Major Sectors and Processes
Fibre Sources for Pulp and Paper
The basic structure of pulp and paper sheets is a felted mat of cellulose fibres held together by hydrogen bonds. Cellulose is a polysaccharide with 600 to 1,500 repeated sugar units. The fibres have high tensile strength, will absorb the additives used to modify pulp into paper and board products, and are supple, chemically stable and white. The purpose of pulping is to separate cellulose fibres from the other components of the fibre source. In the case of wood, these include hemicelluloses (with 15 to 90 repeated sugar units), lignins (highly polymerized and complex, mainly phenyl propane units; they act as the “glue” that cements the fibres together), extractives (fats, waxes, alcohols, phenols, aromatic acids, essential oils, oleoresins, stearols, alkaloids and pigments), and minerals and other inorganics. As shown in table 1, the relative proportions of these components vary according to the fibre source.
Table 1. Chemical constituents of pulp and paper fibre sources (%)
|
Softwoods |
Hardwoods |
Straw |
Bamboo |
Cotton |
|
|
Carbohydrates |
|||||
|
a-cellulose |
38–46 |
38–49 |
28–42 |
26–43 |
80–85 |
|
Hemicellulose |
23–31 |
20–40 |
23–38 |
15–26 |
nd |
|
Lignin |
22–34 |
16–30 |
12–21 |
20–32 |
nd |
|
Extractives |
1–5 |
2–8 |
1–2 |
0.2–5 |
nd |
|
Minerals and other |
|
|
|
|
|
nd = no data available.
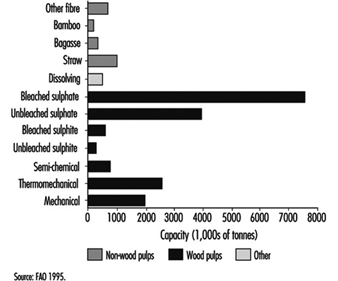
Coniferous and deciduous trees are the major fibre sources for pulp and paper. Secondary sources include straws from wheat, rye and rice; canes, such as bagasse; woody stalks from bamboo, flax and hemp; and seed, leaf or bast fibres, such as cotton, abaca and sisal. The majority of pulp is made from virgin fibre, but recycled paper accounts for an increasing proportion of production, up from 20% in 1970 to 33% in 1991. Wood-based production accounted for 88% of worldwide pulp capacity in 1994 (176 million tonnes, figure 1); therefore, the description of pulp and paper processes in the following article focuses on wood-based production. The basic principles apply to other fibres as well.
Figure 1. Worldwide pulp capacities, by pulp type
Wood Handling
Wood may arrive at a pulp mill woodyard in the form of raw logs or as chips from a lumber mill. Some pulp mill operations have on-site sawmills (often called “woodrooms”) which produce both marketable lumber and stock for the pulp mill. Sawmilling is discussed in detail in the chapter Lumber. This article discusses those elements of wood preparation which are specific to pulp mill operations.
The wood preparation area of a pulp mill has several basic functions: to receive and meter the wood supply to the pulping process at the rate demanded by the mill; to prepare the wood so that it meets the mill’s feed specifications for species, cleanliness and dimensions; and to collect any material rejected by the previous operations and send it to final disposal. Wood is converted into chips or logs suitable for pulping in a series of steps which may include debarking, sawing, chipping and screening.
Logs are debarked because bark contains little fibre, has a high extractives content, is dark, and often carries large quantities of grit. Debarking can be done hydraulically with high-pressure water jets, or mechanically by rubbing logs against each other or with metal cutting tools. Hydraulic debarkers may be used in coastal areas; however, the effluent generated is difficult to treat and contributes to water pollution.
Debarked logs may be sawn into short lengths (1 to 6 metres) for stone groundwood pulping or chipped for refiner mechanical or chemical pulping methods. Chippers tend to produce chips with a considerable size range, but pulping requires chips of very specific dimensions to ensure constant flow through refiners and uniform cooking in digesters. Chips are therefore passed over a series of screens whose function is to separate chips on the basis of length or thickness. Oversized chips are rechipped, while undersized chips are either used as waste fuel or are metered back into the chip flow.
The requirements of the particular pulping process and chip conditions will dictate the duration of chip storage (figure 1; note the different types of chips available for pulping). Depending on fibre supply and mill demand, a mill will maintain a 2 to 6 week unscreened chip inventory, usually in large outdoor chip piles. Chips may degrade through auto-oxidation and hydrolysis reactions or fungal attack of the wood components. In order to avoid contamination, short-term inventories (hours to days) of screened chips are stored in chip silos or bins. Chips for sulphite pulping may be stored outside for several months to allow volatilization of extractives which may cause problems in subsequent operations. Chips used in kraft mills where turpentine and tall oil are recovered as commercial products typically proceed directly to pulping.
Figure 1. Chip storage area with front end loaders
George Astrakianakis
Pulping
Pulping is the process by which the bonds within the wood structure are ruptured either mechanically or chemically. Chemical pulps can be produced by either alkaline (i.e., sulphate or kraft) or acidic (i.e., sulphite) processes. The highest proportion of pulp is produced by the sulphate method, followed by mechanical (including semi-chemical, thermomechanical and mechanical) and sulphite methods (figure 1). Pulping processes differ in the yield and quality of the product, and for chemical methods, in the chemicals used and the proportion that can be recovered for reuse.
Figure 1. Worldwide pulp capacities, by pulp type
Mechanical Pulping
Mechanical pulps are produced by grinding wood against a stone or between metal plates, thereby separating the wood into individual fibres. The shearing action breaks cellulose fibres, so that the resulting pulp is weaker than chemically separated pulps. The lignin connecting cellulose to hemicellulose is not dissolved; it merely softens, allowing the fibres to be ground out of the wood matrix. The yield (proportion of original wood in pulp) is usually greater than 85%. Some mechanical pulping methods also use chemicals (i.e., the chemi-mechanical pulps); their yields are lower since they remove more of the non-cellulosic materials.
In stone groundwood pulping (SGW), the oldest and historically most common mechanical method, fibres are removed from short logs by pressing them against a rotating abrasive cylinder. In refiner mechanical pulping (RMP, figure 2), which gained popularity after it became commercially viable in the 1960s, wood chips or sawdust are fed through the centre of a disc refiner, where they are shredded into finer pieces as they are pushed out through progressively narrower bars and grooves. (In figure 2, the refiners are enclosed in the middle of the picture and their large motors are on the left. Chips are supplied though the large diameter pipes, and pulp exits the smaller ones.) A modification of RMP is thermomechanical pulping (TMP), in which the chips are steamed before and during refining, usually under pressure.
Figure 2. Refiner mechanical pulping
One of the earliest methods of producing chemi-mechanical pulps involved pre-steaming logs before boiling them in chemical pulping liquors, then grinding them in stone grinders to produce “chemi-groundwood” pulps. Modern chemi-mechanical pulping uses disc refiners with chemical treatment (e.g., sodium bisulphite, sodium hydroxide) either prior to, during or after refining. Pulps produced in this manner are referred to either as chemi-mechanical pulps (CMP) or chemi-thermomechanical pulps (CTMP), depending on whether refining was carried out at atmospheric or elevated pressure. Specialized variations of CTMP have been developed and patented by a number of organizations.
Chemical Pulping and Recovery
Chemical pulps are produced by chemically dissolving the lignin between the wood fibres, thereby enabling the fibres to separate relatively undamaged. Because most of the non-fibrous wood components are removed in these processes, yields are usually in the order of 40 to 55%.
In chemical pulping, chips and chemicals in aqueous solution are cooked together in a pressure vessel (digester, figure 3) which can be operated on a batch or continuous basis. In batch cooking, the digester is filled with chips through a top opening, the digestion chemicals are added, and the contents cooked at elevated temperature and pressure. Once the cook is complete, the pressure is released, “blowing” the delignified pulp out of the digester and into a holding tank. The sequence is then repeated. In continuous digesting, pre-steamed chips are fed into the digester at a continuous rate. Chips and chemicals are mixed together in the impregnation zone at the top of the digester and then proceed through the upper cooking zone, the lower cooking zone, and the washing zone before being blown into the blow tank.
Figure 3. Continuous kraft digestor, with chip conveyor under construction
Canfor Library
The digesting chemicals are recovered in most chemical pulping operations today. The principal objectives are to recover and reconstitute digestion chemicals from the spent cooking liquor, and to recover heat energy by burning the dissolved organic material from the wood. The resulting steam and electricity supplies some, if not all, of the mill’s energy needs.
Sulphate Pulping and Recovery
The sulphate process produces a stronger, darker pulp than other methods and requires chemical recovery to compete economically. The method evolved from soda pulping (which uses only sodium hydroxide for digestion) and began to gain prominence in the industry from the 1930s to 1950s with the development of chlorine dioxide bleaching and chemical recovery processes, which also produced steam and power for the mill. The development of corrosion-proof metals, such as stainless steel, to handle the acidic and alkaline pulp mill environments also played a role.
The cooking mixture (white liquor) is sodium hydroxide (NaOH, “caustic”) and sodium sulphide (Na2S). Modern kraft pulping is usually carried out in continuous digesters often lined with stainless steel (figure 3). The temperature of the digester is raised slowly to approximately 170°C and held at that level for approximately 3 to 4 hours. The pulp (called brown stock because of its colour) is screened to remove uncooked wood, washed to remove the spent cooking mixture (now black liquor), and sent either to the bleach plant or to the pulp machine room. Uncooked wood is either returned to the digester or sent to the power boiler to be burned.
The black liquor collected from the digester and brown stock washers contains dissolved organic material whose exact chemical composition depends on the wood species pulped and the cooking conditions. The liquor is concentrated in evaporators until it contains less than 40% water, then sprayed into the recovery boiler. The organic component is consumed as fuel, generating heat which is recovered in the upper section of the furnace as high-temperature steam. The unburned inorganic component collects at the bottom of the boiler as a molten smelt. The smelt flows out of the furnace and is dissolved in a weak caustic solution, producing “green liquor” containing primarily dissolved Na2S and sodium carbonate (Na2CO3). This liquor is pumped to a recausticizing plant, where it is clarified, then reacted with slaked lime
(Ca(OH)2), forming NaOH and calcium carbonate (CaCO3). The white liquor is filtered and stored for subsequent use. CaCO3 is sent to a lime kiln, where it is heated to regenerate lime (CaO).
Sulphite Pulping and Recovery
Sulphite pulping dominated the industry from the late 1800s to the mid-1900s, but the method used during this era was limited by the types of wood which could be pulped and the pollution created by discharging untreated waste cooking liquor into waterways. Newer methods have overcome many of these problems, but sulphite pulping is now a small segment of the pulp market. Although sulphite pulping usually uses acid digestion, both neutral and basic variations exist.
The cooking liquor of sulphurous acid (H2SO3) and bisulphite ion (HSO3–) is prepared on-site. Elemental sulphur is burned to produce sulphur dioxide (SO2), which is passed up through an absorption tower that contains water and one of four alkaline bases (CaCO3, the original sulphite base, Na2CO3, magnesium hydroxide (Mg(OH)2) or ammonium hydroxide (NH4OH)) which produce the acid and ion and control their proportions. Sulphite pulping is usually carried out in brick-lined batch digesters. To avoid unwanted reactions, the digester is heated slowly to a maximum temperature of 130 to 140°C and the chips are cooked for a long time (6 to 8 hours). As the digester pressure increases, gaseous sulphur dioxide (SO2) is bled off and remixed with the raw cooking acid. When approximately 1 to 1.5 hours of cooking time remains, heating is discontinued and the pressure is decreased by bleeding off gas and steam. The pulp is blown into a holding tank, then washed and screened.
The spent digestion mixture, called red liquor, can be used for heat and chemical recovery for all but calcium-bisulphite-base operations. For ammonia-base sulphite pulping, the dilute red liquor is first stripped to remove residual SO2, then concentrated and burned. The flue gas containing SO2 is cooled and passed through an absorption tower where fresh ammonia combines with it to regenerate the cooking liquor. Finally, the liquor is filtered, fortified with fresh SO2 and stored. The ammonia cannot be recovered because it is converted into nitrogen and water in the recovery boiler.
In magnesium-base sulphite pulping, burning the concentrated pulping liquor gives magnesium oxide (MgO) and SO2, which are easily recovered. No smelt is produced in this process; rather MgO is collected from the flue gas and slaked with water to produce magnesium hydroxide (Mg(OH)2). SO2 is cooled and combined with the Mg(OH)2 in an absorption tower to reconstitute the cooking liquor. The magnesium bisulphite (Mg(HSO3)2) is then fortified with fresh SO2 and stored. Recovery of 80 to 90% of the cooking chemicals is possible.
Recovery of sodium-base sulphite cooking liquor is more complicated. Concentrated spent liquor is incinerated, and approximately 50% of the sulphur is converted into SO2. The remainder of the sodium and sulphur is collected at the bottom of the recovery boiler as a smelt of Na2S and Na2CO3. The smelt is dissolved to produce green liquor, which is converted to sodium bisulphite (NaHSO3) in several steps. The NaHSO3 is fortified and stored. The regeneration process produces reduced sulphur gases, in particular hydrogen sulphide (H2S).
Bleaching
Bleaching is a multi-stage process that refines and brightens raw pulp. The objective is to dissolve (chemical pulps) or modify (mechanical pulps) the brown-coloured lignin that was not removed during pulping, while maintaining the integrity of the pulp fibres. A mill produces customized pulp by varying the order, concentration and reaction time of the bleaching agents.
Each bleaching stage is defined by its bleaching agent, pH (acidity), temperature and duration (table 1). After each bleaching stage, the pulp may be washed with caustic to remove spent bleaching chemicals and dissolved lignin before it progresses to the next stage. After the last stage, the pulp is pumped through a series of screens and cleaners to remove any contaminants such as dirt or plastic. It is then concentrated and conveyed to storage.
Table 1. Bleaching agents and their conditions of use
|
Symbol |
Concentration |
pH |
Consistency* |
Temperature |
Time (h) |
|
|
Chlorine (Cl2) |
C |
2.5–8 |
2 |
3 |
20–60 |
0.5–1.5 |
|
Sodium hydroxide (NaOH) |
E |
1.5–4.2 |
11 |
10–12 |
<80 |
1–2 |
|
Chlorine dioxide (ClO2) |
D |
~1 |
0–6 |
10–12 |
60–75 |
2–5 |
|
Sodium hypochlorite (NaOCl) |
H |
1–2 |
9–11 |
10–12 |
30–50 |
0.5–3 |
|
Oxygen (O2) |
O |
1.2–1.9 |
7–8 |
25–33 |
90–130 |
0.3–1 |
|
Hydrogen peroxide (H2O2) |
P |
0.25 |
10 |
12 |
35–80 |
4 |
|
Ozone (O3) |
Z |
0.5–3.5 |
2–3 |
35–55 |
20–40 |
<0.1 |
|
Acid washing (SO2) |
A |
4–6 |
1.8–5 |
1.5 |
30–50 |
0.25 |
|
Sodium dithionite (NaS2O4) |
Y |
1–2 |
5.5–8 |
4–8 |
60–65 |
1–2 |
* Concentration of fibre in water solution.
Historically, the most common bleaching sequence used to produce market-grade bleached kraft pulp is based on the five-stage CEDED process (see table 1 for definition of symbols). The first two stages of bleaching complete the delignification process and are considered extensions of pulping. Because of environmental concerns about chlorinated organics in pulp mill effluents, many mills substitute chlorine dioxide (ClO2) for a portion of the chlorine (Cl2) used in the first bleaching stage (CDEDED) and use oxygen (O2) pre-treatment during the first caustic extraction (CDEODED). The current trend in Europe and North America is towards complete substitution with ClO2 (e.g., DEDED) or elimination of both Cl2 and ClO2. Where ClO2 is used, sulphur dioxide (SO2) is added during the final washing stage as an “antichlor” to stop the ClO2 reaction and to control the pH. Newly developed chlorine-free bleaching sequences (e.g., OAZQP, OQPZP, where Q = chelation) use enzymes, O2, ozone (O3), hydrogen peroxide (H2O2), peracids and chelating agents such as ethylene diamine tetracetic acid (EDTA). Totally chlorine-free bleaching had been adopted at eight mills worldwide by 1993. Because these newer methods eliminate the acidic bleaching steps, acid washing is a necessary addition to the initial stages of kraft bleaching to allow removal of metals bound to the cellulose.
Sulphite pulps are generally easier to bleach than kraft pulps because of their lower lignin content. Short bleaching sequences (e.g., CEH, DCEHD, P, HP, EPOP) can be used for most paper grades. For dissolving-grade sulphite pulps used in the production of rayon, cellophane and so on, both hemicellulose and lignin are removed, requiring more complex bleaching sequences (e.g., C1C2ECHDA). The final acid wash is both for metal control and antichlor purposes. The effluent load for dissolving-grade sulphite pulps is much greater because so much of the raw wood is consumed (typical yield 50%) and more water is used.
The term brightening is used to describe bleaching of mechanical and other high-yield pulps, because they are whitened by destroying chromophoric groups without dissolving the lignin. Brightening agents include H2O2 and/or sodium hydrosulphite (NaS2O4). Historically, zinc hydrosulphite (ZnS2O4) was commonly used, but has been largely eliminated because of its toxicity in effluent. Chelating agents are added before bleaching to neutralize any metal ions, thereby preventing the formation of coloured salts or the decomposition of H2O2. The effectiveness of mechanical pulp bleaching depends on the species of wood. Hardwoods (e.g., poplar and cottonwood) and softwoods (e.g., spruce and balsam) that are low in lignin and extractives can be bleached to a higher brightness level than the more resinous pine and cedar.
Recycled Paper Operations
The use of waste or recycled paper as the raw material for pulp production has increased during the last several decades, and some paper plants depend almost completely on waste paper. In some countries, waste paper is separated from other household waste at the source before it is collected. In other countries separation by grade (e.g., corrugated board, newsprint, high-grade paper, mixed) takes place in special recycling plants.
Recycled paper can be repulped in a relatively mild process which uses water and sometimes NaOH. Small metal pieces and plastics may be separated during and/or after repulping, using a debris rope, cyclones or centrifugation. Filling agents, glues and resins are removed in a cleaning stage by blowing air through the pulp slurry, sometimes with the addition of flocculating agents. The foam contains the unwanted chemicals and is removed. The pulp can be de-inked using a series of washing steps which may or may not include the use of chemicals (i.e., surfactant fatty acid derivatives) to dissolve remaining impurities, and bleaching agents to whiten the pulp. Bleaching has the disadvantage that it may reduce fibre length and therefore lessen final paper quality. The bleaching chemicals used in recycled pulp production are usually similar to those used in brightening operations for mechanical pulps. After the repulping and de-inking operations, sheet production follows in a manner very similar to that using virgin fibre pulp.
Sheet Production and Converting: Market Pulp, Paper, Paperboard
End products of pulp and paper mills depend on the pulping process, and may include market pulp and various types of paper or paperboard products. For example, the relatively weak mechanical pulp is converted into single-use products such as newspapers and tissue. Kraft pulp is converted into multi-use paper products such as high-quality writing paper, books and grocery bags. Sulphite pulp, which is primarily cellulose, can be used in a series of diverse end-products including specialty paper, rayon, photographic film, TNT, plastics, adhesives, and even ice cream and cake mixes. Chemi-mechanical pulps are exceptionally stiff, ideal for the structural support needed for corrugated container board. The fibres in pulp from recycled paper are usually shorter, less flexible and less water permeable, and can therefore not be used for high-quality paper products. Recycled paper is therefore mainly used for the production of soft paper products like tissue paper, toilet paper, paper towelling and napkins.
To produce market pulp, the pulp slurry is usually screened once more and its consistency adjusted (4 to 10%) before it is ready for the pulp machine. The pulp is then spread onto a travelling metal screen or plastic mesh (known as the “wire”) at the “wet end” of the pulp machine, where the operator monitors the speed of the moving wire and the water content of the pulp (figure 1; the presses and the cover of the drier can be seen in the upper left; in modern mills, operators spend a great deal of time in control rooms). Water and filtrate are drawn through the wire, leaving a web of fibres. The pulp sheet is passed through a series of rotating rolls (“presses”) that squeeze out water and air until the fibre consistency is 40 to 45%. The sheet is then floated through a multi-storey sequence of hot-air dryers until the consistency is 90 to 95%. Finally, the continuous pulp sheet is cut into pieces and stacked into bales. The pulp bales are compressed, wrapped and packaged into bundles for storage and transport.
Figure 1. Wet end of pulp machine showing fibre mat on the wire.
Canfor Library
Although similar in principle to making pulp sheets, paper making is considerably more complex. Some mills use a variety of different pulps to optimize paper quality (e.g., a mix of hardwood, softwood, kraft, sulphite, mechanical or recycled pulps). Depending on the type of pulp used, a series of steps is necessary prior to forming the paper sheet. Generally, dried market pulp is rehydrated, while high-consistency pulp from storage is diluted. Pulp fibres may be beaten to increase the fibre-bonding area and thereby improve paper sheet strength. The pulp is then blended with “wet-end” additives (table 1) and passed through a final set of screens and cleaners. The pulp is then ready for the paper machine.
Table 1. Papermaking additives
|
Additive |
Location applied |
Purpose and/or examples of specific agents |
|
Most commonly used additives |
||
|
Talc |
Wet end |
Pitch control (prevent deposition and accumulation |
|
Titanium dioxide |
Wet end |
Pigment (brighten sheet, improve printing) |
|
“Alum”(Al2(SO4)3) |
Wet end |
Precipitates rosin sizing onto fibres |
|
Rosin |
Wet end |
Internal sizing (resist liquid penetration) |
|
Clay (kaolin) |
Wet/dry |
Filler (make brighter, smoother, more opaque) |
|
Starch |
Wet/dry |
Surface sizing (resist liquid penetration) |
|
Dyes and |
Wet/dry |
e.g., acid, basic or direct dyes,colour lakes, |
|
Latex |
Dry end |
Adhesive (reinforce sheet, bind additives to paper, |
|
Other additives |
||
|
Slimicides |
Wet end |
e.g., thiones, thiazoles, thiocyanates, hiocarbamates, thiols, isothiazolinones, |
|
Defoamers |
Wet end |
e.g., pine oil, fuel oil, recycled oils, silicones, alcohols |
|
Wire treatment |
Wet end |
e.g., imidazoles, butyl diglycol, acetone, turpentine, |
|
Wet and dry |
Wet end |
e.g., formaldehyde resins, epichlorohydrin, glyoxal, |
|
Coatings, |
Dry end |
e.g., aluminium hydroxide, polyvinyl acetate, |
|
Others |
Wet/dry |
Corrosion inhibitors, dispersants, flameproofing, |
The flow spreader and headbox distribute a thin suspension (1 to 3%) of refined pulp onto a moving wire (similar to a pulp machine, only at a much higher speed, sometimes in excess of 55 km/h) which forms the fibres into a thin felted sheet. The sheet moves through a series of press rolls to the dryer section, where a series of steam-heated rolls evaporate most of the remaining water. Hydrogen bonds between the fibres have fully developed at this stage. Finally, the paper is calendered and reeled. Calendering is the process by which the paper surface is ironed smooth and its thickness reduced. The dried, calendered paper sheet is wound onto a reel, labelled and transported to the warehouse (figure 2; note waste paper under reel, and unenclosed operator control panel). “Dry-end” additives can be added before calendering on the paper machine or in separate “off-machine” coating operations in the converting sector of the industry.
Figure 2. Dry end of a paper machine showing full paper reel and operator using air slitter to cut end.
George Astrakianakis
A variety of chemicals are used in the papermaking process to provide the paper with specific surface characteristics and sheet properties. The most commonly used additives (table 1) are typically used at the per cent level, though some such as clay and talc may contribute as much as 40% to the dry weight of certain papers. Table 1 also indicates the diversity of chemical additives which may be used for specific production purposes and products; some of these are used at very low concentrations (e.g., slimicides are added to process water in parts per million).
The process of making paperboard is similar to that of making paper or pulp. A suspension of pulp and water is dispersed onto a travelling wire, the water is removed, and the sheet dried and stored as a roll. The process differs in the way that the sheet is formed to give thickness, in the combining of multiple layers, and in the drying process. Board can be made from single or multi-layered sheets with or without a core. The sheets are usually high-quality kraft pulp (or kraft and CTMP blend), while the core is made from either a blend of semi-chemical and low-cost recycled pulp or from entirely recycled pulp and other waste material. Coatings, vapour barriers and multiple layers are added according to the end use to protect the contents from water and physical damage.
Power Generation and Water Treatment
In addition to liquor recovery, pulp mills recover a significant portion of energy from burning waste materials and by-products of the process in power boilers. Materials such as bark, wood waste and dried sludge collected from effluent treatment systems may be burned to provide steam to power electrical generators.
Pulp and paper mills consume vast amounts of fresh water. A 1,000 tonne per day bleached kraft pulp mill may use more than 150 million litres of water a day; a paper mill even more. In order to prevent adverse effects on mill equipment and to maintain product quality, the incoming water must be treated to remove contaminants, bacteria and minerals. Several treatments are applied depending on the quality of the incoming water. Sedimentation beds, filters, flocculants, chlorine and ion exchange resins are all used to treat water before it is used in the process. Water that is used in the power and recovery boilers is further treated with oxygen scavengers and corrosion inhibitors such as hydrazine and morpholine to avoid deposits forming in the boiler tubes, to reduce metal corrosion, and to prevent carry-over of water to the steam turbine.
Chemical and By-product Production
Because many bleaching chemicals are reactive and hazardous to transport, they are produced on-site or nearby. Chlorine dioxide (ClO2), sodium hypochlorite (NaOCl) and peracids are always produced on-site, while chlorine (Cl2) and sodium hydroxide or caustic (NaOH) are usually produced off-site. Tall oil, a product derived from the resin and fatty acids that are extracted during kraft cooking, may be refined on- or off-site. Turpentine, a lighter fraction kraft by-product, is often collected and concentrated on-site, and refined elsewhere.
Chlorine Dioxide
Chlorine dioxide (ClO2) is a highly reactive greenish-yellow gas. It is toxic and corrosive, explodes at high concentrations (10%) and is quickly reduced to Cl2 and O2 in the presence of ultraviolet light. It must be prepared as a dilute gas and stored as a dilute liquid, making bulk transport impossible.
ClO2 is generated by reducing sodium chlorate (Na2ClO3) with either SO2, methanol, salt or hydrochloric acid. The gas leaving the reactor is condensed and stored as a 10% liquid solution. Modern ClO2 generators operate at 95% efficiency or greater, and the small amount of Cl2 that is produced will be collected or scrubbed out of the vent gas. Side reactions may occur depending on the purity of the feed chemicals, the temperature and other process variables. By-products are returned to the process and spent chemicals are neutralized and sewered.
Sodium Hypochlorite
Sodium hypochlorite (NaOCl) is produced by combining Cl2 with a dilute solution of NaOH. It is a simple, automated process that requires almost no intervention. The process is controlled by maintaining the caustic concentration such that the residual Cl2 in the process vessel is minimized.
Chlorine and Caustic
Chlorine (Cl2), used as a bleaching agent since the early 1800s, is a highly reactive, toxic, green-coloured gas which becomes corrosive when moisture is present. Chlorine is usually manufactured by the electrolysis of brine (NaCl) into Cl2 and NaOH at regional installations, and transported to the customer as a pure liquid. Three methods are used to produce Cl2 on an industrial scale: the mercury cell, the diaphragm cell, and the most recent development, the membrane cell. Cl2 is always produced at the anode. It is then cooled, purified, dried, liquefied and transported to the mill. At large or remote pulp mills, local facilities may be constructed, and the Cl2 can be transported as a gas.
The quality of NaOH depends on which of the three processes is used. In the older mercury cell method, the sodium and mercury combine to form an amalgam that is decomposed with water. The resulting NaOH is nearly pure. One of the shortcomings of this process is that mercury contaminates the workplace and has resulted in serious environmental problems. The NaOH produced from the diaphragm cell is removed with the spent brine and concentrated to allow the salt to crystallize and separate. Asbestos is used as the diaphragm. The purest NaOH is produced in membrane cells. A semi-permeable resin-based membrane allows sodium ions to pass through without the brine or chlorine ions, and combine with water added to the cathode chamber to form pure NaOH. Hydrogen gas is a by-product of each process. It is usually treated and used either in other processes or as fuel.
Tall Oil Production
Kraft pulping of highly-resinous species such as pine produces sodium soaps of resin and fatty acids. The soap is collected from black liquor storage tanks and from soap skimming tanks that are located in the evaporator train of the chemical recovery process. Refined soap or tall oil can be used as a fuel additive, dust control agent, road stabilizer, pavement binder and roofing flux.
At the processing plant, soap is stored in primary tanks to allow the black liquor to settle to the bottom. The soap rises and overflows into a second storage tank. Sulphuric acid and the decanted soap are fed into a reactor, heated to 100°C, agitated and then allowed to settle. After settling overnight, the crude tall oil is decanted into a storage vessel and allowed to sit for another day. The top fraction is considered dry crude tall oil and is pumped to storage, ready for shipment. The cooked lignin in the bottom fraction will become part of the subsequent batch. The spent sulphuric acid is pumped to a storage tank, and any entrained lignin is allowed to settle to the bottom. The lignin left in the reactor is concentrated for several cooks, dissolved in 20% caustic and returned to the primary soap tank. Periodically, the collected black liquor and the residual lignin from all sources are concentrated and burned as fuel.
Turpentine Recovery
Gases from the digesters and condensate from black liquor evaporators may be collected for recovery of turpentine. The gases are condensed, combined, then stripped of turpentine, which is recondensed, collected and sent to a decanter. The top fraction of the decanter is drawn off and sent to storage, while the bottom fraction is recycled to the stripper. Raw turpentine is stored separately from the rest of the collection system because it is noxious and flammable, and is usually processed off-site. All the non-condensable gases are collected and incinerated either in the power boilers, the lime kiln or a dedicated furnace. The turpentine can be processed for use in camphor, synthetic resins, solvents, flotation agents and insecticides.
Occupational Hazards and Controls
Table 1 provides an overview of the types of exposures which may be expected in each area of pulp and paper operations. Although exposures may be listed as specific to certain production processes, exposures to employees from other areas may also occur depending on weather conditions, proximity to sources of exposure, and whether they work in more than one process area (e.g., quality control, general labour pool and maintenance personnel).
Table 1. Potential health and safety hazards in pulp and paper production, by process area
|
Process area |
Safety hazards |
Physical hazards |
Chemical hazards |
Biological hazards |
|
Wood preparation |
||||
|
Log pond |
Drowning; mobile equipment; |
Noise; vibration; cold; heat |
Engine exhaust |
|
|
Wood room |
Nip points; slipping, falling |
Noise; vibration |
Terpenes and other wood extracts; wood dust |
Bacteria; fungi |
|
Chip screening |
Nip points; slipping, falling |
Noise; vibration |
Terpenes and other wood extracts; wood dust |
Bacteria; fungi |
|
Chip yard |
Nip points; mobile equipment |
Noise; vibration; cold; heat |
Engine exhaust; terpenes and other wood extracts; wood dust |
Bacteria; fungi |
|
Pulping |
||||
|
Stone groundwood |
Slipping, falling |
Noise; electric and magnetic fields; high humidity |
||
|
RMP, CMP, CTMP |
Slipping, falling |
Noise; electric and magnetic fields; high humidity |
Cooking chemicals and by-products; terpenes and other wood extracts; wood dust |
|
|
Sulphate pulping |
Slipping, falling |
Noise; high humidity; heat |
Acids and alkalis; cooking chemicals and by-products; reduced sulphur gases; terpenes |
|
|
Sulphate recovery |
Explosions; nip points; slipping, |
Noise; heat; steam |
Acids and alkalis; asbestos; ash; cooking chemicals and by-products; fuels; reduced |
|
|
Sulphite pulping |
Slipping, falling |
Noise; high humidity; heat |
Acids and alkalis; cooking chemicals and by-products; sulphur dioxide; terpenes and other wood extracts; wood dust |
|
|
Sulphite recovery |
Explosions; nip points; slipping, |
Noise; heat; steam |
Acids and alkalis; asbestos; ash; cooking chemicals and by-products; fuels; sulphur dioxide |
|
|
Repulping/de-inking |
Slipping, falling |
Acids and alkalis; bleaching chemicals and by- products; dyes and inks; pulp/paper dust; slimicides; solvents |
Bacteria |
|
|
Bleaching |
Slipping, falling |
Noise; high humidity; heat |
Bleaching chemicals and by-products; slimicides; terpenes and other wood extracts |
|
|
Sheet forming and |
||||
|
Pulp machine |
Nip points; slipping, falling |
Noise; vibration; high |
Acids and alkalis; bleaching chemicals and by-products; flocculant; pulp/paper dust; slimicides; solvents |
Bacteria |
|
Paper machine |
Nip points; slipping, falling |
Noise; vibration; high |
Acids and alkalis; bleaching chemicals and by-products; dyes and inks; flocculant; pulp/paper |
Bacteria |
|
Finishing |
Nip points; mobile equipment |
Noise |
Acids and alkalis; dyes and inks; flocculant; |
|
|
Warehouse |
Mobile equipment |
Fuels; engine exhaust; pulp/paper dust |
||
|
Other operations |
||||
|
Power generation |
Nip points; slipping, falling |
Noise; vibration; electric and |
Asbestos; ash; fuels; terpenes and other wood extracts; wood dust |
Bacteria; fungi |
|
Water treatment |
Drowning |
Bleaching chemicals and by-products |
Bacteria |
|
|
Effluent treatment |
Drowning |
Bleaching chemicals and by-products; flocculant; reduced sulphur gases |
Bacteria |
|
|
Chlorine dioxide |
Explosions; slipping, falling |
Bleaching chemicals and by-products |
Bacteria |
|
|
Turpentine recovery |
Slipping, falling |
Cooking chemicals and by-products; reduced sulphur gases; terpenes and other wood extracts |
||
|
Tall oil production |
Acids and alkalis; cooking chemicals and by-products; reduced sulphur gases; terpenes and other wood extracts |
RMP = refining mechanical pulping; CMP = chemi-mechanical pulping; CTMP = chemi-thermomechanical pulping.
Exposure to the potential hazards listed in table 1 is likely to depend on the extent of automation of the plant. Historically, industrial pulp and paper production was a semi-automatic process which required a great deal of manual intervention. In such facilities, operators would sit at open panels adjacent to the processes to view the effects of their actions. The valves at the top and bottom of a batch digester would be manually opened, and during the filling stages, gases in the digester would be displaced by the incoming chips (figure 1). Chemical levels would be adjusted based on experience rather than sampling, and process adjustments would be dependent on the skill and knowledge of the operator, which at times led to upsets. For example, over-chlorination of pulp would expose workers downstream to increased levels of bleaching agents. In most modern mills, progress from manually controlled to electronically controlled pumps and valves allows for remote operation. The demand for process control within narrow tolerances has required computers and sophisticated engineering strategies. Separate control rooms are used to isolate the electronic equipment from the pulp and paper production environment. Consequently, operators usually work in air-conditioned control rooms which offer refuge from the noise, vibration, temperature, humidity and chemical exposures inherent to mill operations. Other controls which have improved the working environment are described below.
Figure 1. Worker opening cap on manually controlled batch digester.
MacMillan Bloedel archives
Safety hazards including nip points, wet walking surfaces, moving equipment and heights are common throughout pulp and paper operations. Guards around moving conveyors and machinery parts, quick clean-up of spills, walking surfaces which allow drainage, and guard-rails on walkways adjacent to production lines or at height are all essential. Lock-out procedures must be followed for maintenance of chip conveyors, paper machine rolls and all other machinery with moving parts. Mobile equipment used in chip storage, dock and shipping areas, warehousing and other operations should have roll-over protection, good visibility and horns; traffic lanes for vehicles and pedestrians should be clearly marked and signed.
Noise and heat are also ubiquitous hazards. The major engineering control is operator enclosures, as described above, usually available in wood preparation, pulping, bleaching and sheet-forming areas. Air-conditioned enclosed cabs for mobile equipment used in chip pile and other yard operations are also available. Outside these enclosures, workers usually require hearing protection. Work in hot process or outdoor areas and in vessel maintenance operations requires workers to be trained to recognize symptoms of heat stress; in such areas, work scheduling should allow acclimatization and rest periods. Cold weather may create frostbite hazards in outdoor jobs, as well as foggy conditions near chip piles, which remain warm.
Wood, its extracts and associated micro-organisms are specific to wood preparation operations and the initial stages of pulping. Control of exposures will depend on the particular operation, and may include operator booths, enclosure and ventilation of saws and conveyors, as well as enclosed chip storage and low chip inventory. Use of compressed air to clear wood dust creates high exposures and should be avoided.
Chemical pulping operations present the opportunity for exposures to digestion chemicals as well as gaseous by-products of the cooking process, including reduced (kraft pulping) and oxidized (sulphite pulping) sulphur compounds and volatile organics. Gas formation may be influenced by a number of operating conditions: the wood species used; the quantity of wood pulped; the amount and concentration of white liquor applied; the amount of time required for pulping; and maximum temperature attained. In addition to automatic digester capping valves and operator control rooms, other controls for these areas include local exhaust ventilation at batch digesters and blow tanks, capable of venting at the rate the vessel’s gases are released; negative pressure in recovery boilers and sulphite-SO2 acid towers to prevent gas leaks; ventilated full or partial enclosures over post-digestion washers; continuous gas monitors with alarms where leaks may occur; and emergency response planning and training. Operators taking samples and conducting tests should be aware of the potential for acid and caustic exposure in process and waste streams, and the possibility of side reactions such as hydrogen sulphide gas (H2S) production if black liquor from kraft pulping comes into contact with acids (e.g., in sewers).
In chemical recovery areas, acidic and alkaline process chemicals and their by-products may be present at temperatures in excess of 800°C. Job responsibilities may require workers to come into direct contact with these chemicals, making heavy duty clothing a necessity. For example, workers rake the spattering molten smelt that collects at the base of the boilers, thereby risking chemical and thermal burns. Workers may be exposed to dust when sodium sulphate is added to concentrated black liquor, and any leak or opening will release noxious (and potentially fatal) reduced sulphur gases. The potential for a smelt water explosion always exists around the recovery boiler. Water leaks in the tube walls of the boiler have resulted in several fatal explosions. Recovery boilers should be shut down at any indication of a leak, and special procedures should be implemented for transferring the smelt. Loading of lime and other caustic materials should be done with enclosed and ventilated conveyors, elevators and storage bins.
In bleach plants, field operators may be exposed to the bleaching agents as well as chlorinated organics and other by-products. Process variables such as bleaching chemical strength, lignin content, temperature and pulp consistency are constantly monitored, with operators collecting samples and performing laboratory tests. Because of the hazards of many of the bleaching agents used, continuous alarm monitors should be in place, escape respirators should be issued to all employees, and operators should be trained in emergency response procedures. Canopy enclosures with dedicated exhaust ventilation are standard engineering controls found at the top of each bleaching tower and washing stage.
Chemical exposures in the machine room of a pulp or paper mill include chemical carry-over from the bleach plant, the papermaking additives and the chemical mixture in the waste water. Dusts (cellulose, fillers, coatings) and exhaust fumes from mobile equipment are present in the dry-end and the finishing operations. Cleaning between product runs may be done with solvents, acids and alkalis. Controls in this area may include complete enclosure over the sheet drier; ventilated enclosure of the areas where additives are unloaded, weighed and mixed; use of additives in liquid rather than powder form; use of water-based rather than solvent-based inks and dyes; and eliminating the use of compressed air to clean up trimmed and waste paper.
Paper production in recycled paper plants is generally dustier than conventional paper production using newly produced pulp. Exposure to micro-organisms can occur from the beginning (paper collection and separation) to the end (paper production) of the production chain, but exposure to chemicals is less important than in conventional paper production.
Pulp and paper mills employ an extensive maintenance group to service their process equipment, including carpenters, electricians, instrument mechanics, insulators, machinists, masons, mechanics, millwrights, painters, pipefitters, refrigeration mechanics, tinsmiths and welders. Along with their trade-specific exposures (see the Metal processing and metal working and Occupations chapters), these tradespeople may be exposed to any of the process-related hazards. As mill operations have become more automated and enclosed, the maintenance, cleaning and quality assurance operations have become the most highly exposed. Plant shutdowns to clean vessels and machines are of special concern. Depending on mill organization, these operations may be carried out by in-house maintenance or production personnel, although subcontracting to non-mill personnel, who may have less occupational health and safety support services, is common.
In addition to process exposures, pulp and paper mill operations entail some noteworthy exposures for maintenance personnel. Because pulping, recovery and boiler operations involve high heat, asbestos was used extensively to insulate pipes and vessels. Stainless steel is often used in vessels and pipes throughout pulping, recovery and bleaching operations, and to some extent in papermaking. Welding this metal is known to generate chromium and nickel fumes. During maintenance shut-downs, chromium-based sprays may be applied to protect the floor and walls of recovery boilers from corrosion during start-up operations. Process quality measurements in the production line are often made using infrared and radio-isotope gauges. Although the gauges are usually well shielded, instrument mechanics who service them may be exposed to radiation.
Some special exposures may also occur among employees in other mill-support operations. Power boiler workers handle bark, waste wood and sludge from the effluent treatment system. In older mills, workers remove ash from the bottom of the boilers and then reseal the boilers by applying a mixture of asbestos and cement around the boiler grate. In modern power boilers, this process is automated. When material is fed into the boiler at too high a moisture level, workers may be exposed to blow-backs of incomplete combustion products. Workers responsible for water treatment may be exposed to chemicals such as chlorine, hydrazine and various resins. Because of the reactivity of ClO2, the ClO2 generator is usually located in a restricted area and the operator is stationed in a remote control room with excursions to collect samples and service the saltcake filter. Sodium chlorate (a strong oxidizer) used to generate ClO2 can become dangerously flammable if it is allowed to spill on any organic or combustible material and then dry. All spills should be wetted down before any maintenance work may proceed, and all equipment should be thoroughly cleaned afterward. Wet clothing should be kept wet and separate from street clothing, until washed.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."