Nerve cells are the functional units of the nervous system. The nervous system is believed to have ten thousand million of such cells, called neurons and glia, the glia being present in greater numbers than neurons.
The Neuron
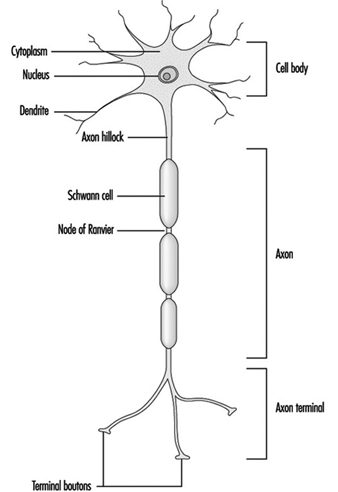
Figure 1 is an idealized diagram of a neuron with its three most important structural features: the cell body, the dendrites and the axon terminal.
Figure 1. The anatomy of the neuron
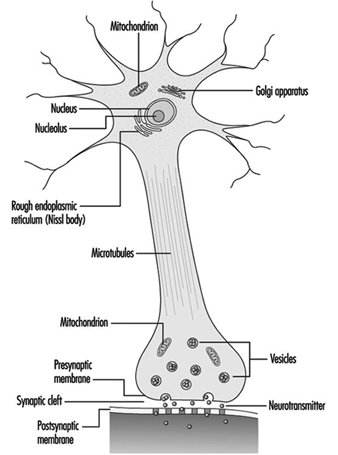
The dendrites are finely branched processes arising near the cell body of a neuron. The dendrites receive excitatory or inhibitory effects via chemical messengers called neurotransmitters. The cytoplasm is the material of the cell body in which the organelles—including the cell nucleus—and other inclusions are found Figure 2. The nucleus contains the cell’s chromatin, or genetic material.
Figure 2. The organelles
The nucleus of the nerve cell is atypical compared with that of other living cells in that, although it contains the genetic material deoxyribonucleic acid (DNA), the DNA is not involved in the process of cell division; that is, after reaching maturity, nerve cells do not divide. (An exception to this rule are the neurons in the nose lining (olfactory epithelium).) The nucleus is rich in ribonucleic acid (RNA), which is necessary for the synthesis of protein. Three types of proteins have been identified: cytosolic proteins, which form the fibrillar elements of the nerve cell; intracondrial proteins, which generate energy for cell activity; and proteins that form membranes and secretory products. Neurons are now conceived of as modified secretory cells. Secretory granules are formed, stored in synaptic vesicles and later released as neurotransmitter substances, the chemical messengers between nerve cells.
The fibrillar elements, which form the skeleton of the neuron, participate in the trophic function of the neuron, acting as vehicles of transmission. Axonal transport can be anterograde (cell body to axon terminal) and retrograde (axon terminal to cell body). From the thickest to the thinnest, three types of fibrillar elements are recognized: microtubules, neurofilaments and microfilaments.
Glial Cells
In contrast to neurons, glial cells do not, by themselves, carry electrical messages. There are two types of glial cells: the macroglia and the microglia. The macroglia is a name given to at least three types of cells: astrocytes, oligodendrocytes and ependymal cells. Microglial cells are primarily scavenger cells for removing debris after neural damage or infection has occurred.
The glial cells also have distinctive microscopic and ultramicroscopic features. Glial cells physically support neurons, but a number of physiological properties are also now beginning to be understood. Among the most important neuron-glial interactions are the glial cell’s role in providing the neurons with nutrients, removing fragments of neurons after their death and, most importantly, contributing to the process of chemical communication. Glial cells, in sharp contrast to neurons, can divide and thus can reproduce themselves. Tumours of the nervous system, for example, result from an abnormal reproduction of glial cells.
Myelin
What appears in the macroscopic observation of neural tissue as “grey matter” and “white matter” has a microscopic and biochemical basis. Microscopically, the grey matter contains the neuronal cell bodies, whereas the white matter is where neural fibres or axons are found. The “white” appearance is due to a sheath—composed of a fatty substance called myelin—covering these fibres. Myelin of the peripheral nerves originates from the membrane of the Schwann cell which wraps around the axon. The myelin of fibres in the central nervous system is provided by the membranes of the oligodendrocytes (a variety of glial cells). Oligodendrocytes usually myelinate several axons, whereas the Schwann cell is associated with only one axon. A discontinuity of the myelin sheath—designated as nodes of Ranvier—exists between continuous Schwann cells or oligodendrocytes. It is estimated that in the longest central motor pathway, up to 2,000 Schwann cells form the myelin cover. Myelin, whose role is to facilitate the propagation of the action potential, may be a specific target of neurotoxic agents. A morphological classification of neurotoxic substances describes characteristic neuropathological changes of the myelin as myelinopathies.
Trophic Function of the Neuron
The normal functions of the neuron include protein synthesis, axonal transport, generation and conduction of the action potential, synaptic transmission, and formation and maintenance of the myelin. Some of the basic trophic functions of the neuron were described as early as the 19th century by sectioning the axons (axotomy). Among the processes uncovered, one of the most important was the Wallerian degeneration—after Waller, the English physiologist who described it.
Wallerian degeneration provides a good opportunity to describe well-known changes in organelles as a result of either traumatic or toxic damage. Parenthetically, the terms used to describe Wallerian degeneration produced by traumatic axotomy are the same ones used to describe changes resulting from neurotoxic agents. At the cellular level, neuropathological changes resulting from toxic damage to neural tissue are far more complex than those occurring as a result of traumatic damage. It is only recently that changes in neurons affected by neurotoxic agents have been observed.
Twenty-four hours after cutting of the axon, the most distinctive feature is swelling of both sides of the mechanical trauma. Swelling results from accumulation of fluids and membranous elements on both sides of the site of injury. These changes are not unlike those observed in a rain-flooded two-way road with vehicles stopped on both sides of the flooded area. In this analogy, stalled vehicles are the swelling. After a few days, regeneration of the ensheathed axons—i.e., those covered with myelin—occurs. Sprouts grow from the proximal stump moving at the rate of 1 to 3 mm per day. Under favourable conditions, sprouts reach the distal (farther from the cell body) stump. When renervation—joining of the stumps—is completed, the basic features of normal transmission have been re-established. The cell body of the injured neuron undergoes profound structural changes in protein synthesis and axonal transport.
If molecular neurobiology is said to be a young discipline, the neurobiology of the neurotoxic processes is even younger, and still in its infancy. True, the molecular basis of action of many neurotoxins and pharmacological agents is now well understood. But with some notable exceptions (e.g., lead, methyl mercury, acrylamide) the molecular basis of toxicity of the vast majority of environmental and neurotoxic agents is unknown. That is why, instead of describing the molecular neurobiology of a select group of occupational and environmental neurotoxic agents, we still are forced to refer to the comparatively abundant strategies and examples from classical neuropharmacology or from work in modern drug manufacture.
Neurotransmitters
A neurotransmitter is a chemical substance which, when released from axon terminals by the action potential, produces the momentary change in electrical potential when another nerve fibre is stimulated. Neurotransmitters stimulate or inhibit adjacent neurons or effector organs such as muscle and glands. Known neurotransmitters and their neural pathways are now being intensively studied, and new ones are constantly being discovered. Some neurological and psychiatric disorders are now understood to be caused by chemical changes in neurotransmission—for example, myasthenia gravis, Parkinson’s disease, certain forms of affective disorders such as depression, severe distortion of thought processes such as in schizophrenia, and Alzheimer’s disease. Although excellent isolated reports on the effect of several environmental and occupational neurotoxic agents on neurotransmission have been published, the body of knowledge is meagre compared with that existing for neuropsychiatric diseases. Pharmacological studies of manufactured drugs require an understanding of how drugs affect neurotransmission. Drug manufacture and neurotransmission research are thus intimately related. The changing views of drug action have been summarized by Feldman and Quenzer (1984).
The effects of neurotoxic agents on neurotransmission are characterized by where in the nervous system they act, their chemical receptors, the time course of their effects, whether neurotoxic agents facilitate, block or inhibit neurotransmission, or whether neurotoxic agents alter the termination or removal of the neurotransmitter’s pharmacological action.
One difficulty experienced by neuroscientists is the need to link known processes that occur at the molecular level in the neuron with events at the cellular level, which in turn may explain how normal and pathological neuropsychological changes occur, as clearly stated in the following which to a large extent still applies: “(A)t the molecular level, an explanation of the action of a drug is often possible; at the cellular level, an explanation is sometimes possible, but at a behavioural level, our ignorance is abysmal” (Cooper, Bloom and Roth 1986).
The Main Components of the Nervous System
Knowledge of the main components of the nervous system is essential for the understanding of the gross neuropsychological manifestations of neurotoxic illness, the rationale for the use of specific techniques for the assessment of nervous system functions, and the understanding of pharmacological mechanisms of neurotoxic action. From a functional standpoint, the nervous system can be divided into two major compartments: The somatic nervous system conveys sensory information (touch, temperature, pain and limb position—even when the eyes are closed) from the body segments and carries the neural pathways that innervate and control the movement of skeletal muscles, such as those of the arms, fingers, legs and toes. The visceral nervous system controls internal organs that are not normally under the influence of blood vessels, the dilation and constriction of the pupils of the eyes and so on.
From an anatomical viewpoint, four main components need to be identified: the central nervous system, the peripheral nervous system including cranial nerves, the autonomic system and the neuroendocrine system.
The Central Nervous System
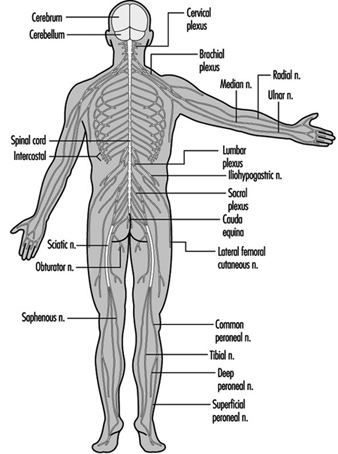
The central nervous system contains the brain and the spinal cord Figure 3. The brain lies in the skull cavity and is protected by the meninges. It is divided into three major components; in ascending order—that is, from the caudal (tail) to cervical (head) portion of the nervous system—they are the hindbrain (also called, the rhombencephalon), the midbrain (the mescencephalon) and the forebrain (the proscencephalon).
Figure 3. The central and peripheral divisions of the nervous system
The hindbrain
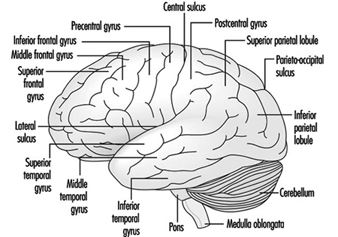
The three major components of the hindbrain are the medulla oblongata, the pons and the cerebellum figure 4.
Figure 4. The brain shown from a lateral side.
The medulla oblongata contains neural structures that control heart rate and breathing, sometimes the targets of neurotoxic agents and drugs causing death. Located between the medulla oblongata and the midbrain, the pons (bridge) derives its names from the large number of fibres traversing its anterior aspect en route to the cerebellar hemispheres. The cerebellum—in Latin, little brain—is characteristically corrugated in appearance. The cerebellum receives sensory information and sends motor messages essential for motor coordination. It is responsible (among other functions) for the execution of fine movements. This scheduling—or programming—requires the adequate timing of sensory inputs and motor responses. The cerebellum is often the target of numerous neurotoxic agents—for example, alcoholic beverages, many industrial solvents, lead—which affect motor responses.
The midbrain
The midbrain is a narrow part of the brain connecting the hindbrain to the forebrain. Structures of the midbrain are the cerebral aqueduct, the tectum, the cerebral peduncles, the substantia nigra and the red nucleus. The cerebral aqueduct is a channel that connects the third with the fourth ventricles (liquid-filled cavities of the brain); the cerebrospinal fluid (CSF) flows through this opening.
The forebrain
This part of the brain is subdivided into diencephalon (“between brain”) and the cerebrum. The major regions of the diencephalon are the thalamus and the hypothalamus. “Thalamus” means “inner room”. The thalami are made up of neuronal groupings, called nuclei, which have five main functions:
- receiving sensory information and sending it to primary areas of the cerebral cortex
- sending information about ongoing movement to motor areas of the cerebral cortex
- sending information on the activity of the limbic system to areas of the cerebral cortex related to this system
- sending information on intrathalamic activity to association areas of the cerebral cortex
- sending information of brain-stem reticular formation activity to widespread areas of the cerebral cortex.
The name hypothalamus means “under the thalamus”. It forms the base of the third ventricle, an important reference point for the imaging of the brain. The hypothalamus is a complex, minute neural structure responsible for many aspects of behaviour such as basic biological drives, motivation and emotion. It is the link between the nervous and the neuroendocrine system, to be reviewed below. The pituitary gland (also called the hypophysis) is linked by neurons to the hypothalamic nuclei. It is well established that the hypothalamic nerve cells perform many neurosecretory functions. The hypothalamus is linked with many other major regions of the brain including the rhinencephalon—the primitive cortex originally associated with olfaction—and the limbic system, including the hippocampus.
The cerebral cortex is the largest component of the brain, consisting of two cerebral hemispheres connected by a mass of white matter called the corpus callosum. The cerebral cortex is the surface layer of each cerebral hemisphere. Deep sulci in the cerebral cortex—the central and the lateral sulci Figure 4 —are taken as reference points to separate anatomical regions of the brain. The frontal lobe lies in front of the central sulcus. The parietal lobe begins at the back of the central sulcus, and lies next to the occipital lobe, which occupies the posterior portion of the brain. The temporal lobe begins well inside the folding of the lateral sulcus and extends into the ventral aspects of the brain hemispheres. Two important components of the cerebrum are the basal ganglia and the limbic system.
The basal ganglia are nuclei—that is, clusters of nerve cells—located toward the centre of the brain. The basal ganglia comprise major centres of the extra-pyramidal motor system. (The pyramidal system, to which the term is contrasted, is involved in the voluntary control of movement.) The extrapyramidal system is selectively affected by many neurotoxic agents (e.g., manganese). In the past two decades, important discoveries have been made concerning the role these nuclei play in several neural degenerative diseases (e.g., Parkinson’s disease, Huntington’s chorea).
The limbic system is comprised of convoluted neural structures branching out into many directions and establishing connections with many “old” regions of the brain, particularly with the hypothalamus. It is involved in the control of emotional expression. The hippocampus is believed to be a structure where many memory processes occur.
The spinal cord
The spinal cord is a whitish structure situated within the vertebral canal. It is divided into four regions: cervical, thoracic, lumbar and sacral-coccyxeal. The two most easily recognizable features of the spinal cord are the grey matter containing the cell bodies of the neurons, and the white matter containing the myelinated axons of the neurons. The ventral region of the spinal cord’s grey matter contains nerve cells that regulate motor function; the middle region of the thoracic spinal cord is associated with autonomic functions. The dorsal portion receives sensory information from the spinal nerves.
The Peripheral Nervous System
The peripheral nervous system includes those neurons that are outside the central nervous system. The term peripheral describes the anatomical distribution of this system, but functionally it is artificial. The cell bodies of peripheral motor fibres, for example, are located within the central nervous system. In experimental, clinical and epidemiological neurotoxicology, the term peripheral nervous system (PNS) describes a system that is selectively vulnerable to the effects of toxic agents and that is able to regenerate.
The spinal nerves
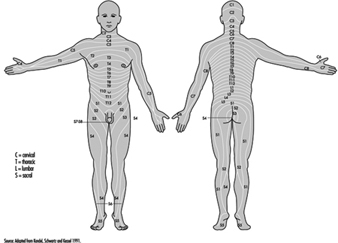
The ventral and dorsal roots are where the peripheral nerves enter and leave the spinal cord along its length. Adjoining vertebrae contain openings to allow root fibres forming the spinal nerves to leave the spinal canal. There are 31 pairs of spinal nerves, which are named according to the region of the vertebral column with which they are associated: 8 cervical, 12 thoracic, 5 lumbar, 5 sacral and 1 coccyxeal. A metamera is a region of the body innervated by a spinal nerve figure 5.
Figure 5. The segmental distribution of the spinal nerves (the metamera).
Carefully examining the motor and sensory functions of metamerae, neurologists can infer the location of lesions where damage has occurred.
Table 1. Names and main functions of each pair of cranial nerves
| Nerve1 | Conducts impulses | Functions |
| I. Olfactory | From nose to brain | Sense of smell |
| II. Optic | From eye to brain | Vision |
| III. Oculomotor | From brain to eye muscles | Eye movements |
| IV. Trochlear | From brain to external eye muscles | Eye movements |
| V. Trigeminal (or trifacial) |
From skin and mucous membrane of head and from teeth to brain; also from brain to chewing muscles | Sensations of face, scalp and teeth; chewing movements |
| VI. Abducens | From brain to external eye muscles | Turning eyes outward |
| VII. Facial | From taste buds of tongue to brain; from brain to face muscles | Sense of taste; contraction of muscles of facial expression |
| VIII. Acoustic | From ear to brain | Hearing; sense of balance |
| IX. Glossopharyngeal | From throat and taste buds of tongue to brain; also from brain to throat muscles and salivary glands | Sensations of throat, taste, swallowing movements, secretion of saliva |
| X. Vagus | From throat, larynx, and organs in thoracic and abdominal cavities to brain; also from brain to muscles of throat and to organs in thoracic and abdominal cavities | Sensations of throat, larynx, and for thoracic and abdominal organs; swallowing, voice production, slowing of heartbeat, acceleration of peristalsis |
| XI. Spinal accessory | From brain to certain shoulder and neck muscles | Shoulder movements; turning movements of head |
| XII. Hypoglossal | From brain to muscles of tongue | Tongue movements |
1 The first letter of the words of the following sentence are the first letters of the names of cranial nerves: “On Old Olympus’ Tiny Tops A Finn and German Viewed Some Hops”. Many generations of students have used this or a similar sentence to help them remember the names of cranial nerves.
The cranial nerves
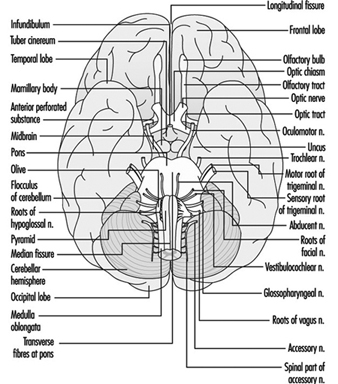
Brain stem is a comprehensive term that designates the region of the nervous system that includes the medulla, the pons and the midbrain. The brain stem is a continuation of the spinal cord upward and forward (ventrally). It is in this region where most of the cranial nerves make their exits and entrances. There are 12 pairs of cranial nerves; Table 1 describes the name and main function of each pair and Figure 6 shows the entrance and exits of some cranial nerves in the brain.
Figure 6. The brain shown from below with the entrance and exits of many cranial nerves.
The Autonomic Nervous System
The autonomic nervous system is that part of the nervous system controlling the activity of the visceral components of the human body. It is called “autonomic” because it performs its functions automatically, meaning that its functioning cannot be easily controlled at will. From an anatomical point of view, the autonomic system has two main components: the sympathetic and the parasympathetic nervous system. The sympathetic nerves controlling visceral activity arise from the thoracic and lumbar portions of the spinal cord; parasympathetic nerves arise from the brain stem and the sacral portion of the spinal cord.
From a physiological point of view, no single generalization can be made that applies to the manner in which the sympathetic and the parasympathetic nervous systems control different body organs. In most cases, visceral organs are innervated by both systems, and each type has an opposite effect in a system of checks and balances. The heart, for example, is innervated by sympathetic nerves whose excitation produces an acceleration of the heartbeat, and by parasympathetic nerves whose excitation produce a slowing of the heartbeat. Either system can stimulate or inhibit the organs it innervates. In other cases, organs are predominantly or exclusively controlled by one system or the other. A vital function of the autonomic nervous system is the maintenance of homeostasis (stable state of equilibrium) and for the adaptation of the animal body to its external environment. Homeostasis is the state of equilibrium of body functions achieved by an active process; the control of body temperature, water and electrolytes are all examples of homeostatic processes.
From the pharmacological point of view, there is no single neurotransmitter associated with either sympathetic or parasympathetic functions, as was once believed. The old view that acetylcholine was the predominant transmitter of the autonomic system had to be abandoned when new classes of neurotransmitters and neuromodulators were found (e.g., dopamine, serotonin, purines and various neuropeptides).
Neuroscientists have recently revived the behavioural point of view of the autonomic nervous system. The autonomic nervous system is involved in the fight-or-flight instinctive reaction still present in humans, which is, for the most part, the basis for the physiological reactions caused by stress. Interactions between the nervous system and immunological functions are possible through the autonomic nervous system. Emotions that originate from the autonomic nervous system can be expressed via the skeletal muscles.
The autonomic control of smooth muscles
The muscles of the viscera—except for the heart—are the smooth muscles. Heart muscle has characteristics of both skeletal and smooth muscle. Like skeletal muscles, smooth muscles also contain the two proteins actin and, in smaller proportions, myosin. Unlike skeletal muscles, they do not present the regular organization of sarcolemes, the contractile unit of the muscle fibre. The heart is unique in that it can generate myogenic activity—even after its neural innervations have been severed, it can contract and relax for several hours by itself.
The neuromuscular coupling in smooth muscles differs from that of skeletal muscles. In skeletal muscles, the neuromuscular junction is the link between the nerve and the muscle fibres. In smooth muscle, there is no neuromuscular junction; the nerve endings enter the muscle, spreading in all directions. Electrical events inside the smooth muscle therefore are much slower than those in skeletal muscles. Finally, smooth muscle has the unique characteristic of exhibiting spontaneous contractions, such as that exhibited by the gut. To a large extent, the autonomic nervous system regulates the smooth muscles’ spontaneous activity.
The central components of the autonomic nervous system
The main role of the autonomic nervous system is to regulate the activity of smooth muscles, heart, glands in the digestive tract, sweat glands, and adrenal and other endocrine glands. The autonomic nervous system has a central component—the hypothalamus, located at the base of the brain—where many autonomic functions are integrated. Most importantly, the central components of the autonomic system are directly involved in the regulation of biological drives (temperature regulation, hunger, thirst, sex, urination, defecation and so on), motivation, emotion and to a great extent in “psychological” functions such as moods, affect and feelings.
Neuroendocrine System
Glands are the organs of the endocrine system. They are called endocrine glands because their chemical messages are delivered inside the body, directly into the blood stream (in contrast with exocrine glands, such as sweat glands, whose secretions appear on the outer surface of the body). The endocrine system provides slow but long-lasting control over organs and tissues through chemical messengers called hormones. Hormones are the main regulators of body metabolism. But, because of intimate links among the central, peripheral, and autonomic nervous systems, the neuroendocrine system—a term that captures such complex links—is now conceived of as a powerful modifier of the structure and function of the human body and behaviour.
Hormones have been defined as chemical messengers which are released from cells into the bloodstream to exert their action on target cells some distance away. Until recently, hormones were distinguished from neurotransmitters, discussed above. The latter are chemical messengers released from neurons onto a synapse between the nerve terminals and another neuron or an effector (i.e., muscle or gland). However, with the discovery that classical neurotransmitters such as dopamine can also act as hormones, the distinction between neurotransmitters and hormones is now less and less clear. Thus, based on purely anatomical considerations, hormones derived from nerve cells may be called neurohormones. From a functional point of view, the nervous system can be thought of as a truly neurosecretory system.
The hypothalamus controls endocrine functions through a link with the pituitary gland (also called the hypophysis, a tiny gland located at the base of the brain). Until the middle 1950s the endocrine glands were viewed as a separate system governed by the pituitary gland, often called the “master gland”. At that time, a neurovascular hypothesis was advanced that established the functional role of the hypothalamic/hypophysial factors in the control of endocrine function. In this view, the endocrine hypothalamus provides the final common neuroendocrine pathway in the control of the endocrine system. It has now been firmly established that the endocrine system is itself regulated by the central nervous system as well as the endocrine inputs. Thus, neuroendocrinology is now an appropriate term to describe the discipline that studies the reciprocal integrated roles of the nervous and the endocrine systems in the control of physiological processes.
With increasing understanding of neuroendocrinology, original divisions are breaking down. The hypothalamus, which is located above and connected to the pituitary gland, is the link between the nervous and the endocrine systems, and many of its nerve cells perform secretory functions. It is also linked with other major regions of the brain, including the rhinencephalon—the primitive cortex originally associated with olfaction or sense of smell—and the limbic system, associated with emotions. It is in the hypothalamus that hormones released by the posterior pituitary gland are produced. The hypothalamus also produces substances that are called releasing and inhibiting hormones. These act on the adenohypophysis, causing it to enhance or inhibit the production of anterior pituitary gland hormones, which act on glands located elsewhere (thyroid, adrenal cortex, ovaries, testicles and others).