Decisions affecting the health, well-being, and employability of individual workers or an employer’s approach to health and safety issues must be based on data of good quality. This is especially so in the case of biological monitoring data and it is therefore the responsibility of any laboratory undertaking analytical work on biological specimens from working populations to ensure the reliability, accuracy and precision of its results. This responsibility extends from providing suitable methods and guidance for specimen collection to ensuring that the results are returned to the health professional responsible for the care of the individual worker in a suitable form. All these activities are covered by the expression of quality assurance.
The central activity in a quality assurance programme is the control and maintenance of analytical accuracy and precision. Biological monitoring laboratories have often developed in a clinical environment and have taken quality assurance techniques and philosophies from the discipline of clinical chemistry. Indeed, measurements of toxic chemicals and biological effect indicators in blood and urine are essentially no different from those made in clinical chemistry and in clinical pharmacology service laboratories found in any major hospital.
A quality assurance programme for an individual analyst starts with the selection and establishment of a suitable method. The next stage is the development of an internal quality control procedure to maintain precision; the laboratory needs then to satisfy itself of the accuracy of the analysis, and this may well involve external quality assessment (see below). It is important to recognize however, that quality assurance includes more than these aspects of analytical quality control.
Method Selection
There are several texts presenting analytical methods in biological monitoring. Although these give useful guidance, much needs to be done by the individual analyst before data of suitable quality can be produced. Central to any quality assurance programme is the production of a laboratory protocol that must specify in detail those parts of the method which have the most bearing on its reliability, accuracy, and precision. Indeed, national accreditation of laboratories in clinical chemistry, toxicology, and forensic science is usually dependent on the quality of the laboratory’s protocols. Development of a suitable protocol is usually a time-consuming process. If a laboratory wishes to establish a new method, it is often most cost-effective to obtain from an existing laboratory a protocol that has proved its performance, for example, through validation in an established international quality assurance programme. Should the new laboratory be committed to a specific analytical technique, for example gas chromatography rather than high-performance liquid chromatography, it is often possible to identify a laboratory that has a good performance record and that uses the same analytical approach. Laboratories can often be identified through journal articles or through organizers of various national quality assessment schemes.
Internal Quality Control
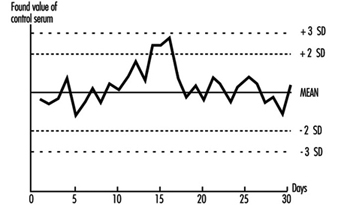
The quality of analytical results depends on the precision of the method achieved in practice, and this in turn depends on close adherence to a defined protocol. Precision is best assessed by the inclusion of “quality control samples” at regular intervals during an analytical run. For example, for control of blood lead analyses, quality control samples are introduced into the run after every six or eight actual worker samples. More stable analytical methods can be monitored with fewer quality control samples per run. The quality control samples for blood lead analysis are prepared from 500 ml of blood (human or bovine) to which inorganic lead is added; individual aliquots are stored at low temperature (Bullock, Smith and Whitehead 1986). Before each new batch is put into use, 20 aliquots are analysed in separate runs on different occasions to establish the mean result for this batch of quality control samples, as well as its standard deviation (Whitehead 1977). These two figures are used to set up a Shewhart control chart (figure 27.2). The results from the analysis of the quality control samples included in subsequent runs are plotted on the chart. The analyst then uses rules for acceptance or rejection of an analytical run depending on whether the results of these samples fall within two or three standard deviations (SD) of the mean. A sequence of rules, validated by computer modelling, has been suggested by Westgard et al. (1981) for application to control samples. This approach to quality control is described in textbooks of clinical chemistry and a simple approach to the introduction of quality assurance is set forth in Whitehead (1977). It must be emphasized that these techniques of quality control depend on the preparation and analysis of quality control samples separately from the calibration samples that are used on each analytical occasion.
Figure 27.2 Shewhart control chart for quality control samples
This approach can be adapted to a range of biological monitoring or biological effect monitoring assays. Batches of blood or urine samples can be prepared by addition of either the toxic material or the metabolite that is to be measured. Similarly, blood, serum, plasma, or urine can be aliquotted and stored deep-frozen or freeze-dried for measurement of enzymes or proteins. However, care has to be taken to avoid infective risk to the analyst from samples based on human blood.
Careful adherence to a well-defined protocol and to rules for acceptability is an essential first stage in a quality assurance programme. Any laboratory must be prepared to discuss its quality control and quality assessment performance with the health professionals using it and to investigate surprising or unusual findings.
External Quality Assessment
Once a laboratory has established that it can produce results with adequate precision, the next stage is to confirm the accuracy (“trueness”) of the measured values, that is, the relationship of the measurements made to the actual amount present. This is a difficult exercise for a laboratory to do on its own but can be achieved by taking part in a regular external quality assessment scheme. These have been an essential part of clinical chemistry practice for some time but have not been widely available for biological monitoring. The exception is blood lead analysis, where schemes have been available since the 1970s (e.g., Bullock, Smith and Whitehead 1986). Comparison of analytical results with those reported from other laboratories analysing samples from the same batch allows assessment of a laboratory’s performance compared with others, as well as a measure of its accuracy. Several national and international quality assessment schemes are available. Many of these schemes welcome new laboratories, as the validity of the mean of the results of an analyte from all the participating laboratories (taken as a measure of the actual concentration) increases with the number of participants. Schemes with many participants are also more able to analyse laboratory performance according to analytical method and thus advise on alternatives to methods with poor performance characteristics. In some countries, participation in such a scheme is an essential part of laboratory accreditation. Guidelines for external quality assessment scheme design and operation have been published by the WHO (1981).
In the absence of established external quality assessment schemes, accuracy may be checked using certified reference materials which are available on a commercial basis for a limited range of analytes. The advantages of samples circulated by external quality assessment schemes are that (1) the analyst does not have fore-knowledge of the result, (2) a range of concentrations is presented, and (3) as definitive analytical methods do not have to be employed, the materials involved are cheaper.
Pre-analytical Quality Control
Effort spent in attaining good laboratory accuracy and precision is wasted if the samples presented to the laboratory have not been taken at the correct time, if they have suffered contamination, have deteriorated during transport, or have been inadequately or incorrectly labelled. It is also bad professional practice to submit individuals to invasive sampling without taking adequate care of the sampled materials. Although sampling is often not under the direct control of the laboratory analyst, a full quality programme of biological monitoring must take these factors into account and the laboratory should ensure that syringes and sample containers provided are free from contamination, with clear instructions about sampling technique and sample storage and transport. The importance of the correct sampling time within the shift or working week and its dependence on the toxicokinetics of the sampled material are now recognized (ACGIH 1993; HSE 1992), and this information should be made available to the health professionals responsible for collecting the samples.
Post-analytical Quality Control
High-quality analytical results may be of little use to the individual or health professional if they are not communicated to the professional in an interpretable form and at the right time. Each biological monitoring laboratory should develop reporting procedures for alerting the health care professional submitting the samples to abnormal, unexpected, or puzzling results in time to allow appropriate action to be taken. Interpretation of laboratory results, especially changes in concentration between successive samples, often depends on knowledge of the precision of the assay. As part of total quality management from sample collection to return of results, health professionals should be given information concerning the biological monitoring laboratory’s precision and accuracy, as well as reference ranges and advisory and statutory limits, in order to help them in interpreting the results.
