc is composed of several types of collagens, with the outer annulus being predominantly type I collagen, and the nucleus and cartilage endplate predominantly type II. Both types form fibrils which provide the structural framework of the disc. The fibrils of the nucleus are much finer (>> mm in diameter) than those of the annulus (0.1 to 0.2 mm in diameter). The disc cells are often surrounded by a capsule of some of the other types of collagen, such as type VI.
Cells: The intervertebral disc has a very low density of cells in comparison to other tissues. Although the density of cells is low, their continuing activity is vital for the health of the disc, as the cells produce macromolecules throughout life, to replace those which break down and are lost with the passage of time.
Function
The main function of the disc is mechanical. The disc transmits load along the spinal column and also allows the spine to bend and twist. The loads on the disc arise from body weight and muscular activity, and change with posture (see figure 4). During daily activities the disc is subject to complex loads. Extending or flexing the spine produces mainly tensile and compressive stresses on the disc, which increase in magnitude going down the spine, due to differences in body weight and geometry. Rotating the spine produces crosswise (shear) stresses.
Figure 4. Relative intradiscal pressures in different postures compared to the pressure in upright standing (100%).
Discs are under pressure, which varies with posture from around 0.1 to 0.2 MPa at rest, to around 1.5 to 2.5 MPa while bending and lifting. The pressure is mainly due to water pressure across the nucleus and inner annulus in a normal disc. When load on the disc is increased, pressure is distributed evenly across the endplate and throughout the disc.
During loading the disc deforms and loses height. The endplate and annulus bulge, increasing the tension on these structures, and the pressure of the nucleus consequently rises. The degree of deformation of the disc depends on the rate at which it is loaded. The disc can deform considerably, compressing or extending by 30 to 60% during flexion and extension. Distances between adjacent spinal processes can increase by over 300%. If a load is re- moved within a few seconds, the disc quickly returns to its former state, but if the load is maintained, the disc continues to lose height. This “creep” results from the continuing deformation of the disc structures and also from fluid loss, because discs lose fluid as a result of the increased pressure. Between 10 and 25% of the disc’s fluid is slowly lost during daily activities, when the disc is under much greater pressures, and regained when lying down at rest. This loss of water can lead to a decrease in an individual’s height of 1 to 2 cm from morning to evening among dayworkers.
As the disc changes its composition because of ageing or degeneration, the response of the disc to mechanical loads also changes. With a loss of proteoglycan and thus water content, the nucleus can no longer respond as efficiently. This change results in uneven stresses across the endplate and the annulus fibres, and, in severe cases of degeneration, the inner fibres may bulge inward when the disc is loaded, which, in turn, may lead to abnormal stresses on other disc structures, eventually causing their failure. The rate of creep is also increased in degenerated discs, which thus lose height faster than normal discs under the same load. Narrowing of the disc space affects other spinal structures, such as muscles and ligaments, and, in particular, leads to an increase in pressure on the facet joints, which may be the cause of the degenerative changes seen in the facet joints of spines with abnormal discs.
Contribution of Major Components to Function
Proteoglycans
Disc function depends on maintaining equilibrium in which the water pressure of the disc is balanced by the disc swelling pressure. The swelling pressure depends on the concentration of ions attracted into the disc by the negatively charged proteoglycans, and thus depends directly on the concentration of proteoglycans. If the load on the disc is increased, water pressure rises and disturbs the equilibrium. To compensate, fluid seeps out of the disc, increasing proteoglycan concentration and disc osmotic pressure. Such fluid expression continues either until the balance is restored or the load on the disc is removed.
Proteoglycans affect fluid movement in other ways, as well. Because of their high concentration in the tissue, the spaces between chains are very small (0.003 to 0.004 mm). Fluid flow through such small pores is very slow, and thus even though there is a large pressure differential, the rate at which fluid is lost, and hence the rate of disc creep, is slow. However, since discs which have degenerated have lower proteoglycan concentrations, fluid can flow through the matrix faster. This may be why degenerated discs lose height more quickly than normal discs. The charge and high concentration of proteoglycans control the entry and movement of other dissolved substances into the disc. Small molecules (nutrients like glucose, oxygen) can easily enter the disc and move through the matrix. Electropositive chemicals and ions, such as Na+or Ca2+, have higher concentrations in the negatively charged disc than in the surrounding interstitial fluid. Large molecules, such as serum albumin or immunoglobulins, are too bulky to enter the disc, and are present only in very low concentrations. Proteoglycans may also affect cellular activity and metabolism. Small proteoglycans, such as biglycan, may bind growth factors and other mediators of cellular activity, releasing them when the matrix is degraded.
Water
Water is the major component of the disc and rigidity of the tissue is maintained by the hydrophilic properties of the proteoglycans. With initial loss of water, the disc becomes more flaccid and deformable as the collagen network relaxes. However, once the disc has lost a significant fraction of water, its mechanical properties change drastically; the tissue behaves more like a solid than a composite under load. Water also provides the medium through which nutrients and wastes are exchanged between the disc and the surrounding blood supply.
Collagen
The collagen network, which can support high tensile loads, provides a framework for the disc, and anchors it to the neighbouring vertebral bodies. The network is inflated by the water taken in by the proteoglycans; in turn the network restrains the proteoglycans and prevents them from escaping from the tissue. These three components together thus form a structure which is able to support high compressive loads.
The organization of the collagen fibrils provides the disc with its flexibility. The fibrils are arranged in layers, with the angle at which the fibrils of each layer run between the neighbouring vertebral bodies, alternating in direction. This highly specialized weave allows the disc to wedge extensively, thus allowing bending of the spine, even though collagen fibrils themselves can extend by only about 3%.
Metabolism
The cells of the disc produce both large molecules and enzymes which can break down matrix components. In a healthy disc, the rates of matrix production and breakdown are balanced. If the balance is upset, the composition of the disc must ultimately change. In growth, synthesis rates for new and replacement mol-ecules are higher than degradation rates, and matrix materials accumulate around the cells. With ageing and degeneration, the reverse occurs. Proteoglycans normally last for about two years. Collagen lasts for many more years. If the balance is disturbed, or if cellular activity falls, the proteoglycan content of the matrix eventually decreases, which affects the mechanical properties of the disc.
Disc cells also respond to changes in mechanical stress. Loading affects disc metabolism, although the mechanisms are not clear. At present it is impossible to predict which mechanical demands encourage a stable balance, and which may favour degradation over synthesis of matrix.
Supply of nutrients
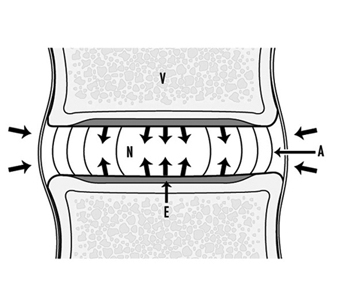
Because the disc receives nutrients from the blood supply of the adjacent tissues, the nutrients such as oxygen and glucose must diffuse through the matrix to the cells in the centre of the disc. Cells may be as much as 7 to 8 mm from the nearest blood supply. Steep gradients develop. At the interface between the disc and the vertebral body, the concentration of oxygen is around 50%, while at the centre of the disc it is below 1%. Disc metabolism is mainly anaerobic. When oxygen falls below 5%, the disc increases production of lactate, a metabolic waste product. The lactate concentration in the centre of the nucleus may be six to eight times higher than that in the blood or interstitium (see figure 5).
Figure 5. The main nutritional pathways to the inter- vertebral disc are by diffusion from the vasculature within the vertebral body (V), through the endplate (E) to the nucleus (N) or from the blood supply outside the annulus (A).
A fall in the supply of nutrients is often suggested to be a major cause of disc degeneration. Endplate permeability of the disc decreases with age, which may impede nutrient transport into the disc and could lead to accumulation of wastes, such as lactate. In discs where nutrient transport has been reduced, oxygen concentrations in the disc centre can fall to very low levels. Here anaerobic metabolism, and consequently lactate production, increases, and the acidity in the disc centre may fall to as low as pH 6.4. Such low values of pH, as well as low oxygen tensions, reduce the rate of matrix synthesis, resulting in a fall in proteoglycan content. In addition, the cells themselves may not survive prolonged exposure to acid pH. A high percentage of dead cells have been found in human discs.
Degeneration of the disc leads to a loss of proteoglycan and a shift in its structure, disorganization of the collagen network and an ingrowth of blood vessels. There is the possibility that some of these changes could be reversed. The disc has been shown to have some capability of repair.
Diseases
Scoliosis: Scoliosis is a sideways bend of the spine, where both the intervertebral disc and vertebral bodies are wedged. It is usually associated with a twisting or rotation of the spine. Because of the manner in which the ribs are attached to the vertebrae this gives rise to a “rib hump”, visible when the affected individual bends forward. Scoliosis may be due to a congenital defect in the spine, such as a wedge-shaped hemi-vertebra, or it may arise secondary to a disorder such as neuromuscular dystrophy. However, in the majority of cases the cause is unknown and it is hence termed idiopathic scoliosis. Pain is rarely a problem in scoliosis and treatment is carried out, mainly to halt further development of the lateral curvature of the spine. (For details on clinical treatment of this and other spinal pathologies see Tidswell 1992.)
Spondylolisthesis: Spondylolisthesis is a forward, horizontal slip of one vertebra in relation to another. It may result from a fracture in the bridge of bone connecting the front to the posterior of the vertebra. Obviously the intervertebral disc between two such vertebrae is stretched and subjected to abnormal loads. The matrix of this disc, and to a lesser extent, adjacent discs, shows changes in composition typical of degeneration—loss of water and proteoglycan. This condition can be diagnosed by x ray.
Ruptured or prolapsed disc: Rupture of the posterior annulus is quite common in physically active young or middle-aged adults. It cannot be diagnosed by x ray unless a discogram is carried out, whereby radio-opaque material is injected into the centre of the disc. A tear can then be demonstrated by the tracking of the discogram fluid. Sometimes isolated and sequestered pieces of disc material can pass through this tear into the spinal canal. Irritation or pressure on the sciatic nerve causes intense pain and paraesthesia (sciatica) in the lower limb.
Degenerative disc disease: This is a term applied to an ill-defined group of patients who present with low-back pain. They may show changes in the x ray appearance, such as a decrease in disc height and possibly osteophyte formation at the rim of the vertebral bodies. This group of patients could represent the endstage of several pathological pathways. For example, untreated annular tears may eventually take on this form.
Spinal stenosis: The narrowing of the spinal canal that occurs in spinal stenosis causes mechanical compression of spinal nerve roots and its blood supply. As such it can lead to symptoms such as weakness, altered reflexes, pain or loss of feeling (paraesthesia), or sometimes have no symptoms. The narrowing of the canal can, in turn, be caused by various factors including protrusion of the intervertebral disc into the canal space, new bone formation in the facet joints (facet hypertrophy) and arthritis with inflammation of other soft connective tissues.
Interpretation of more recent imaging techniques in relation to disc pathology has not been completely established. For example, degenerated discs on magnetic resonance imaging (MRI) give an altered signal from that seen for “normal” discs. However, the correlation between a disc of “degenerate” appearance on MRI and clinical symptoms is poor, with 45% of MRI-degenerate discs being asymptomatic and 37% of patients with low-back pain having normal MRI of the spine.
Risk Factors
Loading
Load on the discs depends on posture. Intradiscal measurements show that the sitting position leads to pressures five times greater than those within the resting spine (see Figure 8). If external weights are lifted this can greatly increase the intradiscal pressure, especially if the weight is held away from the body. Obviously an increased load can lead to a rupture in discs that otherwise might remain intact.
Epidemiological investigations reviewed by Brinckmann and Pope (1990) agree in one respect: repetitive lifting or carrying of heavy objects or performing work in flexed or hyperextended posture represent risk factors for low-back problems. Similarly, certain sports, such as weight lifting, may be associated with a higher incidence of back pain than, for example, swimming. The mechanism is not clear, although the different loading patterns could be relevant.
Smoking
The nutrition of the disc is very precarious, requiring only a small reduction in the flow of nutrients to render it insufficient for the normal metabolism of the disc cells. Cigarette smoking can cause such a reduction because of its effect on the circulatory system outside the intervertebral disc. The transport of nutrients, such as oxygen, glucose or sulphate, into the disc is significantly reduced after only 20 to 30 minutes of smoking, which may explain the higher incidence of low-back pain in individuals who smoke compared to those who do not (Rydevik and Holm 1992).
Vibration
Epidemiological studies have shown that there is an increased incidence of low-back pain in individuals exposed to high levels of vibration. The spine is susceptible to damage at its natural frequencies, particularly from 5 to 10 Hz. Many vehicles excite vibrations at these frequencies. Studies reported by Brinckmann and Pope (1990) have shown a relationship between such vibrations and the incidence of low-back pain. Since vibration has been shown to affect the small blood vessels in other tissues, this may also be the mechanism for its effect on the spine.