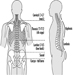
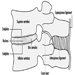
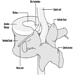
Children categories

1. Blood (3)

1. Blood
Chapter Editor: Bernard D. Goldstein
Table of Contents
Tables
Haematopoietic and Lymphatic System
Bernard D. Goldstein
Leukaemia, Malignant Lymphomas and Multiple Myeloma
Timo Partanen, Paolo Boffetta, Elisabete Weiderpass
Agents or Work Conditions Affecting the Blood
Bernard D. Goldstein
Tables
Click a link below to view the table in the article context.

2. Cancer (4)

2. Cancer
Chapter Editor: Paolo Boffetta
Table of Contents
Tables
Introduction
Neil Pearce, Paolo Boffetta and Manolis Kogevinas
Occupational Carcinogens
Paolo Boffetta, Rodolfo Saracci, Manolis Kogevinas, Julian Wilbourn and Harri Vainio
Environmental Cancer
Bruce K. Armstrong and Paolo Boffetta
Prevention
Per Gustavsson
Tables
Click a link below to view table in article context.
- Occupational cancer: Key facts
- Estimated proportions of cancer (PAR) attributable to occupations
- Evaluation of evidence of carcinogenicity in the IARC Monographs
- IARC Monograph programme classification groups
- Group 1-Chemicals carcinogenic to humans
- Group 2A—Chemicals probably carcinogenic to humans
- Group 2B—Chemicals possibly carcinogenic to humans
- Pesticides evaluated in IARC Monographs, Volumes 1-63 (1972-1995)
- Drugs evaluated in IARC Monographs, Volumes 1-63 (1972-1995)
- Environmental agents/exposures known or suspected of human cancer
- Industries, occupations, exposures presenting a carcinogenic risk
- Industries, occs., exps. with cancer excess not definitive carcinogens
- Registered population variations of incidence of some common cancers

3. Cardiovascular System (7)

3. Cardiovascular System
Chapter Editors: Lothar Heinemann and Gerd Heuchert
Table of Contents
Tables and Figures
Introduction
Lothar Heinemann and Gerd Heuchert
Cardiovascular Morbidity and Mortality in the Workforce
Gottfried Enderlein and Lothar Heinemann
The Risk Factor Concept in Cardiovascular Disease
Lothar Heinemann, Gottfried Enderlein and Heide Stark
Rehabilitation and Prevention Programmes
Lothar Heinemann and Gottfried Enderlein
Physical, Chemical and Biological Hazards
Physical Factors
Heide Stark and Gerd Heuchert
Chemical Hazardous Materials
Ulrike Tittelbach and Wolfram Dietmar Schneider
Biological Hazards
Regina Jäckel, Ulrike Tittelbach and Wolfram Dietmar Schneider
Tables
Click a link below to view table in article context
- Mortality from cardiovascular diseases
- Mortality rates, special cardiovascular diagnosis groups
- Rate of disease and reduced work ability
- Work associated with cardiovascular hazards
- Occupation-related infection and disease
Figures
Point to a thumbnail to see figure caption, click to see the figure in the article context.

4. Digestive System (6)

4. Digestive System
Chapter Editor: Heikki Savolainen
Table of Contents
Figures
Digestive system
G. Frada
Mouth and teeth
F. Gobbato
Liver
George Kazantzis
Peptic ulcer
K. S. Cho
Liver cancer
Timo Partanen, Timo Kauppinen, Paolo Boffetta and Elisabete Weiderpass
Pancreatic cancer
Timo Partanen, Timo Kauppinen, Paolo Boffetta and Elisabete Weiderpass
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

5. Mental Health (8)

5. Mental Health
Chapter Editors: Joseph J. Hurrell, Lawrence R. Murphy, Steven L. Sauter and Lennart Levi
Table of Contents
Tables and Figures
Work and Mental Health
Irene L.D. Houtman and Michiel A.J. Kompier
Work-related Psychosis
Craig Stenberg, Judith Holder and Krishna Tallur
Mood and Affect
Depression
Jay Lasser and Jeffrey P. Kahn
Work-related Anxiety
Randal D. Beaton
Post-traumatic Stress Disorder and its Relationship to Occupational Health and Injury Prevention
Mark Braverman
Stress and Burnout and their Implication in the Work Environment
Herbert J. Freudenberger
Cognitive Disorders
Catherine A. Heaney
Karoshi: Death from Overwork
Takashi Haratani
Tables
Click a link below to view table in article context.
1. Schematic overview of management strategies & examples
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

6. Musculoskeletal System (14)

6. Musculoskeletal System
Chapter Editors: Hilkka Riihimäki and Eira Viikari-Juntura
Table of Contents
Tables and Figures
Overview
Hilkka Riihimäki
Muscles
Gisela Sjøgaard
Tendons
Thomas J. Armstrong
Bones and Joints
David Hamerman
Intervertebral Discs
Sally Roberts and Jill P.G. Urban
Low-back Region
Hilkka Riihimäki
Thoracic Spine Region
Jarl-Erik Michelsson
Neck
Åsa Kilbom
Shoulder
Mats Hagberg
Elbow
Eira Viikari-Juntura
Forearm, Wrist and Hand
Eira Viikari-Juntura
Hip and Knee
Eva Vingård
Leg, Ankle and Foot
Jarl-Erik Michelsson
Other Diseases
Marjatta Leirisalo-Repo
Tables
Click a link below to view table in article context.
- Structure-function of joint components
- Prevalence of back disorders, in Finns over 30 years
- Reducing the risks for low-back pain at work
- Classification-low-back disorders (Quebec Task Force)
- Permissible motions for head in prolonged driving
- Incidence of epicondylitis in various populations
- Incidence of tenosynovitis/peritendinitis
- Primary osteoarthrosis of the hip in Malmö, Sweden
- Guidelines for the treatment of rheumatoid arthritis
- Infections known to trigger reactive arthritis
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

7. Nervous System (9)

7. Nervous System
Chapter Editor: Donna Mergler
Table of Contents
Tables and Figures
Nervous System: Overview
Donna Mergler and José A. Valciukas
Anatomy and Physiology
José A. Valciukas
Chemical Neurotoxic Agents
Peter Arlien-Søborg and Leif Simonsen
Manifestations of Acute and Early Chronic Poisoning
Donna Mergler
Preventing Neurotoxicity at Work
Barry Johnson
Clinical Syndromes Associated with Neurotoxicity
Robert G. Feldman
Measuring Neurotoxic Deficits
Donna Mergler
Diagnosis
Anna Maria Seppäläinen
Occupational Neuroepidemiology
Olav Axelson
Tables
Click a link below to view table in article context.
- Names & main functions of each pair of cranial nerves
- Grouping neurotoxic effects as to neurotoxicity
- Gases associated with neurotoxic effects
- Neurotoxic metals & their inorganic compounds
- Neurotoxic monomers
- Organic solvents associated with neurotoxicity
- Classes of common neurotoxic pesticides
- Other chemicals associated with neurotoxicity
- Chronic symptoms checklist
- Neuro-functional effects of exposures to some neurotoxins
- Chemical exposures & associated neurotoxic syndromes
- Some “core” batteries for assessing early neurotoxic effects
- Decision tree for neurotoxic disease
- Consistent neuro-functional effects of worksite exposures to some leading neurotoxic substances
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

8. Renal-Urinary System (2)

8. Renal-Urinary System
Chapter Editor: George P. Hemstreet
Table of Contents
Tables and Figures
Renal-Urinary Systems
George P. Hemstreet
Renal-Urinary Cancers
Timo Partanen, Harri Vainio, Paolo Boffetta and Elisabete Weiderpass
Tables
Click a link below to view table in article context.
- Drug-metabolism enzymes in kidney
- The most common causes of haematuria, by age & sex
- Criteria for biomarker selection
- Potential biomarkers linked to cell injury
- Acute renal insufficiency & occupation
- Segments of the nephron affected by selected toxicants
- Applications of urinary cytology
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

9. Reproductive System (9)

9. Reproductive System
Chapter Editor: Grace Kawas Lemasters
Table of Contents
Tables and Figures
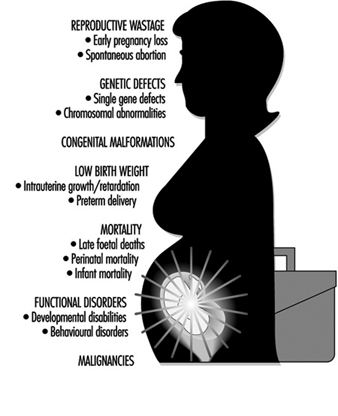
Reproductive System: Introduction
Lowell E. Sever
Introduction to Male and Female Reproductive Function
Donald R. Mattison
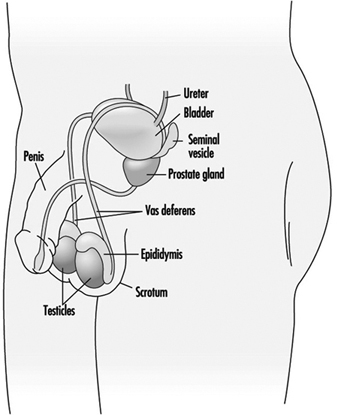
Male Reproductive System and Toxicology
Steven Schrader and Grace Kawas Lemasters
Structure of the Female Reproductive System and Target Organ Vulnerability
Donald R. Mattison
Maternal Occupational Exposures and Adverse Pregnancy Outcomes
Grace Kawas Lemasters
Preterm Delivery and Work
Nicole Mamelle
Occupational and Environmental Exposures to the Newborn
Mary S. Wolff and Patrisha M. Woolard
Maternity Protection in Legislation
Marie-Claire Séguret
Pregnancy and US Work Recommendations
Leon J. Warshaw
Tables
Click a link below to view table in article context.
1. Exposures with multiple adverse endpoints
2. Epidemiological studies of paternal effects on pregnancy outcome
3. Potential female reproductive toxicants
4. Definition of foetal loss & infant death
5. Factors for small for gestational age and foetal loss
6. Identified sources of occupational fatigue
7. Relative risks & fatigue indices for preterm delivery
8. Prematurity risk by number of occupational fatigue indices
9. Relative risks and changes in working conditions
10. Newborn exposure sources and levels
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

10. Respiratory System (18)

10. Respiratory System
Chapters Editors: Alois David and Gregory R. Wagner
Table of Contents
Tables and Figures
Structure and Function
Morton Lippmann
Lung Function Examination
Ulf Ulfvarson and Monica Dahlqvist
Diseases Caused by Respiratory Irritants and Toxic Chemicals
David L.S. Ryon and William N. Rom
Occupational Asthma
George Friedman-Jimenez and Edward L. Petsonk
Diseases Caused by Organic Dusts
Ragnar Rylander and Richard S. F. Schilling
Beryllium Disease
Homayoun Kazemi
Pneumoconioses: Definition
Alois David
ILO International Classification of Radiographs of Pneumoconioses
Michel Lesage
Aetiopathogenesis of Pneumoconioses
Patrick Sébastien and Raymond Bégin
Silicosis
John E. Parker and Gregory R. Wagner
Coal Workers’ Lung Diseases
Michael D. Attfield, Edward L. Petsonk and Gregory R. Wagner
Asbestos-Related Diseases
Margaret R. Becklake
Hard Metal Disease
Gerolamo Chiappino
Respiratory System: The Variety of Pneumoconioses
Steven R. Short and Edward L. Petsonk
Chronic Obstructive Pulmonary Disease
Kazimierz Marek and Jan E. Zejda
Health Effects of Man-Made Fibres
James E. Lockey and Clara S. Ross
Respiratory Cancer
Paolo Boffetta and Elisabete Weiderpass
Occupationally Acquired Infections of the Lung
Anthony A. Marfin, Ann F. Hubbs, Karl J. Musgrave, and John E. Parker
Tables
Click a link below to view table in article context.
1. Respiratory tract regions & particle deposition models
2. Inhalable, thoracic & respirable dust criteria
3. Summary of respiratory irritants
4. Mechanisms of lung injury by inhaled substances
5. Compounds capable of lung toxicity
6. Medical case definition of occupational asthma
7. Steps in diagnostic evaluation of asthma in the workplace
8. Sensitizing agents that can cause occupational asthma
9. Examples of sources of hazards of exposure to organic dust
10. Agents in organic dusts with potential biological activity
11. Diseases induced by organic dusts & their ICD codes
12. Diagnostic criteria for byssinosis
13. Properties of beryllium & its compounds
14. Description of standard radiographs
15. ILO 1980 Classification: Radiographs of Pneumoconioses
16. Asbestos-related diseases & conditions
17. Main commercial sources, products & uses of asbestos
18. Prevalence of COPD
19. Risk factors implicated in COPD
20. Loss of ventilatory function
21. Diagnostic classification, chronic bronchitis & emphysema
22. Lung function testing in COPD
23. Synthetic fibres
24. Established human respiratory carcinogens (IARC)
25. Probable human respiratory carcinogens (IARC)
26. Occupationally acquired respiratory infectious diseases
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

11. Sensory Systems (8)

11. Sensory Systems
Chapter Editor: Heikki Savolainen
Table of Contents
Tables and Figures
The Ear
Marcel-André Boillat
Chemically-Induced Hearing Disorders
Peter Jacobsen
Physically-Induced Hearing Disorders
Peter L. Pelmear
Equilibrium
Lucy Yardley
Vision and Work
Paule Rey and Jean-Jacques Meyer
Taste
April E. Mott and Norman Mann
Smell
April E. Mott
Cutaneous Receptors
Robert Dykes and Daniel McBain
Tables
Click a link below to view table in article context.
1. Typical calculation of functional loss from an audiogram
2. Visual requirements for different activities
3. Recommended illuminance values for the lighting design
4. Visual requirements for a driving licence in France
5. Agents/processes reported to alter the taste system
6. Agents/processes associated with olfactory abnormalities
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

12. Skin Diseases (7)

12. Skin Diseases
Chapter Editor: Louis-Philippe Durocher
Table of Contents
Tables and Figures
Overview: Occupational Skin Diseases
Donald J. Birmingham
Non-Melanocytic Skin Cancer
Elisabete Weiderpass, Timo Partanen, Paolo Boffetta
Malignant Melanoma
Timo Partanen, Paolo Boffetta, Elisabete Weiderpass
Occupational Contact Dermatitis
Denis Sasseville
Prevention of Occupational Dermatoses
Louis-Phillipe Durocher
Occupational Nail Dystrophy
C.D. Calnan
Stigmata
H. Mierzecki
Tables
Click a link below to view table in article context.
1. Occupations at risk
2. Types of contact dermatitis
3. Common irritants
4. Common skin allergens
5. Predisposing factors for occupational dermatitis
6. Examples of skin irritants & sensitizers with occupations
7. Occupational dermatoses in Quebec in 1989
8. Risk factors & their effects on the skin
9. Collective measures (group approach) to prevention
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.

13. Systemic Conditions (3)

13. Systemic Conditions
Chapter Editor: Howard M. Kipen
Table of Contents
Figures
Systemic Conditions: An Introduction
Howard M. Kipen
Sick Building Syndrome
Michael J. Hodgson
Multiple Chemical Sensitivities
Mark R. Cullen
Figures
Point to a thumbnail to see figure caption, click to see figure in article context.
Nervous System: Overview
Knowledge of the nervous system in general and of the brain and human behaviour in particular are of paramount importance to those who are dedicated to a safe and healthy environment. Work conditions, and exposures that directly affect the operations of the brain, influence the mind and behaviour. To evaluate information, to make decisions and to react in a consistent and reasonable manner to perceptions of the world require that the nervous system functions properly and that behaviour not be damaged by dangerous conditions, such as accidents (e.g., a fall from a poorly designed ladder) or exposure to hazardous levels of neurotoxic chemicals.
Damage to the nervous system can cause changes in sensory input (loss of vision, hearing, smell, etc.), can hinder the capacity to control movement and body functions and/or can affect the brain’s capacity to treat or store information. In addition, altered nervous system functioning can cause behavioural or psychological disorders. Mood and personality changes are a common occurrence following physical or organic damage to the brain. As our knowledge develops, we are learning more about the way in which nervous system processes are modified. Neurotoxic substances can cross the brain’s natural barrier and directly interfere with its intricate workings. Although some substances have a particular affinity to certain areas of the nervous system, most neurotoxins have widespread effects, targeting cell processes involved in membrane transport, internal cellular chemical reactions, liberation of secretory substances, and so on.
Damage to the various components of the nervous system can occur in different ways:
- direct physical injury from falling objects, collisions, blows or undue pressure on nerves
- changes in the internal environment, such as insufficient oxygen due to asphyxiants and heat exposure
- interference in the cellular processes through chemical action by substances, such as metals, organic solvents and pesticides
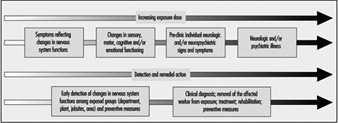
The insidious and multifaceted development of many nervous system disorders requires persons working in the field of occupational health to adopt different but complementary approaches to the study, understanding, prevention and treatment of the problem. Early alterations can be detected in groups of active, exposed workers using sensitive measures of impairment. Identification of initial dysfunction can lead to preventive actions. In the latter stages, a good clinical knowledge is required and differential diagnosis is essential to the adequate treatment and care of disabled workers.
Although chemical substances are mostly examined one by one, it should be remembered that in many workplaces mixtures of potentially neurotoxic chemicals are used, exposing workers to what can be called a “cocktail”. In processes such as printing, painting, cleaning, in poorly ventilated offices, in laboratories, pesticide application, microelectronics and many other sectors, workers are exposed to chemical mixtures. Although there may be information on each one of the substances separately, we have to consider the combined nocivity and possible additive or even synergistic effects on the nervous system. In some cases of multiple exposure, each particular chemical may be present in very small quantity, even below the detection level of exposure assessment techniques; however, when all are added together, the total concentration can be very high.
The reader should be aware of three major difficulties in reviewing facts about the nervous system within the scope of this Encyclopaedia.
First, the understanding of occupational diseases affecting the nervous system and behaviour has changed substantially as new approaches to viewing brain-behavioural relationships have developed. The main interest of characterization of gross morphological changes that occur due to mechanical trauma to the nervous system—particularly, but not exclusively to the brain—was followed by an interest in the absorption of neurotoxic agents by the nervous system; interest in the study of cellular mechanisms of nervous system pathology; and finally, the search for the molecular basis of these pathologic processes began to grow. These approaches coexist today and all contribute information for evaluating the working conditions affecting the brain, mind, and behaviour.
Second, the information generated by neuroscientists is staggering. The third edition of the book Principles of Neural Sciences edited by Kandel, Schwartz and Kessell which appeared in 1991—one of the most valuable reviews of the field—weighs 3.5 kg and is more than 1,000 pages long.
Third, it is very difficult to review knowledge about the functional organization of the nervous system as it applies to all niches of occupational health and safety. Until about 25 years ago, the theoretical views that gave support to those concerned health experts who specialize in the detection, monitoring, prevention, and the clinical treatment of a worker who has absorbed a neurotoxic agent sometimes did not overlap with theoretical views regarding workers’ brain trauma and the behavioural manifestations of minimal brain damage. Behavioural manifestations said to be the consequence of the disruption of specific chemical pathways in the brain were the exclusive territory of the neurotoxicologist; both structural tissue damage of specific regions of the brain, and distant neural structures linked to the area where the lesions occurred, were explanations invoked by neurologists. It is only in the past few years that converging views are appearing.
With this in mind, this chapter addresses issues important to the understanding of the nervous system and the effects of workplace conditions on its functioning. It begins with a description of the anatomy and physiology, followed by a section on neurotoxicity, which reviews exposure, outcomes and prevention.
Since the nervous system is central to the body’s well-being, many non-chemical hazards can likewise affect its normal functioning. Many of these are considered in different chapters dealing with these hazards. Traumatic head injuries are included in First Aid, heat stress is considered in the article “Effects of heat stress and work in the heat”, and decompression sickness is reviewed in the article “Gravitational stress”. Hand-arm vibration (“Hand-transmitted vibration”) and repetitive movement (“Chronic outcomes, musculoskeletal”) in the chapter Musculoskeletal System, which are risk factors for peripheral neuropathies, are likewise considered in these sections of the Encyclopaedia.
The chapter ends with a review of special issues and the outlook for future research avenues.
Anatomy and Physiology
Nerve cells are the functional units of the nervous system. The nervous system is believed to have ten thousand million of such cells, called neurons and glia, the glia being present in greater numbers than neurons.
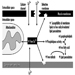
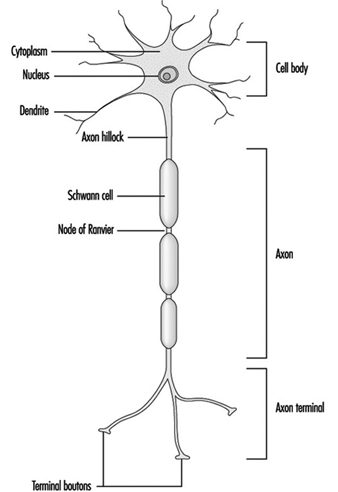
The Neuron
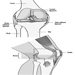
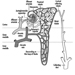
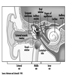
Figure 1 is an idealized diagram of a neuron with its three most important structural features: the cell body, the dendrites and the axon terminal.
Figure 1. The anatomy of the neuron
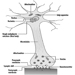
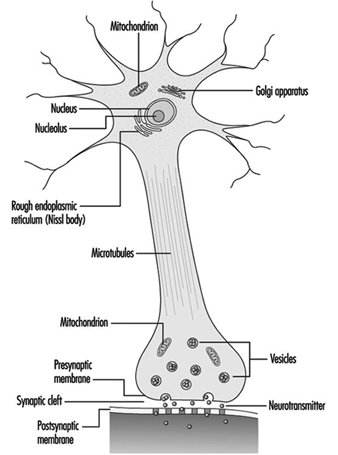
The dendrites are finely branched processes arising near the cell body of a neuron. The dendrites receive excitatory or inhibitory effects via chemical messengers called neurotransmitters. The cytoplasm is the material of the cell body in which the organelles—including the cell nucleus—and other inclusions are found Figure 2. The nucleus contains the cell’s chromatin, or genetic material.
Figure 2. The organelles
The nucleus of the nerve cell is atypical compared with that of other living cells in that, although it contains the genetic material deoxyribonucleic acid (DNA), the DNA is not involved in the process of cell division; that is, after reaching maturity, nerve cells do not divide. (An exception to this rule are the neurons in the nose lining (olfactory epithelium).) The nucleus is rich in ribonucleic acid (RNA), which is necessary for the synthesis of protein. Three types of proteins have been identified: cytosolic proteins, which form the fibrillar elements of the nerve cell; intracondrial proteins, which generate energy for cell activity; and proteins that form membranes and secretory products. Neurons are now conceived of as modified secretory cells. Secretory granules are formed, stored in synaptic vesicles and later released as neurotransmitter substances, the chemical messengers between nerve cells.
The fibrillar elements, which form the skeleton of the neuron, participate in the trophic function of the neuron, acting as vehicles of transmission. Axonal transport can be anterograde (cell body to axon terminal) and retrograde (axon terminal to cell body). From the thickest to the thinnest, three types of fibrillar elements are recognized: microtubules, neurofilaments and microfilaments.
Glial Cells
In contrast to neurons, glial cells do not, by themselves, carry electrical messages. There are two types of glial cells: the macroglia and the microglia. The macroglia is a name given to at least three types of cells: astrocytes, oligodendrocytes and ependymal cells. Microglial cells are primarily scavenger cells for removing debris after neural damage or infection has occurred.
The glial cells also have distinctive microscopic and ultramicroscopic features. Glial cells physically support neurons, but a number of physiological properties are also now beginning to be understood. Among the most important neuron-glial interactions are the glial cell’s role in providing the neurons with nutrients, removing fragments of neurons after their death and, most importantly, contributing to the process of chemical communication. Glial cells, in sharp contrast to neurons, can divide and thus can reproduce themselves. Tumours of the nervous system, for example, result from an abnormal reproduction of glial cells.
Myelin
What appears in the macroscopic observation of neural tissue as “grey matter” and “white matter” has a microscopic and biochemical basis. Microscopically, the grey matter contains the neuronal cell bodies, whereas the white matter is where neural fibres or axons are found. The “white” appearance is due to a sheath—composed of a fatty substance called myelin—covering these fibres. Myelin of the peripheral nerves originates from the membrane of the Schwann cell which wraps around the axon. The myelin of fibres in the central nervous system is provided by the membranes of the oligodendrocytes (a variety of glial cells). Oligodendrocytes usually myelinate several axons, whereas the Schwann cell is associated with only one axon. A discontinuity of the myelin sheath—designated as nodes of Ranvier—exists between continuous Schwann cells or oligodendrocytes. It is estimated that in the longest central motor pathway, up to 2,000 Schwann cells form the myelin cover. Myelin, whose role is to facilitate the propagation of the action potential, may be a specific target of neurotoxic agents. A morphological classification of neurotoxic substances describes characteristic neuropathological changes of the myelin as myelinopathies.
Trophic Function of the Neuron
The normal functions of the neuron include protein synthesis, axonal transport, generation and conduction of the action potential, synaptic transmission, and formation and maintenance of the myelin. Some of the basic trophic functions of the neuron were described as early as the 19th century by sectioning the axons (axotomy). Among the processes uncovered, one of the most important was the Wallerian degeneration—after Waller, the English physiologist who described it.
Wallerian degeneration provides a good opportunity to describe well-known changes in organelles as a result of either traumatic or toxic damage. Parenthetically, the terms used to describe Wallerian degeneration produced by traumatic axotomy are the same ones used to describe changes resulting from neurotoxic agents. At the cellular level, neuropathological changes resulting from toxic damage to neural tissue are far more complex than those occurring as a result of traumatic damage. It is only recently that changes in neurons affected by neurotoxic agents have been observed.
Twenty-four hours after cutting of the axon, the most distinctive feature is swelling of both sides of the mechanical trauma. Swelling results from accumulation of fluids and membranous elements on both sides of the site of injury. These changes are not unlike those observed in a rain-flooded two-way road with vehicles stopped on both sides of the flooded area. In this analogy, stalled vehicles are the swelling. After a few days, regeneration of the ensheathed axons—i.e., those covered with myelin—occurs. Sprouts grow from the proximal stump moving at the rate of 1 to 3 mm per day. Under favourable conditions, sprouts reach the distal (farther from the cell body) stump. When renervation—joining of the stumps—is completed, the basic features of normal transmission have been re-established. The cell body of the injured neuron undergoes profound structural changes in protein synthesis and axonal transport.
If molecular neurobiology is said to be a young discipline, the neurobiology of the neurotoxic processes is even younger, and still in its infancy. True, the molecular basis of action of many neurotoxins and pharmacological agents is now well understood. But with some notable exceptions (e.g., lead, methyl mercury, acrylamide) the molecular basis of toxicity of the vast majority of environmental and neurotoxic agents is unknown. That is why, instead of describing the molecular neurobiology of a select group of occupational and environmental neurotoxic agents, we still are forced to refer to the comparatively abundant strategies and examples from classical neuropharmacology or from work in modern drug manufacture.
Neurotransmitters
A neurotransmitter is a chemical substance which, when released from axon terminals by the action potential, produces the momentary change in electrical potential when another nerve fibre is stimulated. Neurotransmitters stimulate or inhibit adjacent neurons or effector organs such as muscle and glands. Known neurotransmitters and their neural pathways are now being intensively studied, and new ones are constantly being discovered. Some neurological and psychiatric disorders are now understood to be caused by chemical changes in neurotransmission—for example, myasthenia gravis, Parkinson’s disease, certain forms of affective disorders such as depression, severe distortion of thought processes such as in schizophrenia, and Alzheimer’s disease. Although excellent isolated reports on the effect of several environmental and occupational neurotoxic agents on neurotransmission have been published, the body of knowledge is meagre compared with that existing for neuropsychiatric diseases. Pharmacological studies of manufactured drugs require an understanding of how drugs affect neurotransmission. Drug manufacture and neurotransmission research are thus intimately related. The changing views of drug action have been summarized by Feldman and Quenzer (1984).
The effects of neurotoxic agents on neurotransmission are characterized by where in the nervous system they act, their chemical receptors, the time course of their effects, whether neurotoxic agents facilitate, block or inhibit neurotransmission, or whether neurotoxic agents alter the termination or removal of the neurotransmitter’s pharmacological action.
One difficulty experienced by neuroscientists is the need to link known processes that occur at the molecular level in the neuron with events at the cellular level, which in turn may explain how normal and pathological neuropsychological changes occur, as clearly stated in the following which to a large extent still applies: “(A)t the molecular level, an explanation of the action of a drug is often possible; at the cellular level, an explanation is sometimes possible, but at a behavioural level, our ignorance is abysmal” (Cooper, Bloom and Roth 1986).
The Main Components of the Nervous System
Knowledge of the main components of the nervous system is essential for the understanding of the gross neuropsychological manifestations of neurotoxic illness, the rationale for the use of specific techniques for the assessment of nervous system functions, and the understanding of pharmacological mechanisms of neurotoxic action. From a functional standpoint, the nervous system can be divided into two major compartments: The somatic nervous system conveys sensory information (touch, temperature, pain and limb position—even when the eyes are closed) from the body segments and carries the neural pathways that innervate and control the movement of skeletal muscles, such as those of the arms, fingers, legs and toes. The visceral nervous system controls internal organs that are not normally under the influence of blood vessels, the dilation and constriction of the pupils of the eyes and so on.
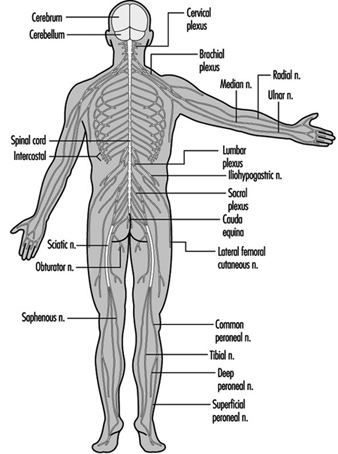
From an anatomical viewpoint, four main components need to be identified: the central nervous system, the peripheral nervous system including cranial nerves, the autonomic system and the neuroendocrine system.
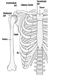
The Central Nervous System
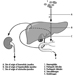
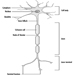
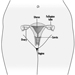
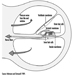
The central nervous system contains the brain and the spinal cord Figure 3. The brain lies in the skull cavity and is protected by the meninges. It is divided into three major components; in ascending order—that is, from the caudal (tail) to cervical (head) portion of the nervous system—they are the hindbrain (also called, the rhombencephalon), the midbrain (the mescencephalon) and the forebrain (the proscencephalon).
Figure 3. The central and peripheral divisions of the nervous system
The hindbrain
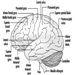
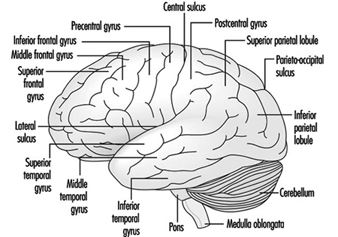
The three major components of the hindbrain are the medulla oblongata, the pons and the cerebellum figure 4.
Figure 4. The brain shown from a lateral side.
The medulla oblongata contains neural structures that control heart rate and breathing, sometimes the targets of neurotoxic agents and drugs causing death. Located between the medulla oblongata and the midbrain, the pons (bridge) derives its names from the large number of fibres traversing its anterior aspect en route to the cerebellar hemispheres. The cerebellum—in Latin, little brain—is characteristically corrugated in appearance. The cerebellum receives sensory information and sends motor messages essential for motor coordination. It is responsible (among other functions) for the execution of fine movements. This scheduling—or programming—requires the adequate timing of sensory inputs and motor responses. The cerebellum is often the target of numerous neurotoxic agents—for example, alcoholic beverages, many industrial solvents, lead—which affect motor responses.
The midbrain
The midbrain is a narrow part of the brain connecting the hindbrain to the forebrain. Structures of the midbrain are the cerebral aqueduct, the tectum, the cerebral peduncles, the substantia nigra and the red nucleus. The cerebral aqueduct is a channel that connects the third with the fourth ventricles (liquid-filled cavities of the brain); the cerebrospinal fluid (CSF) flows through this opening.
The forebrain
This part of the brain is subdivided into diencephalon (“between brain”) and the cerebrum. The major regions of the diencephalon are the thalamus and the hypothalamus. “Thalamus” means “inner room”. The thalami are made up of neuronal groupings, called nuclei, which have five main functions:
- receiving sensory information and sending it to primary areas of the cerebral cortex
- sending information about ongoing movement to motor areas of the cerebral cortex
- sending information on the activity of the limbic system to areas of the cerebral cortex related to this system
- sending information on intrathalamic activity to association areas of the cerebral cortex
- sending information of brain-stem reticular formation activity to widespread areas of the cerebral cortex.
The name hypothalamus means “under the thalamus”. It forms the base of the third ventricle, an important reference point for the imaging of the brain. The hypothalamus is a complex, minute neural structure responsible for many aspects of behaviour such as basic biological drives, motivation and emotion. It is the link between the nervous and the neuroendocrine system, to be reviewed below. The pituitary gland (also called the hypophysis) is linked by neurons to the hypothalamic nuclei. It is well established that the hypothalamic nerve cells perform many neurosecretory functions. The hypothalamus is linked with many other major regions of the brain including the rhinencephalon—the primitive cortex originally associated with olfaction—and the limbic system, including the hippocampus.
The cerebral cortex is the largest component of the brain, consisting of two cerebral hemispheres connected by a mass of white matter called the corpus callosum. The cerebral cortex is the surface layer of each cerebral hemisphere. Deep sulci in the cerebral cortex—the central and the lateral sulci Figure 4 —are taken as reference points to separate anatomical regions of the brain. The frontal lobe lies in front of the central sulcus. The parietal lobe begins at the back of the central sulcus, and lies next to the occipital lobe, which occupies the posterior portion of the brain. The temporal lobe begins well inside the folding of the lateral sulcus and extends into the ventral aspects of the brain hemispheres. Two important components of the cerebrum are the basal ganglia and the limbic system.
The basal ganglia are nuclei—that is, clusters of nerve cells—located toward the centre of the brain. The basal ganglia comprise major centres of the extra-pyramidal motor system. (The pyramidal system, to which the term is contrasted, is involved in the voluntary control of movement.) The extrapyramidal system is selectively affected by many neurotoxic agents (e.g., manganese). In the past two decades, important discoveries have been made concerning the role these nuclei play in several neural degenerative diseases (e.g., Parkinson’s disease, Huntington’s chorea).
The limbic system is comprised of convoluted neural structures branching out into many directions and establishing connections with many “old” regions of the brain, particularly with the hypothalamus. It is involved in the control of emotional expression. The hippocampus is believed to be a structure where many memory processes occur.
The spinal cord
The spinal cord is a whitish structure situated within the vertebral canal. It is divided into four regions: cervical, thoracic, lumbar and sacral-coccyxeal. The two most easily recognizable features of the spinal cord are the grey matter containing the cell bodies of the neurons, and the white matter containing the myelinated axons of the neurons. The ventral region of the spinal cord’s grey matter contains nerve cells that regulate motor function; the middle region of the thoracic spinal cord is associated with autonomic functions. The dorsal portion receives sensory information from the spinal nerves.
The Peripheral Nervous System
The peripheral nervous system includes those neurons that are outside the central nervous system. The term peripheral describes the anatomical distribution of this system, but functionally it is artificial. The cell bodies of peripheral motor fibres, for example, are located within the central nervous system. In experimental, clinical and epidemiological neurotoxicology, the term peripheral nervous system (PNS) describes a system that is selectively vulnerable to the effects of toxic agents and that is able to regenerate.
The spinal nerves
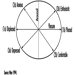
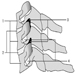
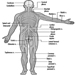
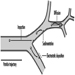
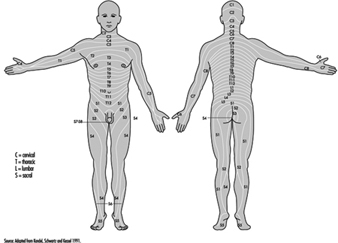
The ventral and dorsal roots are where the peripheral nerves enter and leave the spinal cord along its length. Adjoining vertebrae contain openings to allow root fibres forming the spinal nerves to leave the spinal canal. There are 31 pairs of spinal nerves, which are named according to the region of the vertebral column with which they are associated: 8 cervical, 12 thoracic, 5 lumbar, 5 sacral and 1 coccyxeal. A metamera is a region of the body innervated by a spinal nerve figure 5.
Figure 5. The segmental distribution of the spinal nerves (the metamera).
Carefully examining the motor and sensory functions of metamerae, neurologists can infer the location of lesions where damage has occurred.
Table 1. Names and main functions of each pair of cranial nerves
| Nerve1 | Conducts impulses | Functions |
| I. Olfactory | From nose to brain | Sense of smell |
| II. Optic | From eye to brain | Vision |
| III. Oculomotor | From brain to eye muscles | Eye movements |
| IV. Trochlear | From brain to external eye muscles | Eye movements |
| V. Trigeminal (or trifacial) |
From skin and mucous membrane of head and from teeth to brain; also from brain to chewing muscles | Sensations of face, scalp and teeth; chewing movements |
| VI. Abducens | From brain to external eye muscles | Turning eyes outward |
| VII. Facial | From taste buds of tongue to brain; from brain to face muscles | Sense of taste; contraction of muscles of facial expression |
| VIII. Acoustic | From ear to brain | Hearing; sense of balance |
| IX. Glossopharyngeal | From throat and taste buds of tongue to brain; also from brain to throat muscles and salivary glands | Sensations of throat, taste, swallowing movements, secretion of saliva |
| X. Vagus | From throat, larynx, and organs in thoracic and abdominal cavities to brain; also from brain to muscles of throat and to organs in thoracic and abdominal cavities | Sensations of throat, larynx, and for thoracic and abdominal organs; swallowing, voice production, slowing of heartbeat, acceleration of peristalsis |
| XI. Spinal accessory | From brain to certain shoulder and neck muscles | Shoulder movements; turning movements of head |
| XII. Hypoglossal | From brain to muscles of tongue | Tongue movements |
1 The first letter of the words of the following sentence are the first letters of the names of cranial nerves: “On Old Olympus’ Tiny Tops A Finn and German Viewed Some Hops”. Many generations of students have used this or a similar sentence to help them remember the names of cranial nerves.
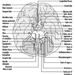
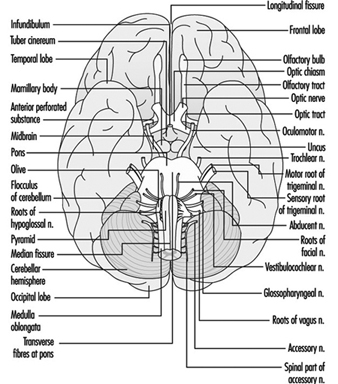
The cranial nerves
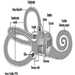
Brain stem is a comprehensive term that designates the region of the nervous system that includes the medulla, the pons and the midbrain. The brain stem is a continuation of the spinal cord upward and forward (ventrally). It is in this region where most of the cranial nerves make their exits and entrances. There are 12 pairs of cranial nerves; Table 1 describes the name and main function of each pair and Figure 6 shows the entrance and exits of some cranial nerves in the brain.
Figure 6. The brain shown from below with the entrance and exits of many cranial nerves.
The Autonomic Nervous System
The autonomic nervous system is that part of the nervous system controlling the activity of the visceral components of the human body. It is called “autonomic” because it performs its functions automatically, meaning that its functioning cannot be easily controlled at will. From an anatomical point of view, the autonomic system has two main components: the sympathetic and the parasympathetic nervous system. The sympathetic nerves controlling visceral activity arise from the thoracic and lumbar portions of the spinal cord; parasympathetic nerves arise from the brain stem and the sacral portion of the spinal cord.
From a physiological point of view, no single generalization can be made that applies to the manner in which the sympathetic and the parasympathetic nervous systems control different body organs. In most cases, visceral organs are innervated by both systems, and each type has an opposite effect in a system of checks and balances. The heart, for example, is innervated by sympathetic nerves whose excitation produces an acceleration of the heartbeat, and by parasympathetic nerves whose excitation produce a slowing of the heartbeat. Either system can stimulate or inhibit the organs it innervates. In other cases, organs are predominantly or exclusively controlled by one system or the other. A vital function of the autonomic nervous system is the maintenance of homeostasis (stable state of equilibrium) and for the adaptation of the animal body to its external environment. Homeostasis is the state of equilibrium of body functions achieved by an active process; the control of body temperature, water and electrolytes are all examples of homeostatic processes.
From the pharmacological point of view, there is no single neurotransmitter associated with either sympathetic or parasympathetic functions, as was once believed. The old view that acetylcholine was the predominant transmitter of the autonomic system had to be abandoned when new classes of neurotransmitters and neuromodulators were found (e.g., dopamine, serotonin, purines and various neuropeptides).
Neuroscientists have recently revived the behavioural point of view of the autonomic nervous system. The autonomic nervous system is involved in the fight-or-flight instinctive reaction still present in humans, which is, for the most part, the basis for the physiological reactions caused by stress. Interactions between the nervous system and immunological functions are possible through the autonomic nervous system. Emotions that originate from the autonomic nervous system can be expressed via the skeletal muscles.
The autonomic control of smooth muscles
The muscles of the viscera—except for the heart—are the smooth muscles. Heart muscle has characteristics of both skeletal and smooth muscle. Like skeletal muscles, smooth muscles also contain the two proteins actin and, in smaller proportions, myosin. Unlike skeletal muscles, they do not present the regular organization of sarcolemes, the contractile unit of the muscle fibre. The heart is unique in that it can generate myogenic activity—even after its neural innervations have been severed, it can contract and relax for several hours by itself.
The neuromuscular coupling in smooth muscles differs from that of skeletal muscles. In skeletal muscles, the neuromuscular junction is the link between the nerve and the muscle fibres. In smooth muscle, there is no neuromuscular junction; the nerve endings enter the muscle, spreading in all directions. Electrical events inside the smooth muscle therefore are much slower than those in skeletal muscles. Finally, smooth muscle has the unique characteristic of exhibiting spontaneous contractions, such as that exhibited by the gut. To a large extent, the autonomic nervous system regulates the smooth muscles’ spontaneous activity.
The central components of the autonomic nervous system
The main role of the autonomic nervous system is to regulate the activity of smooth muscles, heart, glands in the digestive tract, sweat glands, and adrenal and other endocrine glands. The autonomic nervous system has a central component—the hypothalamus, located at the base of the brain—where many autonomic functions are integrated. Most importantly, the central components of the autonomic system are directly involved in the regulation of biological drives (temperature regulation, hunger, thirst, sex, urination, defecation and so on), motivation, emotion and to a great extent in “psychological” functions such as moods, affect and feelings.
Neuroendocrine System
Glands are the organs of the endocrine system. They are called endocrine glands because their chemical messages are delivered inside the body, directly into the blood stream (in contrast with exocrine glands, such as sweat glands, whose secretions appear on the outer surface of the body). The endocrine system provides slow but long-lasting control over organs and tissues through chemical messengers called hormones. Hormones are the main regulators of body metabolism. But, because of intimate links among the central, peripheral, and autonomic nervous systems, the neuroendocrine system—a term that captures such complex links—is now conceived of as a powerful modifier of the structure and function of the human body and behaviour.
Hormones have been defined as chemical messengers which are released from cells into the bloodstream to exert their action on target cells some distance away. Until recently, hormones were distinguished from neurotransmitters, discussed above. The latter are chemical messengers released from neurons onto a synapse between the nerve terminals and another neuron or an effector (i.e., muscle or gland). However, with the discovery that classical neurotransmitters such as dopamine can also act as hormones, the distinction between neurotransmitters and hormones is now less and less clear. Thus, based on purely anatomical considerations, hormones derived from nerve cells may be called neurohormones. From a functional point of view, the nervous system can be thought of as a truly neurosecretory system.
The hypothalamus controls endocrine functions through a link with the pituitary gland (also called the hypophysis, a tiny gland located at the base of the brain). Until the middle 1950s the endocrine glands were viewed as a separate system governed by the pituitary gland, often called the “master gland”. At that time, a neurovascular hypothesis was advanced that established the functional role of the hypothalamic/hypophysial factors in the control of endocrine function. In this view, the endocrine hypothalamus provides the final common neuroendocrine pathway in the control of the endocrine system. It has now been firmly established that the endocrine system is itself regulated by the central nervous system as well as the endocrine inputs. Thus, neuroendocrinology is now an appropriate term to describe the discipline that studies the reciprocal integrated roles of the nervous and the endocrine systems in the control of physiological processes.
With increasing understanding of neuroendocrinology, original divisions are breaking down. The hypothalamus, which is located above and connected to the pituitary gland, is the link between the nervous and the endocrine systems, and many of its nerve cells perform secretory functions. It is also linked with other major regions of the brain, including the rhinencephalon—the primitive cortex originally associated with olfaction or sense of smell—and the limbic system, associated with emotions. It is in the hypothalamus that hormones released by the posterior pituitary gland are produced. The hypothalamus also produces substances that are called releasing and inhibiting hormones. These act on the adenohypophysis, causing it to enhance or inhibit the production of anterior pituitary gland hormones, which act on glands located elsewhere (thyroid, adrenal cortex, ovaries, testicles and others).
Chemical Neurotoxic Agents
Definition of Neurotoxicity
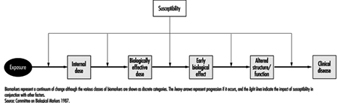
Neurotoxicity refers to the capability of inducing adverse effects in the central nervous system, peripheral nerves or sensory organs. A chemical is considered to be neurotoxic if it is capable of inducing a consistent pattern of neural dysfunction or change in the chemistry or structure of the nervous system.
Neurotoxicity is generally manifested as a continuum of symptoms and effects, which depend on the nature of the chemical, the dose, the duration of exposure and the traits of the exposed individual. The severity of the observed effects, as well as the evidence for neurotoxicity, increases through levels 1 to 6, shown in Table 1. Short-term or low-dose exposure to a neurotoxic chemical may result in subjective symptoms such as headache and dizziness, but the effect usually is reversible. With increasing dose, neurological changes may show up, and eventually irreversible morphological changes are generated. The degree of abnormality needed for implying neurotoxicity of a chemical agent is a controversial issue. According to the definition, a consistent pattern of neural dysfunction or change in the chemistry or structure of the nervous system is considered if there is well-documented evidence for persistent effects on level 3, 4, 5 or 6 in Table 1. These levels reflect the weight of evidence provided by different signs of neurotoxicity. Neurotoxic substances include naturally occurring elements such as lead, mercury and manganese; biological compounds such as tetrodotoxin (from the puffer fish, a Japanese delicacy) and domoic acid (from contaminated mussels); and synthetic compounds including many pesticides, industrial solvents and monomers.
Table 1. Grouping neurotoxic effects to reflect their relative strength for establishing neurotoxicity
|
Level |
Grouping |
Explanation/Examples |
|
6 |
Morphological changes |
Morphological changes include cell death and axonopathy as well as subcellular morphological changes. |
|
5 |
Neurological changes |
Neurological change embraces abnormal findings in neurological examinations on single individuals. |
|
4 |
Physiological/behavioural changes |
Physiological/behavioural changes comprise experimental findings on groups of animals or humans such as changes in evoked potentials and EEG, or changes in psychological and behavioural tests. |
|
3 |
Biochemical changes |
Biochemical changes cover changes in relevant biochemical parameters (e.g., transmitter level, GFA-protein content (glial fibrillary acidic protein) or enzyme activities). |
|
21 |
Irreversible, subjective symptoms |
Subjective symptoms. No evidence of abnormality on neurological, psychological or other medical examination. |
|
11 |
Reversible, subjective symptoms |
Subjective symptoms. No evidence of abnormality on neurological, psychological, or other medical examination. |
1 Humans only
Source: Modified from Simonsen et al. 1994.
In the United States between 50,000 and 100,000 chemicals are in commerce, and 1,000 to 1,600 new chemicals are submitted for evaluation each year. More than 750 chemicals and several classes or groups of chemical compounds are suspected to be neurotoxic (O’Donoghue 1985), but the majority of chemicals have never been tested for neurotoxic properties. Most of the known neurotoxic chemicals available today have been identified by case-reports or through accidents.
Although neurotoxic chemicals often are produced to fulfil specific uses, exposure may arise from several sources—use in private homes, in agriculture and in industries, or from polluted drinking water and so on. Fixed a priori preconceptions about which neurotoxic compounds are expected to be found in which occupations should therefore be viewed with caution, and the following citations should be looked upon as possible examples including a few of the most common neurotoxic chemicals (Arlien-Søborg 1992; O’Donoghue 1985; Spencer and Schaumburg 1980; WHO 1978).
Symptoms of Neurotoxicity
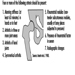
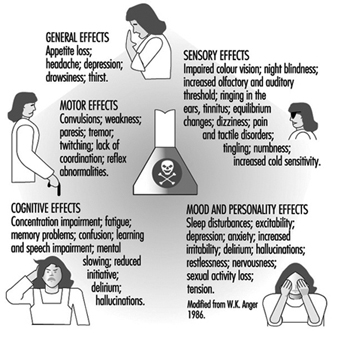
The nervous system generally reacts rather stereotypically to exposure to neurotoxic substances Figure 1. Some typical syndromes are indicated below.
Figure 1. Neurological and behavioural effects of exposure to neurotoxic chemicals.
Polyneuropathy
This is caused by impairment of motor and sensory nerve function leading to weakness of the muscles, with paresis usually most pronounced peripherally in the upper and lower extremities (hands and feet). Prior or simultaneous paraesthesia (tingling or numbness in the fingers and toes) may occur. This may lead to difficulties in walking or in the fine coordination of hands and fingers. Heavy metals, solvents and pesticides, among other chemicals, may result in such disability, even if the toxic mechanism of these compounds may be totally different.
Encephalopathy
This is caused by a diffuse impairment of the brain, and may result in fatigue; impairment of learning, memory and ability to concentrate; anxiety, depression, increased irritability and emotional instability. Such symptoms may indicate early diffuse degenerative brain disorder as well as occupational chronic toxic encephalopathy. Often increased frequency of headaches, dizziness, changes in sleep pattern and reduced sexual activity may also be present from the early stages of the disease. Such symptoms may develop following long-term, low-level exposure to several different chemicals such as solvents, heavy metals or hydrogen sulphide, and are also seen in several dementing disorders not related to work. In some cases more specific neurological symptoms can be seen (e.g., Parkinsonism with tremor, rigidity of the muscles and slowing of movements, or cerebellar symptoms such as tremor and reduced coordination of hand movements and gait). Such clinical pictures can be seen following exposure to some specific chemicals such as manganese, or MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) in the former condition, and toluene or mercury in the latter.
Gases
A wide variety of chemicals with totally different chemical structures are gases at normal temperature and have been proven neurotoxic Table 3. Some of them are extremely toxic even in very small doses, and have even been used as war gases (phosgene and cyanide); others require high doses over longer periods to give symptoms (e.g., carbon dioxide). Some are used for general anaesthesia (e.g., nitrous oxide); others are widely used in industry and in agents used for disinfection (e.g., formaldehyde). The former may induce irreversible changes in the nervous system after repeated low-level exposure, the latter apparently produce only acute symptoms. Exposure in small rooms with poor ventilation is particularly hazardous. Some of the gases are odourless, which makes them particularly dangerous (e.g., carbon monoxide). As shown in Table 2, some gases are important constituents in industrial production, while others are the result of incomplete or complete combustion (e.g., CO and CO2 respectively). This is seen in mining, steel works, power stations and so on, but may also be seen in private homes with insufficient ventilation. Essential for treatment is to stop further exposure and provide fresh air or oxygen, and in severe cases artificial ventilation.
Table 2. Gases associated with neurotoxic effects
|
Chemical |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Carbon dioxide (CO2 ) |
Welding; fermentation; manufacture, storage and use of dry ice |
Metal industry; mining; breweries |
M: Dilate vessels A: Headache; dyspnoea; tremor; loss of consciousness C: Hardly any |
|
Carbon monoxide (CO) |
Car repair; welding; metal melting; drivers; firemen |
Metal industry; mining; transportation; power station |
M: Deprivation of oxygen A: Headache; drowsiness; loss of consciousness |
|
Hydrogen sulphide (H2S) |
Fumigating of green house; manure; fishermen; fish unloading; sewerage handling |
Agriculture; fishing; sewer work |
M: Blocking oxidative metabolism A: Loss of consciousness C: Encephalopathy |
|
Cyanide (HCN) |
Electro-welding; galvanic surface treatment with nickel; copper and silver; fumigation of ships, houses foods and soil in green houses |
Metal industry; chemical industry; nursery; mining; gasworks |
M: Blocking of respiratory enzymes A: Dyspnoea; falling blood pressure; convulsions; loss of consciousness; death C: Encephalopathy; ataxia; neuropathy (e.g., aftereating cavasava) Occupational impairment uncertain |
|
Nitrous oxide (N2O) |
General anaesthesia during operation; light narcosis at dental care and delivery |
Hospitals (anaesthesia); dentists; midwife |
M: Acute change in nerve cell membrane; degeneration of nerve cells after long-term exposure A: Light-headedness; drowsiness; loss of consciousness C: Numbness of fingers and toes; reduced coordination; encephalopathy |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor- and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain.
Ataxia: impaired motor coordination.
Metals
As a rule the toxicity of metals increases with increasing atomic weight, lead and mercury being particularly toxic. Metals are usually found in nature at low concentrations, but in certain industries they are used in great amounts (see Table 3) and may give rise to occupational risk for the workers. Moreover, considerable amounts of metals are found in waste water and may give rise to environmental risk for the residents close to the plants but also at greater distances. Often the metals (or, for example, organic mercury compounds) are taken up into the food chain and will accumulate in fish, birds and animals, representing a risk for consumers. The toxicity and the way in which the metals are handled by the organism may depend on the chemical structure. Pure metals may be taken up by inhalation or skin contact of vapour (mercury) and/or small particles (lead), or orally (lead). Inorganic mercury compounds (e.g., HgCl2) are mainly taken up by mouth, while organic metal compounds (e.g., tetraethyl lead) mainly are taken up by inhalation or by skin contact. The body burden may to a certain degree be reflected in the concentration of metal in the blood or urine. This is the basis for biological monitoring. In treatment it must be recalled that especially lead is released very slowly from deposits in the body. The amount of lead in bones will normally be reduced by only 50% over 10 years. This release may be speeded up by the use of chelating agents: BAL (dimercapto-1-propanol), Ca-EDTA or penicillamine.
Table 3. Metals and their inorganic compounds associated with neurotoxicity
|
Chemical |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Lead |
Melting; soldering; grinding; repair; glazing; plasticizer |
Metal work; mining; accumulator plants; car repair; shipyards; glass workers; ceramics; pottery; plastic |
M: Impairment of oxidative metabolism of nerve cells and glia A: Abdominal pain; headache; encephalopathy; seizures C: Encephalopathy; polyneuropathy, including drop hand |
|
Mercury Elemental |
Electrolysis; electrical instruments (gyroscope; manometer; thermometer; battery; electric bulb; tubes, etc.); amalgam filling |
Chloralkali plants; mining; electronics; dentistry; polymer production; paper and pulp industry |
M: Impairment at multiple sites in nerve cells A: Lung inflammation; headache; impaired speech C: Inflammation of gums; appetite loss; encephalopathy; including tremor; irritability |
|
Calomel Hg2Cl2 |
Laboratories |
A: Low acute toxicity chronic toxic effects, see above |
|
|
Sublimate HgCl2 |
Disinfection |
Hospitals; clinics; laboratories |
M: Acute tubular and glomerular renal degeneration. Verytoxic even in small oral doses, lethal down to 30 mg/kgweight C: See above. |
|
Manganese |
Melting (steel alloy); cutting; welding in steel; dry batteries |
Manganese mining; steel and aluminium production; metal industry; battery production; chemical industry; brickyard |
M: Not known, possible changes in dopamine and catecholamine in basal ganglia in the centre of the brain A: Dysphoria C: Encephalopathy including Parkinsonism; psychosis; appetite loss; irritability; headache; weakness |
|
Aluminium |
Metallurgy; grinding; polishing |
Metal industry |
M: Unknown C: Possibly encephalopathy |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor- and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain.
Monomers
Monomers constitute a large, heterogeneous group of reactive chemicals used for chemical synthesis and production of polymers, resins and plastics. Monomers comprise polyhalogenated aromatic compounds such as p-chlorobenzene and 1,2,4-trichlorbenzene; unsaturated organic solvents such as styrene and vinyltoluene, acrylamide and related compounds, phenols, ɛ-caprolactam and ζ-aminobutyrolactam. Some of the widely used neurotoxic monomers and their effect on the nervous system are listed in Table 3. Occupational exposure to neurotoxic monomers may take place at industries manufacturing, transporting and using chemical products and plastic products. During handling of polymers containing rest monomers, and during moulding in boat yards and in dental clinics, a substantial exposure to neurotoxic monomers takes place. Upon exposure to these monomers uptake may take place during inhalation (e.g., carbon disulphide and styrene) or by skin contact (e.g., acrylamide). As monomers are a heterogeneous group of chemicals, several different mechanisms of toxicity are likely. This is reflected by differences in symptoms (Table 4).
Table 4. Neurotoxic monomers
|
Compound |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Acrylamide |
Employees exposed to the monomer |
Polymer production; tunnelling and drilling operations |
M: Impaired axonal transport C: Polyneuropathy; dizziness; tremor and ataxia |
|
Acrylonitrile |
Accidents in labs and industries; house fumigation |
Polymer and rubber production; chemical synthesis |
A: Hyperexcitability; salivation; vomiting; cyanosis; ataxia; difficulty breathing |
|
Carbon disulphide |
Production of rubber and viscose rayon |
Rubber and viscose rayon industries |
M: Impaired axonal transport and enzyme activity is likely C: Peripheral neuropathy; encephalopathy; headache; vertigo; gastrointestinal disturbances |
|
Styrene |
Production of glass-reinforced plastics; monomer manufacture and transportation; use of styrene-containing resins and coatings |
Chemical industry; fibreglass production; polymer industry |
M: Unknown A: Central nervous system depression; headache C: Polyneuropathy; encephalopathy; hearing loss |
|
Vinyltoluene |
Resin production; insecticide compounds |
Chemical and polymer industry |
C: Polyneuropathy; reduced motor nerve conductionvelocity |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain.
Ataxia: impaired motor coordination.
Organic solvents
Organic solvents is a common designation for a large group of more than 200 lipophilic chemical compounds capable of dissolving fats, oils, waxes, resins, rubber, asphalt, cellulose filaments and plastic materials. They are usually fluids at room temperature with boiling points below 200 to 250°C, and are easily evaporated. They are mainly taken up via the lungs but some may penetrate the skin as well. Due to their lipophilicity they are distributed to organs rich in fat. Thus high concentrations are found in body fat, bone marrow, liver and brain, which also may act as reservoirs of solvents. The partition coefficient octanol/water can indicate whether high brain concentrations are to be expected. The mechanism of toxicity is not yet known, but several possibilities have been envisioned: blocking important enzymes in the metabolic breakdown of glucose and thus reducing energy available for neuronal processing; reducing energy formation in the mitochondria; changing neuronal membranes, leading to impairment of ion channel function; slowing of axonal flow. Methylene chloride is metabolized to CO, which blocks the transport of oxygen in the blood. Large groups of workers in a great variety of professions are exposed daily or at least frequently (see Table 5). In some countries the consumption of organic solvents has declined in some occupations due to hygienic improvements and substitution (e.g., house painters, graphic industry workers, metal workers), while in other occupations the pattern of exposure has changed but the total amount of organic solvents has remained unchanged. For example, trichloroethylene has been replaced by 1,1,1-trichloroethane and freon. So solvents are still a major hygienic problem at many workplaces. People are at particular risk when exposed in small rooms with poor ventilation and with high temperature, increasing the evaporation. Physical work increases the pulmonary uptake of solvents. In several countries (in particular the Nordic countries), compensation has been given to workers who have developed chronic toxic encephalopathy following long-term, low-level exposure to solvents.
Table 5. Organic solvents associated with neurotoxicity
|
Chemical |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Chlorinated hydrocarbons: trichloroethylene; 1,1,1-trichloroethane; tetrachloroethylene |
Degreasing; electroplating; painting; printing; cleaning; general and light anaesthesia |
Metal industry; graphic industry; electronic industry; dry cleaners; anaesthetists |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy; polyneuropathy; trigeminal affection (TRI); hearing loss |
|
Methylene chloride |
Extraction, including extraction of caffeine; paint remover |
Food industry; painters; graphic industry |
M: Metabolism ® CO A: Prenarcotic symptoms; coma C: Encephalopathy |
|
Methyl chloride |
Refrigerator production and repair |
Refrigerator production; rubber industry; plastic industry |
M: Unknown A: Prenarcotic symptoms; loss of consciousness; death C: Encephalopathy |
|
Toluene |
Printing; cleaning; degreasing; electroplating; painting; spray painting |
Graphic industry; electronic industry |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy; cerebellar dysfunction; polyneuropathy; hearing loss; visual disturbance |
|
Xylene |
Printing; synthesis of phthalic anhydride; painting; histology laboratory procedures |
Graphic industry; plastic industry; histology laboratories |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy; visual disturbance; hearing loss |
|
Styrene |
Polymerization; moulding |
Plastic industry; fibreglass production |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy; polyneuropathy; hearing loss |
|
Hexacarbons: n-hexane; methyl butyl ketone (MBK); methyl ethyl ketone (MEK) |
Gluing; printing; plastic coating; painting; extraction |
Leather and shoe industry; graphic industry; painter; laboratories |
M: Impairment of axonal transport A: Prenarcotic C: Polyneuropathy; encephalopathy |
|
Various solvents: Freon 113 |
Refrigerator production and repair; dry cleaning; degreasing |
Refrigerator production; metal industry; electronic industry; dry cleaning |
M: Unknown A: Mild prenarcotic symptoms C: Encephalopathy |
|
Diethylether; halothane |
General anaesthetics (nurses; doctors) |
Hospitals; clinics |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy |
|
Carbon disulphide |
See monomers |
See monomers |
See monomers |
|
Mixtures: white spirit and thinner |
Painting; degreasing; cleaning; printing; impregnation; surface treatment |
Metal industry; graphic industry; wood industry; painters |
M: Unknown A: Prenarcotic symptoms C: Encephalopathy |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor- and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain
Pesticides
Pesticides is used as a generic term for any chemical designed to kill groups of plants or animals that are a human health hazard or may cause economic loss. It includes insecticides, fungicides, rodenticides, fumigants and herbicides. Approximately 5 billion pounds of pesticide products made up of more than 600 active pesticide ingredients are annually used in agriculture worldwide. Organophosphorus, carbamate and organochlorine pesticides together with pyrethroids, chlorophenoxy herbicides and organic metal compounds used as fungicides have neurotoxic properties (Table 6). Among the many different chemicals used as rodenticides, some (e.g., strychnine, zinc phosphide and thallium) are neurotoxic too. Occupational exposure to neurotoxic pesticides is mainly associated with agricultural work such as pesticide handling and working with treated crops, but exterminators, pesticide manufacturing and formulating employees, highway and railway workers, as well as greenhouse, forestry and nursery workers may have a substantial risk of being exposed to neurotoxic pesticides as well. Children, who constitute a significant proportion of the agricultural workforce, are especially vulnerable because their nervous systems are not fully developed. The acute effects of pesticides are generally well described, and long-lasting effects upon repeated exposure or single high dose exposure are often seen (Table 6), but the effect of repeated subclinical exposure is uncertain.
Table 6. Classes of common neurotoxic pesticides, exposure, effects and associated symptoms
|
Compound |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Organo-phosphorus compounds: Beomyl; Demethon; Dichlorvos; Ethyl parathion; Mevinphos; Phosfolan; Terbufos; Malathion |
Handling; treatment of crops; working with treated crops; dock labourer |
Agriculture; forestry; chemical; gardening |
M: Acetyl cholinesterase inhibition A: Hyperactivity; neuromuscular paralysis; visual impairment; breathing difficulty; restlessness; weakness; vomiting; convulsions |
|
Carbamates: Aldicarb; Carbaryl; Carbofuran; Propoxur |
M: Delayed neurotoxicity axonopathy2 C: Polyneuropathy; numbness and tingling in feet; muscle weakness; sensory disturbance; paralysis |
||
|
Organochlorine: Aldrin; Dieldrin; DDT; Endrin; Heptachlor; Lindane; Methoxychlor; Mirex; Toxaphene |
See above |
See above |
A: Excitability; apprehension; dizziness; headache; confusion; loss of balance; weakness; ataxia; tremors; convulsions; coma C: Encephalopathy |
|
Pyrethroids |
See above |
See above |
M: Altering flow of sodium ions through nerve cellmembrane A: Repeated firing of the nerve cell; tremor; convulsion |
|
2,4-D |
Herbicide |
Agriculture |
C: Polyneuropathy |
|
Triethyltin hydroxide |
Surface treatment; handling treated wood |
Wood and wood products |
A: Headache; weakness; paralysis; visual disturbances C: Polyneuropathy; CNS effects |
|
Methyl bromide |
Fumigating |
Greenhouses; insecticide; manufacture of refrigerators |
M: Unknown A: Visual and speech disturbances; delirium; convulsion C: Encephalopathy |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain.
Ataxia: impaired motor coordination.
2 Mainly phosphates or phosphonates.
Other chemicals
Several different chemicals which do not fit into the above-mentioned groups also possess neurotoxicity. Some of these are used as pesticides but also in different industrial processes. Some have well-documented acute and chronic neurotoxic effects; others have obvious acute effects, but the chronic effects are only poorly examined. Examples of these chemicals, their uses and effects are listed in Table 7.
Table 7. Other chemicals associated with neurotoxicity
|
Chemical |
Examples of source of exposure |
Selected industries at risk |
Effects1 |
|
Boric acid |
Welding; fluxes; preservation |
Metal; glass |
A: Delirium; convulsion C: CNS depression. |
|
Disulfiram |
Pharmaceutical |
Rubber |
C: Fatigue; peripheral neuropathy; sleepiness |
|
Hexachlorophene |
Antibacterial soaps |
Chemical |
C: CNS oedema; peripheral nerve damage |
|
Hydrazine |
Reducing agents |
Chemical; army |
A: Excitement; appetite loss; tremor; convulsion |
|
Phenol/Cresol |
Antiseptics |
Plastics; resins; chemical; hospitals; laboratories |
M: Denatures proteins and enzymes A: Reflex loss; weakness; tremor; sweating; coma C: Appetite loss; mental disturbance; ringing in the ears |
|
Pyridine |
Ethanol denaturation |
Chemical; textile |
A: CNS depression; mental depression; fatigue; appetite loss C: Irritability; sleep disorders; polyneuropathy; double vision |
|
Tetraethyl lead |
Gasoline additive |
Chemical; transport |
C: Irritability; weakness; tremor; vision difficulties |
|
Arsine |
Batteries; insecticide; melting |
Smelting; glasswork; ceramics; manufacture of paper |
M: Impairing enzyme function A: Reduced sensation; paresis; convulsion; coma C: Motor impairment; ataxia; vibration sense loss; polyneuropathy |
|
Lithium |
Oil additive; pharmaceutical |
Petrochemical |
A/C: Appetite loss; ringing in the ears; vision blurring; tremor; ataxia |
|
Selenium |
Melting; production of rectifiers; vulcanization; cutting oils; antioxidant |
Electronic; glass works; metal industry; rubber industry |
A: Delirium; anosmia C: Odour of garlic; polyneuropathy; nervousness |
|
Thallium |
Rodenticide |
Glass; glass products |
A: Appetite loss; tiredness; drowsiness; metallic taste; numbness; ataxia |
|
Tellurium |
Melting; rubber production; catalyst |
Metal; chemical; rubber; electronic |
A: Headache; drowsiness; neuropathy C: Odour of garlic; metallic taste; Parkinsonism; depression |
|
Vanadium |
Melting |
Mining; steel production; chemical industry |
A: Appetite loss; ringing in the ears; somnolence, tremor C: Depression; tremor; blindness |
1 M: mechanism; A: acute effects; C: chronic effects.
Neuropathy: dysfunction of motor and sensory peripheral nerve fibres.
Encephalopathy: brain dysfunction due to generalized impairment of the brain.
Ataxia: impaired motor coordination
Manifestations of Acute and Early Chronic Poisoning
Current knowledge of the short- and long-term manifestations of exposure to neurotoxic substances comes from experimental animal studies and human chamber studies, epidemiological studies of active and retired and/or diseased workers, clinical studies and reports, as well as large-scale disasters, such as those that occurred in Bhopal, following a leak of methyl isocyanate, and in Minamata, from methyl mercury poisoning.
Exposure to neurotoxic substances can produce immediate effects (acute) and/or long-term effects (chronic). In both cases, the effects can be reversible and disappear over time following reduction or cessation of exposure, or result in permanent, irreversible damage. The severity of acute and chronic nervous system impairment depends on exposure dose, which includes both the quantity and duration of exposure. Like alcohol and recreational drugs, many neurotoxic substances may initially be excitatory, producing a sensation of well-being or euphoria and/or speeding up motor functions; as the dose increases in quantity or in time, these same neurotoxins will depress the nervous system. Indeed, narcosis (a state of stupor or insensibility) is induced by a large number of neurotoxic substances, which are mind-altering and depress the central nervous system.
Acute Poisoning
Acute effects reflect the immediate response to the chemical substance. The severity of the symptoms and resulting disorders depends on the quantity that reaches the nervous system. With mild exposures, acute effects are mild and transient, disappearing when exposure ceases. Headache, tiredness, light-headedness, difficulty concentrating, feelings of drunkenness, euphoria, irritability, dizziness and slowed reflexes are the types of symptoms experienced during exposure to neurotoxic chemicals. Although these symptoms are reversible, when exposure is repeated day after day, the symptoms recur as well. Moreover, since the neurotoxic substance is not immediately eliminated from the body, symptoms can persist following work. Reported symptoms at a particular workstation are a good reflection of chemical interference with the nervous system and should be considered a warning signal for potential over-exposure; preventive measures to reduce exposure levels should be initiated.
If exposure is very high, as can occur with spills, leaks, explosions and other accidents, symptoms and signs of intoxication are debilitating (severe headaches, mental confusion, nausea, dizziness, incoordination, blurred vision, loss of consciousness); if exposure is high enough, effects can be long-lasting, possibly resulting in coma and death.
Acute pesticide-related disorders are a common occurrence among agricultural workers in food-producing countries, where large amounts of toxic substances are used as insecticides, fungicides, nematicides, and herbicides. Organophosphates, carbamates, organochlorines, pyrethrum, pyrethrin, paraquat and diquat are among the major categories of pesticides; however, there are thousands of pesticide formulations, containing hundreds of different active ingredients. Some pesticides, such as maneb, contain manganese, while others are dissolved in organic solvents. In addition to the symptoms mentioned above, acute organophosphate and carbamate poisoning may be accompanied by salivation, incontinence, convulsions, muscle twitching, diarrhoea, visual disturbances, as well as respiratory difficulties and a rapid heart rate; these result from an excess of the neurotransmitter acetylcholine, which occurs when these substances attack a chemical called cholinesterase. Blood cholinesterase decreases proportionally to the degree of acute organophosphate or carbamate intoxication.
With some substances, such as organophosphorus pesticides and carbon monoxide, high-level acute exposures can produce delayed deterioration of certain parts of the nervous system. For the former, numbness and tingling, weakness and disequilibrium can occur a few weeks after exposure, while for the latter, delayed neurologic deterioration can take place, with symptoms of mental confusion, ataxia, motor incoordination and paresis. Repeated acute episodes of high levels of carbon monoxide have been associated with later-life Parkinsonism. It is possible that high exposures to certain neurotoxic chemicals may be associated with an increased risk for neurodegenerative disorders later on in life.
Chronic Poisoning
Recognition of the hazards of neurotoxic chemicals has led many countries to reduce the permissible exposure levels. However, for most chemicals, the level at which no adverse effect will occur over long-term exposure is still unknown. Repeated exposure to low to medium levels of neurotoxic substances throughout many months or years can alter nervous system functions in an insidious and progressive manner. Continued interference with molecular and cellular processes causes neurophysiological and psychological functions to undergo slow alterations, which in the early stages may go unseen since there are large reserves in the nervous system circuitry and damage can, in the first stages, be compensated through new learning.
Thus, initial nervous system injury is not necessarily accompanied by functional disorders and may be reversible. However, as the damage progresses, symptoms and signs, often non-specific in nature, become apparent, and individuals may seek medical attention. Finally, impairment may become so severe that a clear clinical syndrome, generally irreversible, is manifest.
Figure 1 schematizes the health deterioration continuum associated with exposure to neurotoxic substances. Progression of neurotoxic dysfunction is dependent on both the duration and concentration of exposure (dose), and may be influenced by other workplace factors, individual health status and susceptibility as well as lifestyle, particularly drinking and exposure to neurotoxic substances used in hobbies, such as glues applied in furniture assembly or plastic model building, paints and paint removers.
Figure 1. Health deterioration on a continuum with increasing dosage
Different strategies are adopted for identification of neurotoxin-related illness among individual workers and for the surveillance of early nervous system deterioration among active workers. Clinical diagnosis relies on a constellation of signs and symptoms, coupled to the medical and exposure history for an individual; aetiologies other than exposure must be systematically ruled out. For the surveillance of early dysfunction among active workers, the group portrait of dysfunction is important. Most often, the pattern of dysfunction observed for the group will be similar to the pattern of impairment clinically observed in the disease. It is somewhat like summing early, mild alterations to produce a picture of what is happening to the nervous system. The pattern or profile of the overall early response provides an indication of the specificity and the type of action of the particular neurotoxic substance or mixture. In workplaces with potential exposure to neurotoxic substances, health surveillance of groups of workers may prove particularly useful for prevention and workplace action in order to avoid the development of more severe illness (see Figure 2). Workplace studies carried out throughout the world, with active workers exposed to specific neurotoxic substances or to mixtures of various chemicals, have provided valuable information on early manifestations of nervous system dysfunction in groups of exposed workers.
Figure 2. Preventing neurotoxicity at work.
Early symptoms of chronic poisoning
Altered mood states are most often the first symptoms of the initial changes in nervous system functioning. Irritability, euphoria, sudden mood changes, excessive tiredness, feelings of hostility, anxiousness, depression and tension are among the mood states most often associated with neurotoxic exposures. Other symptoms include memory problems, concentration difficulties, headaches, blurred vision, feelings of drunkenness, dizziness, slowness, tingling sensation in hands or feet, loss of libido and so on. Although in the early stages these symptoms are usually not sufficiently severe to interfere with work, they do reflect diminished well-being and affect one’s capacity to fully enjoy family and social relations. Often, because of the non-specific nature of these symptoms, workers, employers and occupational health professionals tend to ignore them and look for causes other than workplace exposure. Indeed, such symptoms may contribute to or aggravate an already difficult personal situation.
In workplaces where neurotoxic substances are used, workers, employers and occupational health and safety personnel should be particularly aware of the symptomatology of early intoxication, indicative of nervous system vulnerability to exposure. Symptom questionnaires have been developed for worksite studies and surveillance of workplaces where neurotoxic substances are used. Table 1 contains an example of such a questionnaire.
Table 1. Chronic symptoms checklist
Symptoms experienced in the past month
1. Have you tired more easily than expected for the type of activity you do?
2. Have you felt light-headed or dizzy?
3. Have you had difficulty concentrating?
4. Have you been confused or disoriented?
5. Have you had trouble remembering things?
6. Have your relatives noticed that you have trouble remembering things?
7. Have you had to make notes to remember things?
8. Have you found it hard to understand the meaning of newspapers?
9. Have you felt irritable?
10. Have you felt depressed?
11. Have you had heart palpitations even when you are not exerting yourself?
12. Have you had a seizure?
13. Have you been sleeping more often than is usual for you?
14. Have you had difficulty falling asleep?
15. Have you been bothered by incoordination or loss of balance?
16. Have you had any loss of muscle strength in your legs or feet?
17. Have you had any loss of muscle strength in your arms or hands?
18. Have you had difficulty moving your fingers or grasping things?
19. Have you had hand numbness and tingling in your fingers lasting for more than a day?
20. Have you had hand numbness and tinging in your toes lasting more than a day?
21. Have you had headaches at least once a week?
22. Have you had difficulty driving home from work because you felt dizzy or tired?
23. Have you felt “high” from the chemicals used at work?
24. Have you had a lower tolerance for alcohol (takes less to get drunk)?
Source: Taken from Johnson 1987.
Early motor, sensory and cognitive changes in chronicpoisoning
With increasing exposure, changes can be observed in motor, sensory and cognitive functions in workers exposed to neurotoxic substances, who do not present clinical evidence of abnormality. Since the nervous system is complex, and certain areas are vulnerable to specific chemicals, while others are sensitive to the action of a large number of toxic agents, a wide range of nervous system functions may be affected by a single toxic agent or a mixture of neurotoxins. Reaction time, hand-eye coordination, short-term memory, visual and auditory memory, attention and vigilance, manual dexterity, vocabulary, switching attention, grip strength, motor speed, hand steadiness, mood, colour vision, vibrotactile perception, hearing and smell are among the many functions that have been shown to be altered by different neurotoxic substances.
Important information on the type of early deficits that result from exposure has been provided by comparing performance between exposed and non-exposed workers and with respect to the degree of exposure. Anger (1990) provides an excellent review of worksite neurobehavioural research up to 1989. Table 2 adapted from this article, provides an example of the type of neuro-functional deficits that have been consistently observed in groups of active workers exposed to some of the most common neurotoxic substances.
Table 2. Consistent neuro-functional effects of worksite exposures to some leading neurotoxic substances
|
Mixed organic solvents |
Carbon disulphide |
Styrene |
Organophos- |
Lead |
Mercury |
|
|
Acquisition |
+ |
|
|
|
+ |
|
|
Affect |
+ |
|
+ |
|
+ |
|
|
Categorization |
+ |
|
|
|
|
|
|
Coding |
+ |
+ |
|
|
+ |
+ |
|
Colour vision |
+ |
|
+ |
|
|
|
|
Concept shifting |
+ |
|
|
|
|
|
|
Distractibility |
|
|
|
|
+ |
|
|
Intelligence |
+ |
+ |
|
+ |
+ |
+ |
|
Memory |
+ |
+ |
+ |
+ |
+ |
+ |
|
Motor coordination |
+ |
+ |
+ |
|
+ |
+ |
|
Motor speed |
+ |
+ |
+ |
|
+ |
+ |
|
Near visual contrast sensitivity |
+ |
|
|
|
|
|
|
Odour perception threshold |
+ |
|
|
|
|
|
|
Odour identification |
+ |
|
|
|
+ |
|
|
Personality |
+ |
+ |
|
|
|
+ |
|
Spatial relations |
+ |
+ |
|
|
+ |
|
|
Vibrotactile threshold |
+ |
|
|
+ |
|
+ |
|
Vigilance |
+ |
+ |
|
|
+ |
|
|
Visual field |
|
|
|
|
+ |
+ |
|
Vocabulary |
|
|
|
|
+ |
|
Source: Adapted from Anger 1990.
Although at this stage in the continuum from well-being to disease, loss is not in the clinically abnormal range, there can be health-related consequences associated with such changes. For example, decreased vigilance and reduced reflexes may put workers in greater danger of accidents. Smell is used to identify leaks and mask saturation (cartridge breakthrough), and acute or chronic loss of smell renders one less apt to identify a potentially hazardous situation. Mood changes may interfere with inter-personal relations at work, socially and in the home. These initial stages of nervous system deterioration, which can be observed by examining groups of exposed workers and comparing them to non-exposed workers or with respect to their degree of exposure, reflect diminished well-being and may be predictive of risk of more serious neurological problems in the future.
Mental health in chronic poisoning
Neuropsychiatric disorders have long been attributed to exposure to neurotoxic substances. Clinical descriptions range from affective disorders, including anxiety and depression, to manifestations of psychotic behaviour and hallucinations. Acute high-level exposure to many heavy metals, organic solvents and pesticides can produce delirium. “Manganese madness” has been described in persons with long-term exposure to manganese, and the well-known “mad hatter” syndrome results from mercury intoxication. Type 2a Toxic Encephalopathy, characterized by sustained change in personality involving fatigue, emotional lability, impulse control and general mood and motivation, has been associated with organic solvent exposure. There is growing evidence from clinical and population studies that personality disorders persist over time, long after exposure ceases, although other types of impairment may improve.
On the continuum from well-being to disease, mood changes, irritability and excessive fatigue are often the very first indications of over-exposure to neurotoxic substances. Although neuropsychiatric symptoms are routinely surveyed in worksite studies, these are rarely presented as a mental health problem with potential consequences on mental and social well-being. For example, changes in mental health status affect one’s behaviour, contributing to difficult inter-personal relationships and disagreements in the home; these in turn can aggravate one’s mental state. In workplaces with employee aid programmes, designed to help employees with personal problems, ignorance of the potential mental health effects of exposure to neurotoxic substances can lead to treatment dealing with the effects rather than the cause. It is interesting to note that among the many reported outbreaks of “mass hysteria” or psychogenic illness, industries with exposure to neurotoxic substances are over-represented. It is possible that these substances, which, for the large part, went unmeasured, contributed to the reported symptoms.
Mental health manifestations of neurotoxin exposure can be similar to those that are caused by psychosocial stressors associated with poor work organization, as well as psychological reactions to accidents, very stressful occurrences and severe intoxications, called post-traumatic stress disorder (as discussed elsewhere in this Encyclopaedia). A good understanding of the relation between mental health problems and working conditions is important to initiating adequate preventive and curative actions.
General considerations in assessing early neurotoxicdysfunction
When evaluating early nervous system dysfunction among active workers, a number of factors must be taken into account. Firstly, many of the neuropsychological and neurophysiological functions that are examined diminish with age; some are influenced by culture or educational level. These factors must be taken into account when considering the relation between exposure and nervous system alterations. This can be done by comparing groups with similar socio-demographic status or by using statistical methods of adjustment. There are, however, certain pitfalls that should be avoided. For example, older workers may have longer work histories, and it has been suggested that some neurotoxic substances may accelerate ageing. Job segregation may confine poorly educated workers, women and minorities in jobs with higher exposures. Secondly, alcohol consumption, smoking and drugs, which all contain neurotoxic substances, may also affect symptoms and performance. A good understanding of the workplace is important in unravelling the different factors that contribute to nervous system dysfunction and the implementation of preventive measures.
Preventing Neurotoxicity at Work
A worker not exposed to a neurotoxic substance will never develop any adverse neurotoxic health effects from that substance. Zero exposure leads to total protection against neurotoxic health effects. This is the essence of all primary prevention measures.
Toxicity Testing
New chemical compounds introduced into the workplace and in occupational settings should have already been tested for neurotoxicity. Failure to do pre-market toxicity testing can lead to workers’ contact and potentially severe adverse health effects. The introduction of methyl n-butyl ketone into a workplace in the United States is a classic example of the possible hazards of untested neurotoxicants being introduced into the workplace (Spencer and Schaumburg 1980).
Engineering Controls
Engineering controls (e.g., ventilation systems, closed production facilities) are the best means for keeping workers’ exposures below permissible exposure limits. Closed chemical processes that keep all toxicants from being released into the workplace environment are the ideal. If this is not possible, closed ventilation systems that exhaust ambient air vapours and are designed so as to pull contaminated air away from workers are useful when well designed, adequately maintained, and properly operated.
Personal Protection Equipment
In situations where engineering controls are unavailable to reduce workers’ contact with neurotoxicants, personal protective equipment must be provided. Because workplace neurotoxicants are many, and routes of exposure differ across workplaces and work conditions, the kind of personal protective equipment must be carefully selected for the situation at hand. For example, the neurotoxicant lead can exert its toxicity when lead-laden dust is breathed and when lead particles are ingested in food or water. Therefore, personal protective equipment must protect against both routes of exposure. This would mean respiratory protection equipment and adoption of personal hygiene measures to prevent consumption of lead-contaminated food or beverages. For many neurotoxicants (like industrial solvents), absorption of the substance through intact skin is a main route of exposure. Impermeable gloves, aprons and other appropriate equipment must therefore be provided to prevent skin absorption. This would be in addition to engineering controls or personal respiratory protection equipment. Considerable planning must be given to match personal protective equipment to the specific work being performed.
Administrative Controls
Administrative controls consist of managerial efforts to reduce workplace hazards through planning, training, employee rotation on job sites, changes in production processes, and product substitution (Urie 1992), as well as strict adherence to all existing regulations.
Workers’ Right-to-Know
While the employer bears the responsibility for providing a workplace or work experience that does not harm workers’ health, workers have the responsibility to follow workplace rules that are intended to protect them. Workers must be in a position to know what actions to take in protecting themselves. This means workers have the right to know about the neurotoxicity of substances with which they come into contact, and what protective measures they can take.
Worker Health Surveillance
Where conditions permit, workers should be regularly given medical examinations. A regular evaluation by occupational physicians or other medical specialists constitutes worker health surveillance. For workers known to be working with or around neurotoxicants, physicians should be knowledgeable of the effects of exposure. For example, low-level exposure to many organic solvents will produce symptoms of fatigue, sleep disorders, headaches and memory disturbances. For heavy doses of lead, wrist drop and peripheral nerve impairment would be signs of lead intoxication. Any signs and symptoms of neurotoxicant intoxication should result in reassignment of the worker to an area free of the neurotoxicant, and efforts to reduce workplace levels of the neurotoxicant.
Clinical Syndromes Associated with Neurotoxicity
Neurotoxicant syndromes, brought about by substances which adversely affect nervous tissue, constitute one of the ten leading occupational disorders in the United States. Neurotoxicant effects constitute the basis for establishing exposure limit criteria for approximately 40% of agents considered hazardous by the United States National Institute for Occupational Safety and Health (NIOSH).
A neurotoxin is any substance capable of interfering with the normal function of nervous tissue, causing irreversible cellular damage and/or resulting in cellular death. Depending on its particular properties, a given neurotoxin will attack selected sites or specific cellular elements of the nervous system. Those compounds, which are non-polar, have greater lipid solubility, and thus have greater access to nervous tissue than highly polar and less lipid-soluble chemicals. The type and size of cells and the various neurotransmitter systems affected in different regions of the brain, innate protective detoxifying mechanisms, as well as the integrity of cellular membranes and intracellular organelles all influence neurotoxicant responses.
Neurons (the functional cell unit of the nervous system) have a high metabolic rate and are at greatest risk for neurotoxicant damage, followed by oligodendrocytes, astrocytes, microglia and cells of the capillary endothelium. Changes in cellular membrane structure impair excitability and impede impulse transmission. Toxicant effects alter protein form, fluid content and ionic exchange capability of membranes, leading to swelling of neurons, astrocytes and damage to the delicate cells lining blood capillaries. Disruption of neurotransmitter mechanisms block access to post-synaptic receptors, produce false neurotransmitter effects, and alter the synthesis, storage, release, re-uptake or enzymatic inactivation of natural neurotransmitters. Thus, clinical manifestations of neurotoxicity are determined by a number of different factors: the physical characteristics of the neurotoxicant substance, the dose of exposure to it, the vulnerability of the cellular target, the organism’s ability to metabolize and excrete the toxin, and by the reparative abilities of the structures and mechanisms affected. Table 1 lists various chemical exposures and their neurotoxic syndromes.
Table 1. Chemical exposures and associated neurotoxic syndromes
|
Neurotoxin |
Sources of exposure |
Clinical diagnosis |
Locus of pathology1 |
|
Metals |
|||
|
Arsenic |
Pesticides; pigments; antifouling paint; electroplating industry; seafood; smelters; semiconductors |
Acute: encephalopathy Chronic: peripheral neuropathy |
Unknown (a) Axon (c) |
|
Lead |
Solder; lead shot; illicit whiskey; insecticides; auto body shop; storage battery manufacturing; foundries, smelters; lead-based paint; lead pipes |
Acute: encephalopathy Chronic: encephalopathy and peripheral neuropathy |
Blood vessels (a) Axon (c) |
|
Manganese |
Iron, steel industry; welding operations; metal-finishing operations; fertilizers; manufacturers of fireworks, matches; manufacturers of dry cell batteries |
Acute: encephalopathy Chronic: parkinsonism |
Unknown (a) Basal ganglia neurons (c) |
|
Mercury |
Scientific instruments; electrical equipment; amalgams; electroplating industry; photography; felt making |
Acute: headache, nausea, onset of tremor Chronic: ataxia, peripheral neuropathy, encephalopathy |
Unknown (a) Axon (c) Unknown (c) |
|
Tin |
Canning industry; solder; electronic components; polyvinyl plastics; fungicides |
Acute: memory defects, seizures, disorientation Chronic: encephalomyelopathy |
Neurons of the limbic system (a & c) Myelin (c) |
|
Solvents |
|||
|
Carbon disulphide |
Manufacturers of viscose rayon; preservatives; textiles; rubber cement; varnishes; electroplating industry |
Acute: encephalopathy Chronic: peripheral neuropathy, parkinsonism |
Unknown (a) Axon (c) Unknown |
|
n-hexane, methyl butyl ketone |
Paints; lacquers; varnishes; metal-cleaning compounds; quick-drying inks; paint removers; glues, adhesives |
Acute: narcosis Chronic: peripheral neuropathy, unknown (a) Axon (c), |
|
|
Perchloroethylene |
Paint removers; degreasers; extraction agents; dry cleaning industry; textile industry |
Acute: narcosis Chronic: peripheral neuropathy, encephalopathy |
Unknown (a) Axon (c) Unknown |
|
Toluene |
Rubber solvents; cleaning agents; glues; manufacturers of benzene; gasoline, aviation fuels; paints, paint thinners; lacquers |
Acute: narcosis Chronic: ataxia, encephalopathy |
Unknown (a) Cerebellum (c) Unknown |
|
Trichloroethylene |
Degreasers; painting industry; varnishes; spot removers; process of decaffeination; dry cleaning industry; rubber solvents |
Acute: narcosis Chronic: encephalopathy, cranial neuropathy |
Unknown (a) Unknown (c) Axon (c) |
|
Insecticides |
|||
|
Organophosphates |
Agricultural industry manufacturing and application |
Acute: cholinergic poisoning Chronic: ataxia, paralysis, peripheral neuropathy |
Acetylcholinesterase (a) Long tracts of spinal cord (c) Axon (c) |
|
Carbamates |
Agricultural industry manufacturing and application flea powders |
Acute: cholinergic poisoning Chronic: tremor, peripheral neuropathy |
Acetylcholinesterase (a) Dopaminergic system (c) |
1 (a), acute; (c), chronic.
Source: Modified from Feldman 1990, with permission of the publisher.
Establishing a diagnosis of a neurotoxicant syndrome and differentiating it from neurologic diseases of non-neurotoxicant aetiology requires an understanding of the pathogenesis of the neurological symptoms and observed signs and symptoms; an awareness that particular substances are capable of affecting nervous tissue; documentation of exposure; evidence of presence of neurotoxin and/or metabolites in tissues of an affected individual; and careful delineation of a time relationship between exposure and the appearance of symptoms with subsequent decrease in symptoms after exposure is ended.
Proof that a particular substance has reached a toxicant dose level is usually lacking after symptoms appear. Unless environmental monitoring is ongoing, a high index of suspicion is necessary to recognize cases of neurotoxicologic injury. Identifying symptoms referable to the central and/or the peripheral nervous systems can help the clinician focus on certain substances, which have a greater predilection for one part or another of the nervous system, as possible culprits. Convulsions, weakness, tremor/twitching, anorexia (weight loss), equilibrium disturbance, central nervous system depression, narcosis (a state of stupor or unconsciousness), visual disturbance, sleep disturbance, ataxia (inability to coordinate voluntary muscle movements), fatigue and tactile disorders are commonly reported symptoms following exposure to certain chemicals. Constellations of symptoms form syndromes associated with neurotoxicant exposure.
Behavioural Syndromes
Disorders with predominantly behavioural features ranging from acute psychosis, depression and chronic apathy have been described in some workers. It is essential to differentiate memory impairment associated with other neurological diseases, such as Alzheimer’s disease, arteriosclerosis or presence of a brain tumour, from the cognitive deficits associated with toxicant exposure to organic solvents, metals or insecticides. Transient disturbances of awareness or epileptic seizures with or without associated motor involvement must be identified as a primary diagnosis separate from similarly appearing disturbances of consciousness related to neurotoxicant effects. Subjective and behavioural toxicant syndromes such as headache, vertigo, fatigue and personality change manifest as mild encephalopathy with inebriation, and may indicate the presence of exposure to carbon monoxide, carbon dioxide, lead, zinc, nitrates or mixed organic solvents. Standardized neuropsychological testing is necessary to document elements of cognitive impairment in patients suspected of toxicant encephalopathy, and these must be differentiated from those dementing syndromes caused by other pathologies. Specific tests used in the diagnostic batteries of tests must include a broad sampling of cognitive function tests which will generate predictions about the patient’s functioning and daily life, as well as tests which have been demonstrated previously to be sensitive to the effects of known neurotoxins. These standardized batteries must include tests which have been validated on patients with specific types of brain damage and structural deficits, to clearly separate these conditions from neurotoxic effects. In addition, tests must include internal control measures to detect the influence of motivation, hypochondriasis, depression and learning difficulties, and must contain language that takes into account cultural as well as educational background effects.
A continuum exists from mild to severe central nervous system impairment experienced by patients exposed to toxicant substances:
- Organic affective syndrome (Type I Effect), in which mild mood disorders predominate as the patient’s chief complaint, with features most consistent with those of organic affective disorders of the depressive type. This syndrome seems to be reversible following cessation of exposure to the offending agent.
- Mild chronic toxicant encephalopathy, in which, in addition to mood disturbances, central nervous system impairment is more prominent. Patients have evidence of memory and psychomotor function disturbance which can be confirmed by neuropsychological testing. In addition, features of visual spatial impairment and abstract concept formation may be seen. Activities of daily living and work performance are impaired.
- Sustained personality or mood change (Type IIA Effect) or impairment in intellectual function (Type II) may be seen. In mild chronic toxicant encephalopathy, the course is insidious. Features may persist after the cessation of exposure and disappear gradually, while in some individuals, persistent functional impairment may be observed. If exposure continues, the encephalopathy may progress to a more severe stage.
- In severe chronic toxicant encephalopathy (Type III Effect) dementia with global deterioration of memory and other cognitive problems are noted. The clinical effects of toxicant encephalopathy are not specific to a given agent. Chronic encephalopathy associated with toluene, lead and arsenic is not different from that of other toxicant aetiologies. The presence of other associated findings, however (visual disturbances with methyl alcohol), may help differentiate syndromes according to particular chemical aetiologies.
Workers exposed to solvents for long periods of time may exhibit disturbances of central nervous system function which are permanent. Since an excess of subjective symptoms, including headache, fatigue, impaired memory, loss of appetite and diffuse chest pains, have been reported, it is often difficult to confirm this effect in any individual case. An epidemiological study comparing house painters exposed to solvents with unexposed industrial workers showed, for example, that painters had significantly lower mean scores on psychological tests measuring intellectual capacity and psychomotor coordination than referent subjects. The painters also had significantly lower performances than expected on memory and reaction time tests. Differences between workers exposed for several years to jet fuel and unexposed workers, in tests demanding close attention and high sensory motor speed, were apparent as well. Impairments in psychological performance and personality changes have also been reported among car painters. These included visual and verbal memory, reduction of emotional reactivity, and poor performance on verbal intelligence tests.
Most recently, a controversial neurotoxicant syndrome, multiple chemical sensitivity, has been described. Such patients develop a variety of features involving multiple organ systems when they are exposed to even low levels of various chemicals found in the workplace and the environment. Mood disturbances are characterized by depression, fatigue, irritability and poor concentration. These symptoms reoccur on exposure to predictable stimuli, by elicitation by chemicals of diverse structural and toxicological classes, and at levels much lower than those causing adverse responses in the general population. Many of the symptoms of multiple chemical sensitivity are shared by individuals who show only a mild form of mood disturbance, headache, fatigue, irritability and forgetfulness when they are in a building with poor ventilation and with off-gassing of volatile substances from synthetic building materials and carpets. The symptoms disappear when they leave these environments.
Disturbances of consciousness, seizures and coma
When the brain is deprived of oxygen—for example, in the presence of carbon monoxide, carbon dioxide, methane or agents which block tissue respiration such as hydrocyanic acid, or those which cause massive impregnation of the nerve such as certain organic solvents—disturbances of consciousness may result. Loss of consciousness may be preceded by seizures in workers with exposure to anticholinesterase substances such as organophosphate insecticides. Seizures may also occur with lead encephalopathy associated with brain swelling. Manifestations of acute toxicity following organophosphate poisoning have autonomic nervous system manifestations which precede the occurrence of dizziness, headache, blurred vision, myosis, chest pain, increased bronchial secretions, and seizures. These parasympathetic effects are explained by the inhibitory action of these toxicant substances on cholinesterase activity.
Movement disorders
Slowness of movement, increased muscle tone, and postural abnormalities have been observed in workers exposed to manganese, carbon monoxide, carbon disulphide and the toxicity of a meperidine by-product, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). At times, the individuals may appear to have Parkinson’s disease. Parkinsonism secondary to toxicant exposure has features of other nervous disorders such as chorea and athetosis. The typical “pill-rolling” tremor is not seen in these instances, and usually the cases do not respond well to the drug levodopa. Dyskinesia (impairment of the power of voluntary motion) can be a common symptom of bromomethane poisoning. Spasmodic movements of the fingers, face, peribuccal muscles and the neck, as well as extremity spasms, may be seen. Tremor is common following mercury poisoning. More obvious tremor associated with ataxia (lack of coordination of muscular action) is noted in individuals following toluene inhalation.
Opsoclonus is an abnormal eye movement which is jerky in all directions. This is often seen in brain-stem encephalitis, but may also be a feature following chlordecone exposure. The abnormality consists of irregular bursts of abrupt, involuntary, rapid, simultaneous jerking of both eyes in a conjugate manner, possibly multidirectional in severely affected individuals.
Headache
Common complaints of head pain following exposure to various metal fumes such as zinc and other solvent vapours may result from vasodilation (widening of the blood vessels), as well as cerebral oedema (swelling). The experiencing of pain is common to these conditions, as well as carbon monoxide, hypoxia (low oxygen), or carbon dioxide conditions. “Sick building syndrome” is thought to cause headaches because of excess carbon dioxide present in a poorly ventilated area.
Peripheral neuropathy
Peripheral nerve fibres serving motor functions begin in motor neurons in the ventral horn of the spinal cord. The motor axons extend peripherally to the muscles they innervate. A sensory nerve fibre has its nerve cell body in the dorsal root ganglion or in the dorsal grey matter of the spinal cord. Having received information from the periphery detected at distal receptors, nerve impulses are conducted centrally to the nerve cell bodies where they connect with spinal cord pathways transmitting information to the brain stem and cerebral hemispheres. Some sensory fibres have immediate connections with motor fibres within the spinal cord, providing a basis for reflex activity and quick motor responses to noxious sensations. These sensory-motor relationships exist in all parts of the body; the cranial nerves are the peripheral nerve equivalents arising in brain stem, rather than spinal cord, neurons. Sensory and motor nerve fibres travel together in bundles and are referred to as the peripheral nerves.
Toxicant effects of peripheral nerve fibres may be divided into those which primarily affect axons (axonopathies), those which are involved in distal sensory-motor loss, and those which primarily affect myelin sheath and Schwann cells. Axonopathies are evident in early stages in the lower extremities where the axons are the longest and farthest from the nerve cell body. Random demyelination occurs in segments between nodes of Ranvier. If sufficient axonal damage occurs, secondary demyelination follows; as long as axons are preserved, regeneration of Schwann cells and remyelination can occur. A pattern seen commonly in toxicant neuropathies is distal axonopathy with secondary segmental demyelination. The loss of myelin reduces the speed of conducting nerve impulses. Thus, gradual onset of intermittent tingling and numbness progressing to lack of sensation and unpleasant sensations, muscle weakness, and atrophy results from damage to the motor and sensory fibres. Reduced or absent tendon reflexes and anatomically consistent patterns of sensory loss, involving the lower extremities more than upper, are features of peripheral neuropathy.
Motor weaknesses may be noted in distal extremities and progress to unsteady gait and inability to grasp objects. The distal portions of the extremities are involved to a greater extent, but severe cases may produce proximal muscle weakness or atrophy as well. Extensor muscle groups are involved before the flexors. Symptoms may sometimes progress for a few weeks even after removal from exposure. Deterioration of nerve function may persist for several weeks after removal from exposure.
Depending on the type and severity of neuropathy, an electrophysiological examination of the peripheral nerves is useful to document impaired function. Slowing of conduction velocity, reduced amplitudes of sensory or motor action potentials, or prolonged latencies can be observed. Slowing of motor or sensory conduction velocities is generally associated with demyelination of nerve fibres. Preservation of normal conduction velocity values in the presence of muscle atrophy suggests axonal neuropathy. Exceptions occur when there is progressive loss of motor and sensory nerve fibres in axonal neuropathy which affects the maximal conduction speed as a result of the dropping out of the larger diameter faster conducting nerve fibres. Regenerating fibres occur in early stages of recovery in axonopathies, in which conduction is slowed, especially in the distal segments. The electrophysiological study of patients with toxicant neuropathies should include measurements of motor and sensory conduction velocity in the upper and lower extremities. Special attention should be given to the primarily sensory conducting characteristics of the sural nerve in the leg. This is of great value when the sural nerve is then used for biopsy, providing anatomical correlation between the histology of teased nerve fibres and the conduction characteristics. A differential electrophysiological study of the conducting capabilities of proximal segments versus distal segments of a nerve is useful in identifying a distal toxicant axonopathy, or to localize a neuropathic block of conduction, probably due to demyelination.
Understanding the pathophysiology of a suspected neurotoxicant polyneuropathy has great value. For example, in patients with neuropathy caused by n-hexane and methylbutyl ketone, motor nerve conduction velocities are reduced, but in some cases, the values may fall within the normal range if only the fastest firing fibres are stimulated and used as the measured outcome. Since neurotoxicant hexacarbon solvents cause axonal degeneration, secondary changes arise in myelin and explain overall reduction in conduction velocity despite the value within the normal range produced by the preserved conducting fibres.
Electrophysiological techniques include special tests other than the direct conduction velocity, amplitude and latency studies. Somatosensory evoked potentials, auditory evoked potentials, and visual evoked potentials are ways of studying the characteristics of the sensory conducting systems, as well as specific cranial nerves. Afferent-efferent circuitry can be tested by using blink reflex tests involving the 5th cranial nerve to 7th cranial innervated muscle responses; H-reflexes involve segmental motor reflex pathways. Vibration stimulation selects out larger fibres from smaller fibre involvements. Well-controlled electronic techniques are available for measuring the threshold needed to elicit a response, and then to determine the speed of travel of that response, as well as the amplitude of the muscle contraction, or the amplitude and pattern of an evoked sensory action potential. All physiological results must be evaluated in light of the clinical picture and with an understanding of the underlying pathophysiological process.
Conclusion
The differentiation of a neurotoxicant syndrome from a primary neurological disease poses a formidable challenge to physicians in the occupational setting. Obtaining a good history, maintaining a high degree of suspicion and adequate follow-up of an individual, as well as groups of individuals, is necessary and rewarding. Early recognition of illness related to toxicant agents in their environment or to a particular occupational exposure is critical, since proper diagnosis can lead to early removal of an individual from the hazards of ongoing exposure to a toxicant substance, preventing possible irreversible neurological damage. Furthermore, recognition of the earliest affected cases in a particular setting may result in changes that will protect others who have not yet become affected.
Measuring Neurotoxic Deficits
Neuro-functional Test Batteries
Sub-clinical neurologic signs and symptoms have long been noted among active workers exposed to neurotoxins; however, it is only since the mid-1960s that research efforts have focused on the development of sensitive test batteries capable of detecting subtle, mild changes that are present in the early stages of intoxication, in perceptual, psychomotor, cognitive, sensory and motor functions, and affect.
The first neurobehavioural test battery for use in worksite studies was developed by Helena Hänninen, a pioneer in the field of neurobehavioural deficits associated with toxic exposure (Hänninen Test Battery) (Hänninen and Lindstrom 1979). Since then, there have been worldwide efforts to develop, refine and, in some cases, computerize neurobehavioural test batteries. Anger (1990) describes five worksite neurobehavioural test batteries from Australia, Sweden, Britain, Finland and the United States, as well as two neurotoxic screening batteries from the United States, that have been used in studies of neurotoxin-exposed workers. In addition, the computerized Neurobehavioral Evaluation System (NES) and the Swedish Performance Evaluation System (SPES) have been extensively used around the world. There are also test batteries designed to assess sensory functions, including measures of vision, vibrotactile perception threshold, smell, hearing and sway (Mergler 1995). Studies of various neurotoxic agents using one or another of these batteries have greatly contributed to our knowledge of early neurotoxic impairment; however, cross-study comparisons have been difficult since different tests are used and tests with similar names may be administered using a different protocol.
In an attempt to standardize information from studies on neurotoxic substances, the notion of a “core” battery was put forward by a working committee of the World Health Organization (WHO) (Johnson 1987). Based on knowledge at the time of the meeting (1985), a series of tests were selected to make up the Neurobehavioral Core Test Battery (NCTB), a relatively inexpensive, hand-administered battery, which has been successfully used in many countries (Anger et al. 1993). The tests that make up this battery were chosen to cover specific nervous system domains, which had been previously shown to be sensitive to neurotoxic damage. A more recent core battery, which comprises both hand-administered and computerized tests, has been proposed by a workgroup of the United States Agency for Toxic Substances and Disease Registry (Hutchison et al. 1992). Both batteries are presented in Table 1.
Table 1. Examples of "core" batteries for assessment of early neurotoxic effects
|
Neurobehavioural Core Test Battery (NCTB)+ |
Test order |
Agency for Toxic Substances and Disease Registry Adult Environmental Neurobehavioural Test Battery (AENTB)+ |
||
|
Functional domain |
Test |
Functional domain |
Test |
|
|
Motor steadiness |
Aiming (Pursuit Aiming II) |
1 |
Vision |
Visual acuity, near contrast sensitivity |
|
Attention/response speed |
Simple Reaction Time |
2 |
Colour vision (Lanthony D-15 desaturated test) |
|
|
Perceptual motor speed |
Digit Symbol (WAIS-R) |
3 |
Somatosensory |
Vibrotactile perception threshold |
|
Manual dexterity |
Santa Ana (Helsinki Version) |
4 |
Motor strength |
Dynamometer (including fatigue assessment) |
|
Visual perception/memory |
Benton Visual Retention |
5 |
Motor coordination |
Santa Ana |
|
Auditory memory |
Digit Span (WAIS-R, WMS) |
6 |
Higher intellectual function |
Raven Progressive Matrices (Revised) |
|
Affect |
POMS (Profile of Mood States) |
7 |
Motor coordination |
Fingertapping Test (one hand)1 |
|
8 |
Sustained attention (cognitive), speed (motor) |
Simple Reaction Time (SRT) (extended)1 |
||
|
9 |
Cognitive coding |
Symbol-digit with delayed recall1 |
||
|
10 |
Learning and memory |
Serial Digit Learning1 |
||
|
11 |
Index of educational level |
Vocabulary1 |
||
|
12 |
Mood |
Mood Scale1 |
||
1 Available in computerized version; WAIS = Wechsler Adult Intelligence Scale; WMS = Wechsler Memory Scale.
The authors of both core batteries stress that, although the batteries are useful to standardize results, they by no means provide complete assessment of nervous system functions. Additional tests should be used depending upon the type of exposure; for example, a test battery to assess nervous system dysfunction among manganese-exposed workers would include more tests of motor functions, particularly those that require rapid alternating movements, while one for methylmercury-exposed workers would include visual field testing. The choice of tests for any particular workplace should be made on the basis of current knowledge on the action of the particular toxin or toxins to which the persons are exposed.
More sophisticated test batteries, administered and interpreted by trained psychologists, are an important part of the clinical assessment for neurotoxic poisoning (Hart 1988). It includes tests of intellectual ability, attention, concentration and orientation, memory, visuo-perceptive, constructive and motor skills, language, conceptual and executive functions, and psychological well-being, as well as an assessment of possible malingering. The profile of the patient’s performance is examined in the light of past and present medical and psychological history, as well as exposure history. The final diagnosis is based on a constellation of deficits interpreted in relation to the type of exposure.
Measures of Emotional State and Personality
Studies of the effects of neurotoxic substances usually include measures of affective or personality disturbance, in the form of symptoms questionnaires, mood scales or personality indices. The NCTB, described above, includes the Profile of Mood States (POMS), a quantitative measure of mood. Using 65 qualifying adjectives of mood states over the past 8 days, the degrees of tension, depression, hostility, vigour, fatigue and confusion are derived. Most comparative workplace studies of neurotoxic exposure indicate differences between exposed and non-exposed. A recent study of styrene-exposed workers shows dose-response relations between post-shift urinary mandelic acid level, a biological indicator of styrene, and scale scores of tension, hostility, fatigue and confusion (Sassine et al. 1996).
Lengthier and more sophisticated tests of affect and personality, such as the Minnesota Multiphasic Personality Index (MMPI), which reflect both emotional states and personality traits, have been used primarily for clinical evaluation, but also in workplace studies. The MMPI likewise provides an assessment of symptom exaggeration and inconsistent responses. In a study of microelectronics workers with a history of exposure to neurotoxic substances, results from the MMPI indicated clinically significant levels of depression, anxiety, somatic concerns and disturbances of thinking (Bowler et al. 1991).
Electrophysiological Measures
Electrical activity generated by the transmission of information along nerve fibres and from one cell to another, can be recorded and used in the determination of what is happening in the nervous system of persons with toxic exposures. Interference with neuronal activity can slow down transmission or modify the electrical pattern. Electrophysiological recordings require precise instruments and are most frequently carried out in a laboratory or hospital setting. There have, however, been efforts to develop more portable equipment for use in workplace studies.
Electrophysiological measures record a global response of a large number of nerve fibres and/or fibres, and a fair amount of damage must exist before it can be adequately recorded. Thus, for most neurotoxic substances, symptoms, as well as sensory, motor and cognitive changes, usually can be detected in groups of exposed workers before electrophysiological differences are observed. For clinical examination of persons with suspected neurotoxic disorders, electrophysiological methods provide information concerning the type and extent of nervous system damage. A review of electrophysiological techniques used in the detection of early neurotoxicity in humans is provided by Seppalaïnen (1988).
The nerve conduction velocity of sensory nerves (going towards the brain) and motor nerves (going away from the brain) are measured by electroneurography (ENG). By stimulating at different anatomical positions and recording at another, the conduction velocity can be calculated. This technique can provide information about the large myelinated fibres; slowing of conduction velocity occurs when demyelination is present. Reduced conduction velocities have frequently been observed among lead-exposed workers, in the absence of neurological symptoms (Maizlish and Feo 1994). Slow conduction velocities of peripheral nerves have also been associated with other neurotoxins, such as mercury, hexacarbons, carbon disulphide, styrene, methyl-n-butyl ketone, methyl ethyl ketone, and certain solvent mixtures. The trigeminal nerve (a facial nerve) is affected by trichloroethylene exposure. However, if the toxic substance acts primarily on thinly myelinated or unmyelinated fibres, conduction velocities usually remain normal.
Electromyography (EMG) is used for measuring the electrical activity in muscles. Electromyographic abnormalities have been observed among workers with exposure to such substances as n-hexane, carbon disulphide, methyl-n-butyl ketone, mercury and certain pesticides. These changes are often accompanied by changes in ENG and symptoms of peripheral neuropathy.
Changes in brainwaves are evidenced by electroencephalography (EEG). In patients with organic solvent poisoning, local and diffuse slow wave abnormalities have been observed. Some studies report evidence of dose-related EEG alterations among active workers, with exposure to organic solvent mixtures, styrene and carbon disulphide. Organochlorine pesticides can cause epileptic seizures, with EEG abnormalities. EEG changes have been reported with long-term exposure to organophosphorus and zinc phosphide pesticides.
Evoked potentials (EP) provides another means of examining nervous system activity in response to a sensory stimulus. Recording electrodes are placed on the specific area of the brain that responds to the particular stimuli, and the latency and amplitude of the event-related slow potential are recorded. Increased latency and/or reduced peak amplitudes have been observed in response to visual, auditory and somatosensory stimuli for a wide range of neurotoxic substances.
Electrocardiography (ECG or EKG) records changes in the electrical conduction of the heart. Although it is not often used in studies of neurotoxic substances, changes in ECG waves have been observed among persons with exposure to trichloroethylene. Electro-oculographic (EOG) recordings of eye movements have shown alterations among workers with exposure to lead.
Brain Imaging Techniques
In recent years, different techniques have been developed for brain imaging. Computed tomographic (CT) images reveal the anatomy of the brain and spinal cord. They have been used to study cerebral atrophy among solvent-exposed workers and patients; however, the results are not consistent. Magnetic resonance imaging (MRI) examines the nervous system using a powerful magnetic field. It is particularly useful clinically to rule out an alternative diagnosis, such as brain tumours. Positron Emission Tomography (PET), which yields images of biochemical processes, has been successfully used to study changes in the brain induced by manganese intoxication. Single photon emission computed tomography (SPECT) provides information about brain metabolism and may prove to be an important tool in understanding how neurotoxins act on the brain. These techniques are all very costly, and not readily available in most hospitals or laboratories throughout the world.
Diagnosis
The diagnosis of neurotoxic disease is not easy. The errors are usually of two types: either it is not recognized that a neurotoxic agent is the cause of neurological symptoms, or neurological (and especially neurobehavioural) symptoms are erroneously diagnosed as resulting from an occupational, neurotoxic exposure. Both of these errors can be hazardous since an early diagnosis is important in the case of neurotoxic disease, and the best treatment is avoiding further exposure for the individual case and the surveillance of the condition of other workers in order to prevent their exposure to the same danger. On the other hand, sometimes undue alarm can develop in the workplace if a worker claims to have serious symptoms and suspects a chemical exposure as the cause but in fact, either the worker is mistaken or the hazard is not actually present for others. There is practical reason for correct diagnostic procedures, as well, since in many countries, the diagnosis and treatment of occupational diseases and the loss of working capacity and invalidity caused by those diseases are covered by insurance; thus the financial compensation may be disputed, if the diagnostic criteria are not solid. An example of a decision tree for neurological assessment is given in Table 1.
Table 1. Decision tree for neurotoxic disease
I. Relevant exposure level, length and type
II. Appropriate symptoms insidiously increasing central (CNS) or peripheral (PNS) nervous system symptoms
III. Signs and additional tests CNS dysfunction: neurology, psychology tests PNS dysfunction: quantitative sensory test, nerve conduction studies
IV. Other diseases excluded in differential diagnosis
Exposure and Symptoms
Acute neurotoxic syndromes occur mainly in accidental situations, when workers are exposed short-term to very high levels of a chemical or to a mixture of chemicals generally through inhalation. The usual symptoms are vertigo, malaise and possible loss of consciousness as a result of depression of the central nervous system. When the subject is removed from the exposure, the symptoms disappear rather quickly, unless the exposure has been so intense that it is life-threatening, in which case coma and death may follow. In these situations recognition of the hazard must occur at the workplace, and the victim should be taken out into the fresh air immediately.
In general, neurotoxic symptoms arise after short-term or long-term exposures, and often at relatively low-level occupational exposure levels. In these cases acute symptoms may have occurred at work, but the presence of acute symptoms is not necessary for diagnosis of chronic toxic encephalopathy or toxic neuropathy to be made. However, patients do often report headache, light-headedness or mucosal irritation at the end of a working day, but these symptoms initially disappear during the night, weekend or vacation. A useful checklist can be found in Table 2.
Table 2. Consistent neuro-functional effects of worksite exposures to some leading neurotoxic substances
|
Mixed organic solvents |
Carbon disulphide |
Styrene |
Organophos- |
Lead |
Mercury |
|
|
Acquisition |
+ |
|
|
|
+ |
|
|
Affect |
+ |
|
+ |
|
+ |
|
|
Categorization |
+ |
|
|
|
|
|
|
Coding |
+ |
+ |
|
|
+ |
+ |
|
Colour vision |
+ |
|
+ |
|
|
|
|
Concept shifting |
+ |
|
|
|
|
|
|
Distractibility |
|
|
|
|
+ |
|
|
Intelligence |
+ |
+ |
|
+ |
+ |
+ |
|
Memory |
+ |
+ |
+ |
+ |
+ |
+ |
|
Motor coordination |
+ |
+ |
+ |
|
+ |
+ |
|
Motor speed |
+ |
+ |
+ |
|
+ |
+ |
|
Near visual contrast sensitivity |
+ |
|
|
|
|
|
|
Odour perception threshold |
+ |
|
|
|
|
|
|
Odour identification |
+ |
|
|
|
+ |
|
|
Personality |
+ |
+ |
|
|
|
+ |
|
Spatial relations |
+ |
+ |
|
|
+ |
|
|
Vibrotactile threshold |
+ |
|
|
+ |
|
+ |
|
Vigilance |
+ |
+ |
|
|
+ |
|
|
Visual field |
|
|
|
|
+ |
+ |
|
Vocabulary |
|
|
|
|
+ |
|
Source: Adapted from Anger 1990.
Assuming that the patient has been exposed to neurotoxic chemicals, the diagnosis of neurotoxic disease starts with symptoms. In 1985, a joint working group of the World Health Organization and the Nordic Council of Ministers discussed the matter of chronic organic solvent intoxication and found a set of core symptoms, which are found in most cases (WHO/Nordic Council 1985). The core symptoms are fatigability, memory loss, difficulties in concentration, and loss of initiative. These symptoms usually start after a basic change in personality, which develops gradually and affects energy, intellect, emotion and motivation. Among other symptoms of chronic toxic encephalopathy are depression, dysphoria, emotional lability, headache, irritability, sleep disturbances and dizziness (vertigo). If there is also involvement of the peripheral nervous system, numbness and possibly muscular weakness develop. Such chronic symptoms last for at least a year after the exposure itself has ended.
Clinical Examination and Testing
The clinical examination should include a neurological examination, where attention should be paid to impairment of higher nervous functions, such as memory, cognition, reasoning and emotion; to impaired cerebellar functions, like tremor, gait, station and coordination; and to peripheral nervous functions, especially vibration sensitivity and other tests of sensation. Psychological tests can provide objective measures of higher nervous system functions, including psychomotor, short-term memory, verbal and non-verbal reasoning and perceptual functions. In individual diagnosis the tests should include some tests that give a clue as to the person’s premorbid intellectual level. History of school performance and previous job performance as well as possible psychological tests administered previously, for example in connection with military service, can help in the evaluation of the person’s normal level of performance.
The peripheral nervous system can be studied with quantitative tests of sensory modalities, vibration and thermosensibility. Nerve conduction velocity studies and electromyography can often reveal neuropathy at an early stage. In these tests special emphasis should be on sensory nerve functions. The amplitude of the sensory action potential (SNAP) decreases more often than the sensory conduction velocity in axonal neuropathies, and most toxic neuropathies are axonal in character. Neuroradiological studies such as computed tomography (CT) and magnetic resonance imaging (MRI) usually do not reveal anything pertinent to chronic toxic encephalopathy, but they may be useful in the differential diagnosis.
In the differential diagnosis other neurological and psychiatric diseases should be considered. Dementia of other aetiology should be ruled out, as well as depression and stress symptoms of various causes. Psychiatric consultation may be necessary. Alcohol abuse is a relevant confounding factor; excessive use of alcohol causes symptoms similar to those of solvent exposure, and on the other hand there are papers indicating that solvent exposure may induce alcohol abuse. Other causes for neuropathy also have to be ruled out, especially entrapment neuropathies, diabetes and kidney disease; also alcohol causes neuropathy. The combination of encephalopathy and neuropathy is more likely of toxic origin than either of these alone.
In the final decision the exposure should be evaluated again. Was there relevant exposure, considering the level, length and quality of exposure? Solvents are more likely to induce psycho-organic syndrome or toxic encephalopathy; hexacarbons, however, usually first cause neuropathy. Lead and some other metals cause neuropathy, although CNS involvement can be detected later on.
Occupational Neuroepidemiology
Olav Axelson*
*Adapted from Axelson 1996.
Early knowledge about the neurotoxic effects of occupational exposures appeared through clinical observations. The observed effects were more or less acute and concerned exposure to metals such as lead and mercury or solvents like carbon disulphide and trichloroethylene. With time, however, more chronic and clinically less obvious effects of neurotoxic agents have been assessed through modern examination methods and systematic studies of larger groups. Still, the interpretation of the findings has been controversial and debated such as the chronic effects of solvent exposure (Arlien-Søborg 1992).
The difficulties met in interpreting chronic neurotoxic effects depend on both the diversity and vagueness of symptoms and signs and the associated problem of defining a proper disease entity for conclusive epidemiological studies. For example, in solvent exposure, the chronic effects might include memory and concentration problems, tiredness, lack of initiative, affect liability, irritability, and sometimes dizziness, headache, alcohol intolerance, and reduced libido. Neurophysiological methods have also revealed various functional disturbances, again difficult to condense into any single disease entity.
Similarly, a variety of neurobehavioural effects also seems to occur due to other occupational exposures, such as moderate lead exposure or welding with some exposure to aluminium, lead, and manganese or exposure to pesticides. Again there are also neurophysiological or neurological signs, among others, polyneuropathy, tremor, and disturbance of equilibrium, in individuals exposed to organochlorine, organophosphorus and other insecticides.
In view of the epidemiological problems involved in defining a disease entity out of the many types of neurobehavioural effects referred to, it has also become natural to consider some clinically, more or less well-defined neuropsychiatric disorders in relation to occupational exposures.
Since the 1970s several studies have especially focused on solvent exposure and the psycho-organic syndrome, when of disabling severity. More recently also Alzheimer’s dementia, multiple sclerosis, Parkinson’s disease, amyotrophic lateral sclerosis, and related conditions have attracted interest in occupational epidemiology.
Regarding solvent exposure and the psycho-organic syndrome (or toxic chronic encephalopathy in clinical occupational medicine, when exposure is taken into diagnostic account), the problem of defining a proper disease entity was apparent and first led to considering en bloc the diagnoses of encephalopathia, dementia, and cerebral atrophy, but neurosis, neurasthenia, and nervositas were also included as not necessarily distinct from each other in medical practice (Axelson, Hane and Hogstedt 1976). Recently, more specific disease entities, such as organic dementia and cerebral atrophy, have also been associated with solvent exposure (Cherry, Labréche and McDonald 1992). The findings have not been totally consistent, however, as no excess of “presenile dementia” appeared in a large-scale case-referent study in the United States with as many as 3,565 cases of various neuropsychiatric disorders and 83,245 hospital referents (Brackbill, Maizlish and Fischbach 1990). However, in comparison with bricklayers, there was about a 45% excess of disabling neuropsychiatric disorders among white male painters, except spray painters.
Occupational exposures also seem to play a role for disorders more specific than the psycho-organic syndrome. Hence, in 1982, an association between multiple sclerosis and solvent exposure from glues was first indicated in the Italian shoe industry (Amaducci et al. 1982). This relationship has been considerably strengthened by further studies in Scandinavia (Flodin et al. 1988; Landtblom et al. 1993; Grönning et al. 1993) and elsewhere, so that 13 studies with some information on solvent exposure could be considered in a review (Landtblom et al. 1996). Ten of these studies provided enough data for inclusion in a meta-analysis, showing about a twofold risk for multiple sclerosis among individuals with solvent exposure. Some studies also associate multiple sclerosis with radiological work, welding, and work with phenoxy herbicides (Flodin et al. 1988; Landtblom et al. 1993). Parkinson’s disease seems to be more common in rural areas (Goldsmith et al. 1990), especially at younger ages (Tanner 1989). More interestingly, a study from Calgary, Canada, showed a threefold risk for herbicide exposure (Semchuk, Love and Lee 1992).
All the case persons who recalled specific exposures reported exposure to phenoxy herbicides or thiocarbamates. One of them recalled exposure to paraquat, which is chemically similar to MPTP (N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine), an inducer of a Parkinson-like syndrome. Paraquat workers have not yet been found to suffer from such a syndrome, however (Howard 1979). Case-referent studies from Canada, China, Spain, and Sweden have indicated a relation with exposure to unspecified industrial chemicals, pesticides, and metals, especially manganese, iron and aluminium (Zayed et al. 1990).
In a study from the United States, an increased risk of motor neuron disease (encompassing amyotrophic lateral sclerosis, progressive bulbar palsy and progressive muscular atrophy) appeared in connection with welding and soldering (Armon et al. 1991). Welding also appeared as a risk factor, as did electricity work, and also work with impregnating agents in a Swedish study (Gunnarsson et al. 1992). Hereditability for neurodegenerative and thyroid disease, combined with solvent exposure and male gender, showed a risk as high as 15.6. Other studies also indicate that exposure to lead and solvents could be of importance (Campbell, Williams and Barltrop 1970; Hawkes, Cavanagh and Fox 1989; Chio, Tribolo and Schiffer 1989; Sienko et al. 1990).
For Alzheimer’s disease, no clear indication of any occupational risk appeared in a meta-analysis of eleven case-referent studies (Graves et al. 1991), but more recently an increased risk was connected with blue-collar work (Fratiglioni et al. 1993). Another new study, which included also the oldest ages, indicated that solvent exposure could be a rather strong risk factor (Kukull et al. 1995). The recent suggestion that Alzheimer’s disease might be related to exposure to electromagnetic fields was perhaps even more surprising (Sobel et al. 1995). Both these studies are likely to stimulate interest in several new investigations along the indicated lines.
Hence, in view of the current perspectives in occupational neuroepidemiology, as briefly outlined, there seems to be a reason for conducting additional work-related studies of different, hitherto more or less neglected, neurological and neuropsychiatric disorders. It is not unlikely that there are some contributing effects from various occupational exposures, in the same manner as we have seen for many cancer types. In addition, as in etiologic cancer research, new clues suggesting ultimate causes or triggering mechanisms behind some of the serious neurological disorders may be obtained from occupational epidemiology.
Renal-Urinary Systems
The renal and urinary systems are comprised of a complex series of organs which together function to filter wastes from the blood, and manufacture, store and discharge urine. These organ systems are vital to homeostasis through maintaining fluid balance, acid-base balance and blood pressure. The primary organs of the renal-urinary systems are the two kidneys and the urinary bladder. In the process of filtering waste products from the blood the kidneys are potentially exposed to high concentrations of endogenous and exogenous toxic substances. Thus, some kidney cells are exposed to concentrations a thousand times higher than in blood.
Problems that result in damage to the kidney may be pre-renal (affect blood supply to the kidney), renal (affect the kidney itself) or post-renal (affect any point along the path which the urine travels from the kidney to the end of the urethra or penis). Post-renal problems are usually obstructive in nature; a common site of obstruction is the prostate, juxtapositioned between the bladder and the urethra. Pre-existing disease of the prostate, bladder or ureters, particularly infection, obstruction or foreign bodies such as stones, can compromise kidney function and increase susceptibility to either acquired or genetic defects.
Understanding the microanatomy and molecular mechanisms of the kidneys and bladder is important to assessing susceptibility to, and monitoring and prevention of, occupational exposures. Toxicants seem to target specific parts of the kidney or bladder and result in the expression of specific biomarkers directly related to the damaged segment. Historically, predisposition to disease has been viewed from the epidemiological perspective of identifying a group of workers at risk. Today, with better understanding of the fundamental mechanisms of disease, individual risk assessment through the use of biomarkers of susceptibility, exposure, effect and disease is on the horizon. New ethical issues arise because of the pressure to develop cost-effective strategies to protect workers from occupational hazards. The pressure arises, in part, because genetic testing is gaining acceptance for evaluating disease predisposition and biomarkers of exposure and effect can serve as intermediate end-points at which intervention may be beneficial. The purpose of this chapter is to provide a medical review of the renal and urinary systems on the basis of which guidelines for assessing and reducing individual risk in the workplace could be set forth with due account taken of the ethical aspects involved.
Anatomy and Pathophysiology of the Kidney
The human kidney is a complex organ which functions to filter wastes from the blood through the production of urine. The two kidneys also perform a variety of other vital functions including maintaining homeostasis, regulating blood pressure, osmotic pressure and acid-base balance. The kidneys receive 25% of the total cardiac output of blood, potentially exposing them to endogenous and exogenous toxins.
The kidneys are located on each side of the spine in the lower portion of the back. Each weighs about 150 grams and is about the size of an orange. The kidney consists of three layers: the cortex (outer layer), the medulla and the renal pelvis. Blood flows into the cortex and medulla through the renal artery and branches into increasingly smaller arteries. Each of the arteries ends in a blood filtration unit called a nephron. A healthy kidney contains approximately 1.2 million nephrons, strategically positioned within the cortex and medulla.
A nephron consists of the glomerulus (a group of tiny blood vessels) surrounded by Bowman’s capsule (a two-layer membrane) that opens into a convoluted tubule. The fluid portion of blood, plasma, is forced through the glomerulus into Bowman’s capsule and then, as filtered plasma, passes into the convoluted tubule. About 99% of the water and essential nutrients that have been filtered are reabsorbed by the tubule cells and passed into the capillaries which surround the convoluted tubule. The unfiltered blood which remains in the glomerulus also flows into capillaries and returns through the renal vein to the heart.
The nephrons appear as long, looped ducts comprised of multiple segments each of which performs a variety of different functions designed to maintain the body’s homeostatic mechanisms. Figure 1 depicts a nephron and its orientation within the renal cortex and the medulla. Each nephron segment has a differential blood supply regulating the ionic gradient. Certain chemicals may directly affect specific segments of the nephron acutely or chronically depending on the type and dose of xenobiotic exposure. Depending on the segment of the microanatomy targeted, various aspects of kidney function may be affected.
Figure 1. Relationships of the vascular supply, the glomerulus and the tubular components of the nephron to each other and the orientation of these components within the renal cortex and medulla
Blood vessels to the kidney supply only the glomerular and tubular elements, delivering wastes to be filtered and absorbing nutrients, proteins and electrolytes in addition to supplying oxygen for organ viability. Ninety per cent of the blood flow is to the cortex, with a gradient decrease to the medulla. Such differential blood flow, and the positioning of the nephron units, are vital to the countercurrent mechanism which further concentrates the urine and potential nephrotoxins.
The glomerulus is positioned between the afferent and efferent arterioles. The efferent arterioles form a web of capillaries around each nephron unit with the exception of the distal tubule juxtaposition adjacent to the afferent blood supply of the glomerulus. Afferent and efferent tubules enervated by the sympathetic nerves respond to autonomic stimulation and hormonal mediators such as vasopression and antidiuretic hormone (ADH). An area called the macula densa, part of the juxtaglomerular apparatus, produces renin, a mediator of blood pressure, in response to osmotic changes and blood pressure. Renin is converted by liver enzymes to an octapeptide, angiotensin II, that regulates blood flow to the kidneys preferentially targeting the afferent arterioles and the mesangial cells of the glomerulus.
The glomerulus allows only certain size proteins with defined charge to pass through during filtration. Plasma filtration is controlled by a balance of osmotic and hydrostatic pressure. Specialized sugar molecules, glycosaminoglycans, provide negative anionic charge which inhibit, by electrostatic forces, the filtration of negatively charged materials. The three-cell layer of the glomerular basement membrane consists of multiple foot processes that increase the absorption area and create the pores through which the filtrate passes. Damage to the specialized basement membrane or the capillary endothelium may permit albumin, a type of protein, to be spilled in increased amounts into the urine. The presence of an excess amount of albumin or other micro-proteins in the urine serves as a marker of glomerular or tubular damage.
The renal interstitium is the space between the nephron units and is more prominent in the central medullary portion than in the outer cortex. Within the interstitium are interstitial cells that are in close proximity to the medullary blood vessels and tubule cells. With ageing there may be an increased prominence of interstitial cells in the cortex with associated fibrosis and scarring. The interstitial cells contain lipid droplets and may be involved in the control of blood pressure with the release of vascular relaxing or constricting factors. Chronic disease of the interstitium may affect the glomerulus and tubules, or conversely, disease of the glomerulus and tubules may affect the interstitium. Thus, in end-stage kidney disease it is sometimes difficult to precisely define the pathological mechanisms of renal failure.
The proximal collecting tubules absorb 80% of the sodium, water and chloride, and 100% of the urea. Each proximal tubule has three segments, with the last segment (P-3) the most vulnerable to xenobiotic (toxic foreign substance) exposures. When the proximal cells are damaged by heavy metals such as chromium, the concentrating ability of the kidney is impaired and urine may be more dilute. Toxicity to the P-3 segment results in the release into the urine of enzymes, such as intestinal alkaline phosphatase, N-acetyl-beta-D-glucosaminidase (NAG), or Tamm-Horsfall protein, which is associated with the brush-like border of the proximal tubule cells increasing the effective absorbing area.
Diagnosis and Testing for Nephrotoxicity
Serum creatinine is another substance filtered by the glomerulus but minimally absorbed by the proximal tubules. Damage to the glomerulus results in its inability to remove toxins produced by the body and there is an accumulation of serum creatinine. Because serum creatinine is a product of muscle metabolism and dependent on the patient’s body mass, it has low sensitivity and specificity for measuring renal function, but it is used frequently because it is convenient. A more sensitive and specific test is to quantitate the filtrate by measuring the creatinine (Cr) clearance; serum urinary creatinine clearance is calculated by the general formula CCr=UCr V/PCr, where UCrV is the amount of Cr excreted per unit time and PCr is the plasma concentration of the Cr. However, creatinine clearance is more complex, in terms of sampling for the test, and is thus impractical for occupational testing. Isotope clearance tests performed by radioactive labelling of compounds such as ortho-iodohippurate which are also cleared by the kidney are also effective, but not practical or cost-effective in the workplace setting. Differential function of individual kidneys may be determined using differential renal nuclear scans or selective catheterization of both kidneys by passage of a catheter from the bladder up through the ureter into the kidney. However these methods also are not readily employed for large-scale workplace testing. Because kidney function may be reduced by 70 to 80% prior to a detectable elevation in serum creatinine, and because other existing tests are either impractical or costly, non-invasive biomarkers are needed to detect low-dose acute intermittent exposures to the kidney. A number of biomarkers for detecting low-dose kidney damage or early changes associated with carcinogenesis are discussed in the section on biomarkers.
Although the proximal tubule cells absorb 80% of the fluids, the countercurrent mechanism and the distal collecting ducts fine-tune the amount of fluids absorbed by regulating ADH. ADH is released from the pituitary gland deep within the brain and responds to osmotic pressures and fluid volume. Exogenous compounds such as lithium may damage the distal collecting ducts and result in renal diabetes insipidus (passage of dilute urine). Inherited genetic disorders may also cause this defect. Xenobiotics normally affect both kidneys but complexities of interpretation arise when exposures are difficult to document or when there is pre-existing renal disease. Consequently, high-dose accidental exposures have served as markers for identifying nephrotoxic compounds in many instances. The majority of occupational exposures occur at low doses, and are masked by the reserve filtration and repair compensatory capability (hypertrophy) of the kidney. The challenge which remains is to detect low-dose exposures clinically undetected by current methods.
Anatomy and Pathophysiology of the Bladder
The urinary bladder is a hollow pouch in which urine is stored; normally, it contracts on demand for controlled emptying through the urethra. The bladder is located in the front, lower part of the pelvic cavity. The bladder is joined on either side to the two kidneys by muscular, peristaltic tubes, the ureters, which carry the urine from the kidneys to the bladder. The renal pelvis, ureters and bladder are lined with transitional epithelium. The outer layer of the urothelium consists of umbrella cells coated with a carbohydrate, glycosaminoglycan (GAG), layer. The transitional cells extend to the basement membrane of the bladder. The deep basal cells are thus protected by the umbrella cells but if the protective GAG layer is damaged the basal cells are susceptible to injury from urinary components. The microanatomy of the transitional epithelium allows it to expand and contract, and even with normal shedding of the umbrella cells the protective integrity of the basal cells is maintained.
The balanced neurological system that regulates storage and emptying may be damaged during electroshock or other trauma, such as spinal cord injury, occurring in the workplace. A major cause of death among quadraplegics is loss of bladder function resulting in chronic renal damage secondary to infection and stone formation. Chronic infection from incomplete emptying due to neurogenic or obstructive causes such as pelvic fracture or other trauma to the urethra and subsequent stricture formation is common. Persistent bacterial infection or stone formation that results in chronic inflammatory and malignant conditions of the bladder may be caused by reduced resistance (i.e., susceptibility) to exogenous exposures in the workplace.
Molecules associated with damage and repair within the bladder serve as potential intermediate end-point markers for both toxic and malignant conditions because many biochemical alterations occur during the changes related to cancer development. Like the kidney, bladder cells have active enzyme systems such as the cytochrome P-450 which may activate or inactivate xenobiotics. The functional activity of the enzymes is determined by genetic inheritance and exhibits genetic polymorphism. Voided urine contains cells exfoliated from the kidney, ureters, bladder, prostate and urethra. These cells provide targets, through the use of biomarkers, for evaluating changes in bladder and renal pathology. Remembering Virchow’s comment that all diseases start in the cells focuses our attention on the importance of cells, which are the molecular mirror of exposure episodes.
Environmental and Occupational Toxicology
A considerable volume of epidemiological data supports the causal relationship of occupational exposures in bladder cancer, but the precise contributions of workplace exposures to kidney failure and kidney cancer are difficult to estimate. In a recent report, it was estimated that up to 10% of end-stage renal disease could be attributed to workplace exposures, but results are difficult to validate because of changing environmental and chemical hazards, variations in diagnostic criteria and the often long latency period between exposure and disease. It is estimated that function of two-thirds of the nephrons of both kidneys may be lost before renal damage is clinically evident. However, evidence is mounting that what were previously thought to be socioeconomic or ethnic causes of nephrotoxicity may in fact be environmental, adding validity to the role of toxicants in disease development.
Nephrotoxicity may be directly related to the xenobiotic, or the xenobiotic may go through a single-step or multi-step activation or inactivation in the kidney or the liver. Activation of xenobiotics is regulated by complex sets of enzymes identified as Phase I, II and Ancillary. One Phase I enzyme is the P-450 oxidative system which acts through reduction or hydrolysis pathways. Phase II enzymes catalyse conjugation while ancillary enzymes regulate drug metabolism (Table 1 lists these enzymes). Various animal models have provided insight into metabolic mechanisms, and studies of kidney slices and microdissection of the kidney nephron units in tissue culture add insight into the pathological mechanisms. However, species and individual variables are considerable and, although mechanisms may be similar, caution is mandated in extrapolating results to humans in the workplace. The primary issues now are to determine which xenobiotics are nephrotoxic and/or carcinogenic, and to what target sites, and to develop methods to identify more accurately subclinical toxicity in the renal-urinary system.
Table 1. Drug-metabolism enzymes in kidney1
| ENZYMES | ||
| Phase I | Phase II | Ancillary |
| Cytochrome P-450 | Esterase | GSH peroxides |
| Microsomal FAD-containing mono-oxygenase | N-Acetyltransferase | GSSG reductase |
| Alcohol and aldehyde dehydrogenases | GSH S-transferase | Superoxide dismutase |
| Epoxide hydrolase | Thiol S-methyl-transferase | Catalase |
| Prostaglandin synthase | UDP glucuronosyltransferase | DT-diaphorase |
| Monoamine oxidase | Sulphotransferase | NADPH-generating pathways |
1 Phase I enzymes catalyse oxidation, reduction or hydrolysis.
Phase II enzymes generally catalyse conjugation.
Ancillary enzymes function in a secondary or supporting manner to facilitate drug metabolism.
Source: National Research Council 1995.
Non-malignant Renal-Urinary Disorders
Glomerulonephritis is an inflammatory reactive condition of the glomerular basement membrane or capillary endothelium. Acute and chronic forms of the disease are caused by a variety of infectious, autoimmune or inflammatory conditions or by exposure to toxic agents. Glomerulonephritis is associated with vasculitis, either systemic or limited to the kidney. Secondary chronic damage to the glomerulus also occurs during an intense cycle of assault from nephrotoxicity to the interstitium of the tubule cells. Epithelial glomerular crescents or proliferative forms are a hallmark of glomerulonephritis in kidney biopsy specimens. Blood, red blood cell (RBC) casts, or protein in the urine, and hypertension are symptoms of glomerulonephritis. A change in blood proteins may occur with lowering of certain fractions of the serum complement, a complex set of interacting proteins involved in the immune system, host defenses and clotting functions. Direct and indirect evidence supports the significance of xenobiotics as a causal factor of glomerulonephritis.
The glomerulus protects the oxygen-carrying red blood cells from passing through its filter. After centrifugation, normal urine contains only one RBC in 10 ml when viewed with high-power light microscopy. When RBCs leak through the glomerular filter and perhaps become individually dysmorphic, RBC casts that assume the cylindrical shape of the collecting nephrons form.
In support of the importance of toxins as an aetiological factor in glomerulonephritis, epidemiological studies reveal increased evidence of toxic exposures in patients who have undergone dialysis or who have been diagnosed with glomerulonephritis. Evidence of glomerular injury from acute hydrocarbon exposure is rare, but has been observed in epidemiological studies, with odds ratios ranging from 2.0 to 15.5. One example of acute toxicity is Goodpasture’s disease which results from hydrocarbon stimulation of antibody production to liver and lung proteins that cross-react with the basement membrane. Exacerbation of nephrotic syndrome, large amounts of protein in the urine, has also been observed in individuals re-exposed to organic solvents, while other studies reveal an historic relationship with a spectrum of renal disorders. Other solvents such as degreasing agents, paints and glues are implicated in more chronic forms of the disease. Awareness of the mechanisms of solvent excretion and reabsorption assists in identifying biomarkers because even minimal damage to the glomerulus results in increased leakage of RBCs into the urine. Although RBCs in the urine are a cardinal sign of glomerular injury, it is important to rule out other causes of haematuria.
Interstitial and tubular nephritis. As mentioned previously, the aetiology of chronic end-stage renal disease is frequently difficult to ascertain. It may be primarily glomerular, tubular or interstitial in origin and occur because of multiple acute episodes or chronic, low-dose processes. Chronic interstitial nephritis involves fibrosis and tubular atrophy. In its acute form, the disease is expressed by marked inflammatory infiltrate with accompanying fluid collection in the interstitial spaces. Interstitial nephritis may involve primarily the interstitium, or be manifest as a secondary event from chronic tubular injury, or it may result from post-renal causes such as obstruction. Prostaglandin-A synthase, an enzyme, is found primarily in the interstitium and is associated with the endoplasmic reticulum, a part of the protein machinery of the cell. Certain xenobiotics, such as benzidine and nitrofuranes, are reducing co-substrates for prostaglandin synthase and are toxic to the tubular interstitium.
Tubular and interstitial injury may occur from exposure to cadmium, lead or a variety of organic solvents. Most of the exposures are chronic, low-dose and toxicity is masked by the renal function reserve and the ability of the kidney to recover some functions. Interstitial nephritis may also result from vascular injury as caused, for example, by chronic carbon monoxide exposure. Proximal tubule cells are the most vulnerable to toxic substances in the blood because of intense exposure to toxins which filter through the glomerulus, internal enzyme systems that activate toxicants and the selective transport of toxicants. The epithelium in the various segments of the proximal tubule has slightly different qualities of lysosomal peroxidase enzymes and other compounds of genetic machinery. Thus, chromium exposure may result in both interstitial and tubular injury. Damage to the collecting tubules may occur when specific enzymes activate various xenobiotics such as chloroform, acetaminophen and p-aminophenol, and antibiotics such as Loradine. A secondary result of damage to the collecting ducts is the inability of the kidney to acidify the urine and the subsequent development of a metabolic acid state.
Nephrogenic diabetes insipidus, the condition in which urine becomes dilute, may be genetic or acquired. The genetic form involves mutations of the ADH receptors which are located on the basal lateral membrane of the collecting ducts, in the descending loop of Henle. ADH fine-tunes the reabsorption of water and certain ions such as potassium. Acquired diabetes insipidus may involve the tubule cells or the associated interstitium, both of which may be diseased because of a variety of conditions. Nephrogenic diabetes insipidus may accompany end-stage renal disease because of diffuse involvement of the interstitium. Consequently, the interstitium is unable to maintain a hypertonic environment for passive water movement from the tubular collecting ducts. Conditions which may cause diffuse interstitial changes are pyelonephritis, sickle cell anaemia and obstructive uropathy. The possible association of these conditions in relation to occupational exposure is an increased susceptibility of the kidney to xenobiotics. A limited number of nephrotoxic compounds have been identified that especially target the collecting tubule cells. Frequency, nocturia (more frequent voiding at night) and polydipsia (chronic thirst) are symptoms of nephrogenic diabetes insipidus. Movement of fluids through the collecting duct cells results in channels that form in response to ADH, affecting the microtubular function of the cells; consequently, drugs such as colchicine may affect the ADH. Two drugs which appear to act by slightly different mechanisms to correct ADH are hydrochlorothiazide and indomethacin, a prostaglandin synthase inhibitor.
Lithium-induced diabetes insipidus correlates with the duration of lithium therapy, average serum lithium level and total lithium carbonate dose. Interestingly, lithium concentrates in the collecting ducts and affects cyclic AMP, part of the energy metabolic pump pathway. Exposure to other compounds such as methoxyflurane and demeclocycline, the latter of which is used for the treatment of acne, also results in nephrogenic diabetes insipidus through an alternative pathway rendering the epithelial cells unresponsive to ADH.
Hypertension, or elevated blood pressure, the second most common cause of end-stage renal disease, is associated with multiple aetiological pathways. Hypertension can be caused by diabetic nephropathy, obstructive nephropathy, glomerulonephritis, polycystic kidney disease, pyelonephritis and vasculitis, and many of those diseases are associated with exposure to toxic compounds. A limited number of occupational exposures are directly associated with hypertension. One is lead, which causes renal vascular ischaemia and injury. The mechanism for lead-induced hypertension is probably regulated through the juxtaglomerular apparatus, the release of renin and the cleavage of renin by liver enzymes to angiotensin II. Drugs implicated in hypertension include amphetamines, oestrogens and oral contraceptives, steroids, cis-platinum, alcohol and tricyclic antidepressants. Hypertension may be gradual in onset or acute and malignant in nature. Malignant hypertension in which diastolic pressure is greater than 110 mm Hg is associated with nausea, vomiting and severe headache, and constitutes a medical emergency. Numerous drugs are available for the treatment of hypertension but over-treatment may result in decreased renal perfusion and a further loss of renal function. Whenever possible, withdrawal of the nephrotoxicant is the treatment of choice.
Differential diagnosis of haematuria and proteinuria
Haematuria (RBCs in the urine) and pyuria (white blood cells in the urine) are primary symptoms of many diseases of the renal-urinary system, and for categorical purposes may be considered non-specific cellular biomarkers. Because of their importance they are discussed separately here. A challenge to the occupational practitioner is to determine if haematuria signifies a permanent underlying medical condition that may be potentially life threatening or if it is attributable to occupational exposures. Clinical assessment of haematuria requires standardization and determination of whether it is pre-renal, renal or post-renal in origin.
Haematuria may be derived from lesions in the kidney per se or anywhere along the pathway of voided urine. Sites of origin include the kidney, collecting renal pelvis, ureters, bladder, prostate and urethra. Because of the serious diseases associated with haematuria, a single episode warrants a medical or urological evaluation. Greater than one RBC per high-power field can be a signal of disease, but significant haematuria may be missed on microscopic analysis in the presence of hypotonic (dilute) urine which may lyse RBCs. Pseudo-haematuria may be caused by beets, berries, vegetable dyes and concentrated urates. Initial haematuria suggests a urethral origin, terminal haematuria is usually prostatic in origin, and blood throughout voiding is from the bladder, kidney or ureter. Gross haematuria is associated with bladder tumours in 21% of the cases, but microscopic haematuria is much less frequently associated (2.2 to 12.5%).
Finding dysmorphic cells when haematuria is quantitatively assessed suggests an upper tract origin, particularly when associated with red blood cell casts. Understanding haematuria in relation to proteinuria provides additional information. The glomerular filtration device almost completely excludes proteins of a molecular weight greater than 250,000 Daltons, while low molecular weight proteins are freely filtered and absorbed normally by the tubule cells. The presence of high molecular weight proteins in the urine suggests lower tract bleeding while low molecular weight proteins are associated with tubular injury. Evaluation of the ratio of α-microglobulin to albumin and α-macroglobulin to albumin helps delineate glomerular from tubular interstitial nephropathy and lower tract bleeding potentially associated with urothelial neoplasia and other post-renal causes such as urinary tract infections.
A special diagnostic problem arises when two or more disease processes that cause the same symptoms are present concurrently. For example, haematuria is seen in both urothelial neoplasia and urinary tract infections. In a patient with both diseases, if the infection is treated and resolved, the cancer would remain. Therefore, it is important to identify the true cause of the symptoms. Haematuria is present in 13% of screened populations; approximately 20% of individuals have significant renal or bladder disorders and 10% of those will go on to develop genitourinary malignancy. Consequently, haematuria is an important biomarker of disease that must be appropriately evaluated.
Clinical interpretation of haematuria is enhanced by a knowledge of the patient’s age and sex, as indicated in Table 2 which shows causes of haematuria relative to the age and sex of the patient. Other causes of haematuria include renal vein thrombosis, hypercalcuria and vasculitis, as well as trauma such as jogging or other sports, and occupational events or exposures. Clinical evaluation of haematuria requires an x ray of the kidney, intravenous pyelogram (IVP), to rule out upper tract diseases including kidney stones and tumours, and a cystoscopy (looking into the bladder through a lighted instrument) to exclude bladder, prostate or urothelial cancers. Subtle vaginal causes must be excluded in women. Regardless of a patient’s age, a clinical evaluation is indicated if haematuria occurs and, depending on the identified aetiology, sequential follow-up evaluations may be indicated.
Table 2. The most common causes of haematuria, by age and sex
| 0–20 Years | 40–60 Years (females) |
| Acute glomerulonephritis Acute urinary tract infection Congenital urinary tract anomalies with obstruction |
Acute urinary tract infection Stones Bladder tumour |
| 20–40 Years | 60+Years (males) |
| Acute urinary tract infection Stones Bladder tumour |
Benign prostatic hyperplasia Bladder tumour Acute urinary tract infection |
| 40–60 Years (males) | 60+Years (females) |
| Bladder tumour Stones Acute urinary tract infection |
Bladder tumours Acute urinary tract infection |
Source: Wyker 1991.
The use of recently identified biomarkers in conjunction with conventional cytology for evaluation of haematuria helps to assure that no occult or incipient malignancy is missed (see next section on biomarkers). For the occupational specialist, determining whether haematuria is a result of toxic exposure or occult malignancy is important. Knowledge of exposure and the patient’s age are critical parameters for making an informed clinical management decision. A recent study has demonstrated that together haematuria and biomarker analysis on exfoliated urinary cells from the bladder were the two best markers for detecting premalignant bladder lesions. Haematuria is observed in all cases of glomerular injury, in only 60% of patients with bladder cancer and in only 15% of patients with malignancies of the kidney itself. Thus, haematuria remains a cardinal symptom of renal and post-renal disease, but the final diagnosis may be complex.
Tests for nephrotoxicity: biomarkers
Historically, monitoring of toxins in the work environment has been the primary method of identifying risk. However, not all toxicants are known and, therefore, cannot be monitored. Also, susceptibility is a factor in whether xenobiotics will affect individuals.
Figure 2. Categories of biomarkers.
Biomarkers provide new opportunities for defining individual risk. For descriptive purposes and to provide a framework for interpretation, biomarkers have been classified according to the schema depicted in Figure 2. As in other diseases, biomarkers of nephrotoxicity and genitourinary toxicity may be related to susceptibility, exposure, effect or disease. Biomarkers may be genotypic or phenotypic, and may be functional, cellular or soluble in urine, blood or other body fluids. Examples of soluble markers are proteins, enzymes, cytokines and growth factors. Biomarkers may be assayed as the gene, message or protein product. These variable systems add to the complexity of biomarker evaluation and selection. One advantage of assaying the protein is that it is the functional molecule. The gene may not be transcribed and the quantity of message may not correspond to the protein product. A list of criteria for biomarker selection is shown in Table 3.
Table 3. Criteria for biomarker selection
| Clinical utility | Assay considerations |
| Strong biomarker | Stability of reagent |
| Sensitivity | Cost of reagent |
| Specificity | Fixation requirements |
| Negative predictive value | Reproducibility of the assay |
| Positive predictive value | Machine sensible parameters |
| Functional role | Contribution to biomarker profile |
| Sequence in oncogenesis | Adaptability to automation |
Source: Hemstreet et al. 1996.
The international scientific commitment to map the human genome made possible by advances in molecular biology established the basis for identifying biomarkers of susceptibility. Most instances of human disease, especially those resulting from environmental exposure to toxicants, involve a constellation of genes reflecting marked genetic diversity (genetic polymorphism). An example of such a gene product, as mentioned previously, is the P-450 oxidative enzyme system which may metabolize xenobiotics in the liver, kidney or bladder. Susceptibility factors may also control the basic mechanism for DNA repair, influence the susceptibility of various signalling pathways important to tumourigenesis (i.e., growth factors) or be related to inherited conditions that predispose to disease. An important example of an inherited susceptibility factor is the slow or fast acetylation phenotype that regulates the acetylation and inactivation of certain aromatic amines known to cause bladder cancer. Biomarkers of susceptibility may include not only genes that regulate the activation of xenobiotics but also proto-oncogenes and suppressor-oncogenes. The control of tumour cell growth involves a number of complex, interacting systems. These include a balance of positive (proto) oncogenes and negative (suppressor) oncogenes. Proto-oncogenes control normal cell growth and development, while suppressor-oncogenes control normal cellular division and differentiation. Other genes may contribute to pre-existing conditions such as a propensity to renal failure triggered by underlying conditions such as polycystic kidney disease.
A biomarker of exposure may be the xenobiotic itself, the metabolic metabolite or markers such as DNA adducts. In some instances the biomarker may be bound to a protein. Biomarkers of exposure may also be biomarkers of effect, if the effect is transient. If a biomarker of effect persists, it may become a biomarker of disease. Useful biomarkers of effect have a high association with a toxicant and are indicative of exposure. For disease detection, expression of the biomarker in close sequence to the onset of disease will have the highest specificity. The expected sensitivity and specificity of a biomarker depends on the risk versus benefit of the intervention. For instance, a biomarker such as F-actin, a cytoskeletal protein differentiation marker, that appears altered in early carcinogenesis may have a poor specificity for detection of pre-cancerous states because not all individuals with an abnormal marker will progress to disease. It may, however, be useful for selecting individuals and monitoring them while undergoing chemoprevention, provided the therapy is non-toxic. Understanding the time-frame and functional linkage between individual biomarkers is extremely important to individual risk assessment and to comprehending the mechanisms of carcinogenesis and nephrotoxicity.
Biomarkers of nephrotoxicity
Biomarkers of nephrotoxicity may be related to the aetiology of kidney failure (i.e., pre-renal, renal or post-renal) and the mechanisms involved in the pathogenesis of the process. This process includes cellular damage and repair. Toxic injury can affect the cells, glomerulus, interstitium or tubules with release of corresponding biomarkers. Xenobiotics may affect more than one compartment or may cause biomarker changes because of the interdependence of cells within the compartment. Inflammatory changes, autoimmune processes and immunological processes further promote the release of biomarkers. Xenobiotics may target one compartment in some circumstances and another under different conditions. One example is mercury which is, acutely, nephrotoxic to the proximal tubule while chronically it affects the arterioles. Response to injury can be divided into several major categories including hypertrophy, proliferation, degeneration (necrosis and apoptosis, or programmed cell death) and membrane alterations.
The majority of susceptibility factors are related to non-xenobiotic-associated renal disease. However, 10% of renal failure cases are attributed to environmental exposures to toxic compounds or iatrogenic induction by various compounds, such as antibiotics, or procedures such as administration of kidney x-ray contrast to a diabetic. In the workplace, identifying subclinical renal failure prior to potential additional nephrotoxic stress has potential practical utility. If a compound is suspected to be xenobiotic and it results in an effect specifically in the causal pathway of disease, intervention to reverse the effect is a possibility. Thus, biomarkers of effect eliminate many of the problems of calculating exposure and defining individual susceptibility. Statistical analysis of biomarkers of effect in relation to biomarkers of susceptibility and exposure should improve marker specificity. The more specific the biomarker of effect the less the requirement for a large sample size required for scientifically identifying potential toxins.
Biomarkers of effect are the most important class of markers and link exposure to susceptibility and disease. We have previously addressed the combining of cellular and soluble biomarkers to differentiate between haematuria originating in the upper tract or the lower tract. A list of soluble biomarkers potentially related to cellular nephrotoxicity is shown in Table 4. To date, none of these alone or as multiple biomarker panels detects subclinical toxicity with adequate sensitivity. Some problems with using soluble biomarkers are lack of specificity, enzyme instability, the dilutional effect of urine, variations in renal function, and non-specific protein interactions that may cloud the specificity of analysis.
Table 4. Potential biomarkers linked to cell injury
|
Immunological factors: -Humoral-antibodies and antibody fragments; components of complement cascade, and coagulation factors -Cellular-lymphocytes, mononuclear phagocytes, and other marrow- derived effectors (oesinophils, basophils, neutrophils and platelets) Lymphokines Major histocompatibility antigens Growth factors and cytokines: platelet- derived growth factor, epidermal growth factor, transforming growth factor (TGF), tumour-necrosis factor, interleukin-1, etc. Lipid mediators: prostaglandins Endothelin |
Extracellular-matrix components: -Collagens -Procollagen -Laminin -Fibronectin Reactive oxygen and nitrogen species Transcription factors and proto-oncogenes: c-myc, c-fos, c-jun, c-Haras, c-Ki-ras, and Egr-1 Heat shock proteins |
Source: Finn, Hemstreet et al. in National Research Council 1995.
One soluble growth factor with potential clinical application is urinary epidermal growth factor (EGF) which may be excreted by the kidney and is also altered in patients with transitional cell carcinoma of the bladder. Quantitation of urinary enzymes has been investigated but the usefulness of this has been limited by the inability to determine the origin of the enzyme and lack of assay reproducibility. The use of urinary enzymes and their widespread acceptance has been slow because of the restrictive criteria mentioned previously. Enzymes evaluated include alaminopeptidase, NAG and intestinal alkaline phosphatase. NAG is perhaps the most widely accepted marker for monitoring proximal tubule cell injury because of its localization in the S3 segment of the tubule. Because the precise cell of origin and pathological cause of urinary enzyme activity are unknown, interpretation of results is difficult. Furthermore, drugs, diagnostic procedures and co-existing diseases such as myocardial infarction may cloud the interpretation.
An alternative approach is to use monoclonal antibody biomarkers to identify and quantitate tubular cells in urine from various areas of the nephron segment. The utility of this approach will depend on maintaining the integrity of the cell for quantification. This requires appropriate fixation and sample handling. Monoclonal antibodies are now available which target specific tubule cells and distinguish, for example, proximal tubule cells from distal tubule cells or convoluted tubule cells. Transmission microscopy cannot effectively resolve differences between leukocytes and various types of tubule cells in contrast to electron microscopy which has been effective in detecting transplant rejection. Techniques such as high-speed quantitative fluorescence image analysis of tubular cells stained with monoclonal antibodies should solve this problem. In the near future, it should be possible to detect subclinical nephrotoxicity with a high degree of certainty as exposure occurs.
Biomarkers of malignant disease
Solid cancers arise in many cases from a field of biochemically altered cells which may or may not be histologically or cytologically altered. Technologies such as quantitative fluorescence image analysis capable of detecting biomarkers associated with premalignant conditions with certainty provide the horizon for targeted chemoprevention. Biochemical alterations may occur in a varied or ordered process. Phenotypically, these changes are expressed by a gradual morphological progression from atypia to dysplasia and finally to overt malignancy. Knowledge of the “functional role” of a biomarker and “when in the sequence of tumorigenesis it is expressed” assists in defining its utility for identifying premalignant disease, for making an early diagnosis and for developing a panel of biomarkers to predict tumour recurrence and progression. A paradigm for biomarker evaluation is evolving and requires the identification of single and multiple biomarker profiles.
Bladder cancer appears to develop along two separate pathways: a low-grade pathway seemingly associated with alterations on chromosome 9 and a second pathway associated with P-53 suppressor gene genetically altered on chromosome 17. Clearly, multiple genetic factors are related to cancer development, and defining the genetic factors in each individual is a difficult task, particularly when the genetic pathway must be linked to a complexity of perhaps multiple exposures. In epidemiological studies, exposures over prolonged intervals have been difficult to reconstruct. Batteries of phenotypic and genotypic markers are being identified to define individuals at risk in occupational cohorts. One profile of phenotypic biomarkers and their relationship to bladder cancer is shown in Figure 3, which illustrates that G-actin, a precursor protein to the cytoskeletal protein F-actin, is an early differentiation marker and may be followed by sequential alterations of other intermediate end-point markers such as M344, DD23 and DNA ploidy. The strongest biomarker panels for detecting premalignant disease and overt cancer, and for prognostication, remain to be determined. As machine-sensible biochemical criteria are defined it may be possible to detect disease risk at prescribed points in the disease continuum.
Figure 3. Four biomarkers, G-actin, P-300, DD23 and DNA, in relation to tumour progression and response to surgical treatment and chemoprevention.
Diagnosis and management of work-relatedrenal-urinary disease
Pre-existing renal disease
Changes in health care delivery systems worldwide bring into focus issues of insurability and protection of workers from additional exposure. Significant pre-existing renal disease is manifest by increased serum creatinine, glucosuria (sugar in the urine), proteinuria, haematuria and dilute urine. Immediately ruling out systemic underlying causes such as diabetes and hypertension is required, and depending on the age of the patient other congenital aetiologies such as multiple cysts in the kidney should be investigated. Thus, the urinalysis, both dipstick and microscopic evaluations, for detection of biochemical and cellular alterations, is useful to the occupational physician. Tests of serum creatinine and creatinine clearance are indicated if significant haematuria, pyuria or proteinuria suggests underlying pathology.
Multiple factors are important to assess risk for progression of chronic disease or acute kidney failure. The first is inherent or acquired limitation of the kidney to resist xenobiotic exposure. The kidney’s response to the nephrotoxicant, such as an increase in the amount of toxicant absorbed or alterations in kidney metabolism, may be influenced by a pre-existing condition. Of particular importance is a decrease in detoxifying function in the very young or the very old. In one study susceptibility to occupational exposure was correlated highly with family history of renal disease, signifying the importance of hereditary predisposition. Underlying conditions, such as diabetes and hypertension, increase susceptibility. Rare conditions, such as lupus erythematosis and vasculitis, may be additional susceptibility factors. In the majority of cases, increased susceptibility is multifactorial and frequently involves a battery of insults which occur either alone or simultaneously. Thus, the occupational physician should be cognizant of the patient’s family history of renal disease and pre-existing conditions affecting renal function, as well as any vascular or cardiac disease, particularly in older workers.
Acute renal failure
Acute renal failure may arise from pre-renal, renal, or post-renal causes. The condition is usually caused by an acute insult resulting in rapid, progressive loss of kidney function. When the nephrotoxicant or precipitating causal factor is removed there is a progressive return of renal function with a gradual decline of serum creatinine and improved renal concentrating ability. A listing of occupational causes of acute renal failure is shown in Table 5. Acute renal failure from high-dose xenobiotic exposure has been useful to signal potential aetiological causes that may also contribute to more chronic forms of progressive renal disease. Acute renal failure from obstruction of the outflow tract caused by benign disease or malignancy is relatively rare, but surgical causes may contribute more frequently. Ultrasound of the upper tract delineates the problem of obstruction, whatever the contributing factor. Renal failure associated with drug or occupational toxicants results in a mortality rate of approximately 37%; the remainder of affected individuals improve to various degrees.
Table 5. Principal causes of acute renal insufficiency of occupational origin
| Renal ischaemia | Tubular necrosis | Haemoglobinuria, myoglobinuria |
| Traumatic shock Anaphylactic shock Acute carbon monoxide poisoning Heat stroke |
Mercury Chromium Arsenic Oxalic acid Tartrates Ethylene glycol Carbon tetrachloride Tetrachlorethane |
Arsine Crush syndrome Struck by lightning |
Source: Crepet 1983.
Acute renal failure may be attributed to a variety of pre-renal causes which have as an underlying theme renal ischaemia resulting from a prolonged decreased renal perfusion. Cardiac failure and renal artery obstruction are two examples. Tubular necrosis may be caused by an ever-growing number of nephrotoxicants present in the workplace. Herbicides and pesticides have all been implicated in a number of studies. In a recent report, hemlock poisoning resulted in the deposition of the myosin and actin from the breakdown of muscle cells in the tubules and an acute decrease in renal function. Endosulfan, an insecticide, and triphenyltin acetate (TPTA), an organotin, both were initially classified as neurotoxins but have recently been reported to be associated with tubular necrosis. Anecdotal reports of additional cases bring into perspective the need for finding biomarkers to identify more subtle subclinical toxicants that may not yet have resulted in high-dose toxic exposures.
Signs and symptoms of acute renal failure are: no urine output (anuria); oliguria (decreased urine output); decreased renal concentrating capacity; and/or a rising serum potassium that may stop the heart in a relaxed state (diastolic arrest). Treatment involves clinical support and, whenever possible, removal from exposure to the toxicant. Rising serum potassium or excessive fluid retention are the two primary indicators for either haemodialysis or peritoneal dialysis, with the choice dependent on the patient’s cardiovascular stability and vascular access for haemodialysis. The nephrologist, a medical kidney specialist, is key in the management strategy for these patients who may also require the care of a urological surgical specialist.
Long-term management of patients following renal failure is largely dependent on the degree of recovery and rehabilitation and the patient’s overall health status. A return to limited work and avoiding conditions that will stress the underlying condition are desirable. Patients with persistent haematuria or pyuria require careful monitoring, possibly with biomarkers, for 2 years following recovery.
Chronic renal disease
Chronic or end-stage renal disease is most frequently the result of a chronic, ongoing subclinical process that involves a multiplicity of factors most of which are poorly understood. Glomerulonephritis, cardiovascular causes and hypertension are major contributing factors. Other factors include diabetes and nephrotoxicants. Patients present with progressive elevations in serum blood urea nitrogen, creatinine, serum potassium and oliguria (decreased urine output). Improved biomarkers or biomarker panels are needed to identify more precisely subclinical nephrotoxicity. For the occupational practitioner, the methods of assessment need to be non-invasive, highly specific and reproducible. No single biomarker has as yet met these criteria to become practical on a large clinical scale.
Chronic renal disease may result from a variety of nephrotoxicants, the pathogenesis of which is better understood for some than others. A list of nephrotoxicants and sites of toxicity is shown in Table 6. As mentioned, toxins may target the glomerulus, segments of the tubules or the interstitial cells. Symptoms of xenobiotic exposure may include haematuria, pyuria, glucosuria, amino acids in the urine, frequent urination and decreased urine output. The precise mechanisms of renal damage for many nephrotoxicants have not been defined but the identification of specific biomarkers of nephrotoxicity should assist in addressing this problem. Although some protection of the kidney is afforded by the prevention of vasoconstriction, tubular injury persists in most cases. As an example, lead toxicity is primarily vascular in origin, while chromium at low doses affects the proximal tubule cells. These compounds appear to affect the metabolic machinery of the cell. Multiple forms of mercury have been implicated in acute elemental nephrotoxicity. Cadmium, in contrast to mercury and like many other occupational nephrotoxicants, first targets the proximal tubule cells.
Table 6. Segments of the nephron affected by selected toxicants
|
Proximal tubule Antibiotics -Cephalosporins Aminoglycosides Antineoplastics -Nitrosoureas -Cisplatin and analogs Radiographic contrast agents Halogenated hydrocarbons -Chlorotrifluoroethylene -Hexafluropropene -Hexachlorobutadiene -Trichloroethylene -Chloroform -Carbon tetrachloride Maleic acid Citrinin Metals -Mercury -Uranyl nitrate -Cadmium -Chromium |
Glomerulus Immune complexes Aminoglycoside antibiotics Puromycin aminonucleoside Adriamycin Penicillamine Distal tubule/collecting duct -Lithium -Tetracyclines -Amphotericin -Fluoride -Methoxyflurane
Papilla -Aspirin -Phenacetin -Acetaminophen -Non-steroidal anti-inflammatory agents -2-Bromoethylamine |
Source: Tarloff and Goldstein 1994.
More...
Renal-Urinary Cancers
Kidney Cancer
Epidemiology
Historically, kidney cancer has been used to mean either all malignancies of the renal system (renal cell carcinoma (RCC), ICD-9 189.0; renal pelvis, ICD-9 189.1; and ureter, ICD-9 189.2) or RCC only. This categorization has led to some confusion in epidemiological studies, resulting in a need to scrutinize previously reported data. RCC comprises 75 to 80% of the total, with the remainder being primarily transitional cell carcinomas of the renal pelvis and ureter. Separation of these two cancer types is appropriate since the pathogenesis of RCC and of transitional cell carcinoma is quite different, and epidemiological risk factors are distinct as are the signs and symptoms of the two diseases. This section focuses on RCC.
The major identified risk factor for kidney cancer is tobacco smoking, followed by suspected but poorly defined occupational and environmental risk factors. It is estimated that the elimination of tobacco smoking would decrease the incidence of kidney cancer by 30 to 40% in industrialized countries, but occupational determinants of RCC are not well established. The population attributable risk due to occupational exposures has been estimated to be between zero, based on recognized carcinogenesis, and 21%, based on a multicentric multisite case-control study in the Montreal area of Canada. Early biomarkers of effect in association with biomarkers of exposure should assist in clarifying important risk factors. Several occupations and industries have been found in epidemiological studies to entail an increased risk of renal cancer. However, with the possible exception of agents used in dry cleaning and exposures in petroleum refining, the available evidence is not consistent. Statistical analysis of epidemiological exposure data in relation to biomarkers of susceptibility and effect will clarify additional aetiological causes.
Several epidemiological studies have associated specific industries, occupations and occupational exposures with increased risks of renal cell carcinoma. The pattern that emerges from these studies is not fully consistent. Oil refining, printing, dry cleaning and truck driving are examples of jobs associated with excess risk of kidney cancer. Farmers usually display decreased risk of RCC, but a Danish study linked long-term exposure to insecticides and herbicides with an almost fourfold excess of RCC risk. This finding requires confirmation in independent data, including specification of the possible causal nature of the association. Other products suspected of being associated with RCC include: various hydrocarbon derivatives and solvents; products of oil refining; petroleum, tar and pitch products; gasoline exhaust; jet fuel; jet and diesel engine emissions; arsenic compounds; cadmium; chromium (VI) compounds; inorganic lead compounds; and asbestos. Epidemiological studies have associated occupational gasoline vapour exposure with kidney cancer risk, some in a dose-response fashion, a phenomenon observed in the male rat for unleaded gasoline vapour exposure. These findings gain some potential weight, given the widespread human exposure to gasoline vapours in retail service stations and the recent increase in kidney cancer incidence. Gasoline is a complex mixture of hydrocarbons and additives, including benzene, which is a known human carcinogen.
The risk of kidney cancer is not consistently linked with social class, although increased risk has occasionally been associated with higher socio-economic status. However, in some populations a reverse gradient was observed, and in yet others, no clear pattern emerged. Possibly these variations may be related to lifestyle. Studies with migrant people show modification in RCC risk towards the level of the host country population, suggesting that environmental factors are important in the development of this malignancy.
Except for nephroblastoma (Wilms’ tumour), which is a childhood cancer, kidney cancer usually occurs after 40 years of age. An estimated 127,000 new cases of kidney cancer (including RCC and transitional cell carcinoma (TCC) of the renal pelvis and ureter), corresponding to 1.7% of the world total cancer incidence, occurred globally in 1985. The incidence of kidney cancer varies among populations. High rates have been reported for both men and women in North America, Europe, Australia and New Zealand; low rates in Melanesia, middle and eastern Africa and southeastern and eastern Asia. The incidence of kidney cancer has been increasing in most western countries, but stagnated in a few. Age-standardized incidence of kidney cancer in 1985 was highest in North America and western, northern and eastern Europe, and lowest in Africa, Asia (except in Japanese men) and the Pacific. Kidney cancer is more frequent in men than in women and ranks among the ten most frequent cancers in a number of countries.
Transitional cell carcinoma (TCC) of the renal pelvis is associated with similar aetiological agents as bladder cancer, including chronic infection, stones and phenacetin-containing analgesics. Balkan nephropathy, a slowly progressive, chronic and fatal nephropathy prevalent in the Balkan countries, is associated with high rates of tumours of the renal pelvis and ureter. The causes of Balkan nephropathy are unknown. Excessive exposure to ochratoxin A, which is considered possibly carcinogenic to humans, has been associated with the development of Balkan nephropathy, but the role of other nephrotoxic agents cannot be excluded. Ochratoxin A is a toxin produced by fungi which can be found in many food stuffs, particularly cereals and pork products.
Screening and diagnosis of kidney cancer
The sign and symptom pattern of RCC varies among patients, even up to the stage when metastasis appears. Because of the location of the kidneys and the mobility of contiguous organs to the expanding mass, these tumours are frequently very large at the time of clinical detection. Although haematuria is the primary symptom of RCC, bleeding occurs late compared to transitional cell tumours because of the intra-renal location of RCC. RCC has been considered the “medical doctor’s dream” but the “surgeon’s curse” because of the interesting constellation of symptoms related to paraneoplastic syndromes. Substances that increase the red blood cell count, calcium and factors which mimic abnormal adrenal gland function have been reported, and abdominal mass, weight loss, fatigue, pain, anaemia, abnormal liver function and hypertension have all been observed. Computerized axial tomography (CAT scan) of the abdomen and ultrasound are being ordered by physicians with increased frequency so, consequently, it is estimated that 20% of RCCs are diagnosed serendipitously as a result of evaluation for other medical problems.
Clinical evaluation of an RCC case consists of a physical examination to identify a flank mass, which occurs in 10% of patients. A kidney x ray with contrast may delineate a renal mass and the solid or cystic nature is usually clarified by ultrasound or CAT scan. The tumours are highly vascular and have a characteristic appearance when the artery is injected with radio-opaque contrast material. Arteriography is performed to embolize the tumour if it is very large or to define the arterial blood supply if a partial nephrectomy is anticipated. Fine-needle aspiration may be used to sample suspect RCC.
Localized RCC tumours are surgically removed with regional lymph nodes and, operatively, early ligation of the artery and vein is important. Symptomatically, the patient may be improved by removing large or bleeding tumours that have metastasized, but this does not improve survival. For metastatic tumours, localized pain control may be achieved with radiation therapy but the treatment of choice for metastatic disease is biological response modifiers (Interleukin-2 or α-interferon), although chemotherapy is occasionally used alone or in combination with other therapies.
Markers such as the cancer gene on chromosome 3 observed in cancer families and in von Hippel-Lindau disease may serve as biomarkers of susceptibility. Although tumour marker antigens have been reported for RCC, there is currently no way to detect these reliably in the urine or blood with adequate sensitivity and specificity. The low prevalence of this disease in the general population requires a high specificity and sensitivity test for early disease detection. Occupational cohorts at risk could potentially be screened with ultrasound. Evaluation of this tumour remains a challenge to the basic scientist, molecular epidemiologist and clinician alike.
Bladder Cancer
Epidemiology
More than 90% of bladder cancers in Europe and North America are transitional cell carcinomas (TCC). Squamous cell carcinoma and adenocarcinoma account for 5 and 1%, respectively, of bladder cancer in these regions. The distribution of histopathological types in bladder cancer is strikingly different in regions such as the Middle East and Africa where bladder cancer is associated with schistosomal infection. For instance, in Egypt, where schistosomiasis is endemic and bladder cancer is the major oncogenic problem, the most common type is squamous cell carcinoma, but the incidence of TCC is increasing with the rising prevalence of cigarette smoking. The discussion which follows focuses on TCC.
Bladder cancer continues to be a disease of significant importance. It accounted for about 3.5% of all malignancies in the world in 1980. In 1985, bladder cancer was estimated to be 11th in frequency on a global scale, being the eighth most frequent cancer among men, with an expected total of 243,000 new cases. There is a peak incidence in the seventh decade of life, and worldwide the male to female ratio is around three to one. Incidence has been increasing in almost all populations in Europe, particularly in men. In Denmark, where annual incidence rates are among the highest in the world, at 45 per 100,000 in men and 12 per 100,000 in women, the recent trend has been a further rise of 8 to 9% every 5 years. In Asia, the very high rates among the Chinese in Hong Kong have declined steadily, but in both sexes bladder cancer incidence is still much higher than elsewhere in Asia and more than twice as high as that among the Chinese in Shanghai or Singapore. Bladder cancer rates among the Chinese in Hawaii are also high.
Cigarette smoking is the single most important aetiological factor in bladder cancer, and occupational exposures rank second. It has been estimated that tobacco is responsible for one-third of all bladder cancer cases outside of regions where schistosomal infection is prevalent. The number of bladder cancer cases attributed in 1985 to tobacco smoking has been estimated at more than 75,000 worldwide, and may account for 50% of bladder cancer in western populations. The fact that all individuals who smoke similar amounts do not develop bladder cancer at the same rate suggests genetic factors are important in controlling the susceptibility. Two aromatic amines, 4-aminobiphenyl and 2-naphthylamine, are carcinogens associated with cigarette smoking; these are found in higher concentrations in “black tobacco” (air-cured) than in “blend tobacco” (flue-cured). Passive smoke increases the adducts in the blood and a dose-response of adduct formation has been correlated with increased risk of bladder cancer. Higher levels of adduct formation have been observed in cigarette smokers who are slow acetylators compared to fast acetylators, which suggests that genetically inherited acetylation status may be an important biomarker of susceptibility. The lower incidence of bladder cancer in Black compared to White races may be attributed to conjugation of carcinogenic metabolic intermediates by sulphotransferases that produce electrophiles. Detoxified phenolic sulphates may protect the urothelium. Liver sulphotransferase activity for N-hydroxyarylamines has been reported to be higher in Blacks than Whites. This may result in a decrease in the amount of free N-hydroxymetabolites to function as carcinogens.
Occupational bladder cancer is one of the earliest known and best documented occupational cancers. The first identified case of occupational bladder cancer appeared some 20 years after the inception of the synthetic dye industry in Germany. Numerous other occupations have been identified in the last 25 years as occupational bladder cancer risks. Occupational exposures may contribute to up to 20% of bladder cancers. Workers occupationally exposed include those working with coal-tar pitches, coal gasification and production of rubber, aluminium, auramine and magenta, as well as those working as hairdressers and barbers. Aromatic amines have been shown to cause bladder cancer in workers in many countries. Notable among this class of chemicals are 2-naphthylamine, benzidine, 4-nitrobiphenyl and 3,3r´-dichlorobenzidine. Two other aromatic amines, 4,4´-methylene dianiline (MDA) and 4,4´-methylene-bis-2-chloroaniline (MOCA) are among the most widely used of the suspected bladder carcinogens. Other carcinogens associated with industrial exposures are largely undetermined; however, aromatic amines are frequently present in the workplace.
Screening and diagnosis of bladder cancer
Screening for bladder cancer continues to receive attention in the quest to diagnose bladder cancer before it becomes symptomatic and, presumably, less amenable to curative treatment. Voided urine cytology and urinalysis for haematuria have been considered candidate screening tests. A pivotal question for screening is how to identify high-risk groups and then individuals within these groups. Epidemiological studies identify groups at risk while biomarkers potentially identify individuals within groups. In general, occupational screening for bladder cancer with haematuria testing and Papanicolaou cytology has been ineffective.
Improved detection of bladder cancer may be possible using the 14-day hemastick testing described by Messing and co-workers. A positive test was observed at least once in 84% of 31 patients with bladder cancer at least 2 months prior to the cystoscopic diagnosis of disease. This test suffers from a false-positive rate of 16 to 20% with half of these patients having no urological disease. The low cost may make this a useful test in a two-tier screen in combination with biomarkers and cytology (Waples and Messing 1992).
In a recent study, the DD23 monoclonal antibody using quantitative fluorescence image analysis detected bladder cancer in exfoliated uroepithelial cells. A sensitivity of 85% and specificity of 95% were achieved in a mixture of low- and high-grade transitional cell carcinomas including TaT1 tumours. The M344 tumour-associated antigen in conjunction with DNA ploidy had a sensitivity approaching 90%.
Recent studies indicate combining biomarkers with haematuria testing may be the best approach. A list of the applications of quantitative fluorescence urinary cytology in combination with biomarkers is summarized in Table 1. Genetic, biochemical and morphological early cell changes associated with premalignant conditions support the concept that individuals at risk can be identified years in advance of the development of overt malignancy. Biomarkers of susceptibility in combination with biomarkers of effect promise to detect individuals at risk with an even higher precision. These advances are made possible by new technologies capable of quantitating phenotypic and genotypic molecular changes at the single cell level thus identifying individuals at risk. Individual risk assessment facilitates stratified, cost-effective monitoring of selected groups for targeted chemoprevention.
Table 1. Applications of urinary cytology
Detection of CIS1 and bladder cancer
Monitoring surgical therapy:
Monitoring bladder following TURBT2
Monitoring upper urinary tract
Monitoring urethral remnant
Monitoring urinary diversion
Monitoring intravesical therapy
Selecting intravesical therapy
Monitoring effect of laser therapy
Evaluation of patients with haematuria
Establishing need for cystoscopy
Screening high-risk populations:
Occupational exposure groups
Drug abuse groups at risk for bladder cancer
Decision criteria for:
Cystectomy
Segmental ureteral resection versus nephroureterectomy
Other indications:
Detecting vesicoenteric fistula
Extraurological tumours invading the urinary tract
Defining effective chemopreventive agents
Monitoring effective chemotherapy
1 CIS, carcinoma in situ.
2 TURBT, transurethral resection for bladder tumour.
Source: Hemstreet et al. 1996.
Signs and symptoms of bladder cancer are similar to those of urinary tract infection and may include pain on urination, frequent voiding and blood and pus cells in the urine. Because symptoms of a urinary tract infection may herald a bladder tumour particularly when associated with gross haematuria in older patients, confirmation of the presence of bacteria and a keen awareness by the examining physician is needed. Any patient treated for a urinary tract infection which does not resolve immediately should be referred to a urology specialist for further evaluation.
Diagnostic evaluation of bladder cancer first requires an intravenous pyelogram (IVP) to exclude upper tract disease in the renal pelvis or ureters. Confirmation of bladder cancer requires looking in the bladder with a light (cystoscope) with multiple biopsies performed with a lighted instrument through the urethra to determine if the tumour is non-invasive (i.e., papillary or CIS) or invasive. Random biopsies of the bladder and prostatic urethra help to define field cancerization and field effect changes. Patients with non-invasive disease require close monitoring, as they are at risk of subsequent recurrences, although stage and grade progression are uncommon. Patients who present with bladder cancer that is already high-grade or invasive into the lamina propria are at equally high risk of recurrence but stage progression is much more likely. Thus, they usually receive intravesical instillation of immuno- or chemotherapeutic agents following transurethral resection. Patients with tumours invading the muscularis propria or beyond are much more likely to have metastasis already and can rarely be managed by conservative means. However, even when treated by total cystectomy (the standard therapy for muscle-invading bladder cancer), 20 to 60% eventually succumb to their disease, almost always due to metastasis. When regional or distal metastasis is present at diagnosis, the 5-year survival rates drop to 35 and 9%, respectively, despite aggressive treatment. Systemic chemotherapy for metastatic bladder cancer is improving with complete response rates reported at 30%. Recent studies suggest chemotherapy prior to cystectomy may improve survival in selected patients.
Bladder cancer staging is predictive of the biological potential for progression, metastasis, or recurrence in 70% of the cases. Staging of bladder cancer usually requires CAT scan to rule out liver metastasis, radioisotope bone scan to exclude spread to the bone, and chest x ray or CAT scan to exclude lung metastasis. A search continues for biomarkers in the tumour and the bladder cancer field that will predict which tumours will metastasize or recur. The accessibility of exfoliated bladder cells in voided specimens shows promise for using biomarkers for monitoring recurrence and for cancer prevention.
Reproductive System: Introduction
Male and female reproductive toxicity are topics of increasing interest in consideration of occupational health hazards. Reproductive toxicity has been defined as the occurrence of adverse effects on the reproductive system that may result from exposure to environmental agents. The toxicity may be expressed as alterations to the reproductive organs and/or the related endocrine system. The manifestations of such toxicity may include:
- alterations in sexual behaviour
- reduced fertility
- adverse pregnancy outcomes
- modifications of other functions that are dependent on the integrity of the reproductive system.
Mechanisms underlying reproductive toxicity are complex. More xenobiotic substances have been tested and demonstrated to be toxic to the male reproductive process than to the female. However, it is not known whether this is due to underlying differences in toxicity or to the greater ease of studying sperm than oocytes.
Developmental Toxicity
Developmental toxicity has been defined as the occurrence of adverse effects on the developing organism that may result from exposure prior to conception (either parent), during pprenatal development or postnatally to the time of sexual maturation. Adverse developmental effects may be detected at any point in the life span of the organism. The major manifestations of developmental toxicity include:
- death of the developing organism
- structural abnormality
- altered growth
- functional deficiency.
In the following discussion, developmental toxicity will be used as an all-inclusive term to refer to exposures to the mother, father or conceptus that lead to abnormal development. The term teratogenesis will be used to refer more specifically to exposures to the conceptus which produce a structural malformation. Our discussion will not include the effects of postnatal exposures on development.
Mutagenesis
In addition to reproductive toxicity, exposure to either parent prior to conception has the potential of resulting in developmental defects through mutagenesis, changes in the genetic material that is passed from parent to offspring. Such changes can occur either at the level of individual genes or at the chromosomal level. Changes in individual genes can result in the transmission of altered genetic messages while changes at the chromosomal level can result in the transmission of abnormalities in chromosomal number or structure.
It is interesting that some of the strongest evidence for a role for preconception exposures in developmental abnormalities comes from studies of paternal exposures. For example, Prader-Willi syndrome, a birth defect characterized by hypotonicity in the newborn period and, later, marked obesity and behaviour problems, has been associated with paternal occupational exposures to hydrocarbons. Other studies have shown associations between paternal preconception exposures to physical agents and congenital malformations and childhood cancers. For example, paternal occupational exposure to ionizing radiation has been associated with an increased risk of neural tube defects and increased risk of childhood leukaemia, and several studies have suggested associations between paternal preconception occupational exposure to electromagnetic fields and childhood brain tumours (Gold and Sever 1994). In assessing both reproductive and developmental hazards of workplace exposures increased attention must be paid to the ppossibleeffects among males.
It is quite likely that some defects of unknown aetiology involve a genetic component which may be related to parental exposures. Because of associations demonstrated between father’s age and mutation rates it is logical to believe that other paternal factors and exposures may be associated with gene mutations. The well-established association between maternal age and chromosomal non-disjunction, resulting in abnormalities in chromosomal number, suggests a significant role for maternal exposures in chromosomal abnormalities.
As our understanding of the human genome increases it is likely that we will be able to trace more developmental defects to mutagenic changes in the DNA of single genes or structural changes in portions of chromosomes.
Teratogenesis
The adverse effects on human development of exposure of the conceptus to exogenous chemical agents have been recognized since the discovery of the teratogenicity of thalidomide in 1961. Wilson (1973) has developed six “general principles of teratology” that are relevant to this discussion. These principles are:
- The final manifestations of abnormal development are death, malformation, growth retardation and functional disorder.
- Susceptibility of the conceptus to teratogenic agents varies with the developmental stage at the time of exposure.
- Teratogenic agents act in specific ways (mechanisms) on developing cells and tissues in initiating abnormal embryogenesis (pathogenesis).
- Manifestations of abnormal development increase in degree from the no-effect to the totally lethal level as dosage increases.
- The access of adverse environmental influences to developing tissues depends on the nature of the agent.
- Susceptibility to a teratogen depends on the genotype of the conceptus and on the manner in which the genotype interacts with environmental factors.
The first four of these principles will be discussed in further detail, as will the combination of principles 1, 2 and 4 (outcome, exposure timing and dose).
Spectrum of Adverse Outcomes Associatedwith Exposure
There is a spectrum of adverse outcomes potentially associated with exposure. Occupational studies that focus on a single outcome risk overlooking other important reproductive effects.
Figure 1 lists some examples of developmental outcomes potentially associated with exposure to occupational teratogens. Results of some occupational studies have suggested that congenital malformations and spontaneous abortions are associated with the same exposures—for example, anaesthetic gases and organic solvents.
Spontaneous abortion is an important outcome to consider because it can result from different mechanisms through several pathogenic processes. A spontaneous abortion can be the result of toxicity to the embryo or foetus, chromosomal alterations, single gene effects or morphological abnormalities. It is important to try to differentiate between karyotypically normal and abnormal conceptuses in studies of spontaneous abortions.
Figure 1. Developmental abnormalities and reproductive outcomes potentially associated with occupational exposures.
Timing of Exposure
Wilson’s second principle relates susceptibility to abnormal development to the time of exposure, that is, the gestational age of the conceptus. This principle has been well established for the induction of structural malformations, and the sensitive periods for organogenesis are known for many structures. Considering an expanded array of outcomes, the sensitive period during which any effect can be induced must be extended throughout gestation.
In assessing occupational developmental toxicity, exposure should be determined and classified for the appropriate critical period—that is, gestational age(s)—for each outcome. For example, spontaneous abortions and congenital malformations are likely to be related to first and second trimester exposure, whereas low birth weight and functional disorders such as seizure disorders and mental retardation are more likely to be related to second and third trimester exposure.
Teratogenic Mechanisms
The third principle is the importance of considering the potential mechanisms that might initiate abnormal embryogenesis. A number of different mechanisms have been suggested which could lead to teratogenesis (Wilson 1977). These include:
- mutational changes in DNA sequences
- chromosomal abnormalities leading to structural or quantitative changes in DNA
- alteration or inhibition of intracellular metabolism, e.g., metabolic blocks and lack of co-enzymes, precursors or substrates for biosynthesis
- interruption of DNA or RNA synthesis
- interference with mitosis
- interference with cell differentiation
- failure of cell-to-cell interactions
- failure of cell migrations
- cell death through direct cytotoxic effects
- effects on cell membrane permeability and osmolar changes
- physical disruption of cells or tissues.
By considering mechanisms, investigators can develop biologically meaningful groupings of outcomes. This can also provide insight into potential teratogens; for example, relationships between carcinogenesis, mutagenesis and teratogenesis have been discussed for some time. From the perspective of assessing occupational reproductive hazards, this is of particular importance for two distinct reasons: (1) substances that are carcinogenic or mutagenic have an increased probability of being teratogenic, suggesting that particular attention should be paid to the reproductive effects of such substances, and (2) effects on deoxyribonucleic acid (DNA), producing somatic mutations, are thought to be mechanisms for both carcinogenesis and teratogenesis.
Dose and Outcome
The fourth principle concerning teratogenesis is the relationship of outcome to dose. This principle is clearly established in many animal studies, and Selevan (1985) has discussed its potential relevance to the human situation, noting the importance of multiple reproductive outcomes within specific dose ranges and suggesting that a dose-response relationship could be reflected in an increasing rate of a particular outcome with increasing dose and/or a shift in the spectrum of the outcomes observed.
In regard to teratogenesis and dose, there is considerable concern about functional disturbances resulting from the ppossiblebehavioural effects of pprenatal exposure to environmental agents. Animal behavioural teratology is expanding rapidly, but human behavioural environmental teratology is in a relatively early stage of development. At present, there are critical limitations in the definition and ascertainment of appropriate behavioural outcomes for epidemiological studies. In addition it is ppossiblethat low-level exposures to developmental toxicants are important for some functional effects.
Multiple Outcomes and Exposure Timing and Dose
Of particular importance with respect to the identification of workplace developmental hazards are the concepts of multiple outcomes and exposure timing and dose. On the basis of what we know about the biology of development, it is clear that there are relationships between reproductive outcomes such as spontaneous abortion and intrauterine growth retardation and congenital malformations. In addition, multiple effects have been shown for many developmental toxicants (table 1).
Table 1. Examples of exposures associated with multiple adverse reproductive end-points
| Exposure | Outcome | |||
| Spontaneous abortion | Congenital malformation | Low birth weight | Developmental disabilities | |
| Alcohol | X | X | X | X |
| Anaesthetic gases |
X | X | ||
| Lead | X | X | X | |
| Organic solvents | X | X | X | |
| Smoking | X | X | X | |
Relevant to this are issues of exposure timing and dose-response relationships. It has long been recognized that the embryonic period during which organogenesis occurs (two to eight weeks post-conception) is the time of greatest sensitivity to the induction of structural malformations. The foetal period from eight weeks to term is the time of histogenesis, with rapid increase in cell number and cellular differentiation occurring during this time. It is then that functional abnormalities and growth retardation are most likely to be induced. It is ppossiblethat there may be relationships between dose and response during this period where a high dose might lead to growth retardation and a lower dose might result in functional or behavioural disturbance.
Male-Mediated Developmental Toxicity
While developmental toxicity is usually considered to result from exposure of the female and the conceptus—that is, teratogenic effects—there is increasing evidence from both animal and human studies for male-mediated developmental effects. Proposed mechanisms for such effects include transmission of chemicals from the father to the conceptus via seminal fluid, indirect contamination of the mother and the conceptus by substances carried from the workplace into the home environment through personal contamination, and—as noted earlier—paternal preconception exposures that result in transmissible genetic changes (mutations).
Introduction to the Male and Female Reproductive Function
Reproductive toxicity has many unique and challenging differences from toxicity to other systems. Whereas other forms of environmental toxicity typically involve development of disease in an exposed individual, because reproduction requires interaction between two individuals, reproductive toxicity will be expressed within a reproductive unit, or couple. This unique, couple- dependent aspect, although obvious, makes reproductive toxicology distinct. For example, it is ppossiblethat exposure to a toxicant by one member of a reproductive couple (e.g., the male) will be manifest by an adverse reproductive outcome in the other member of the couple (e.g., increased frequency of spontaneous abortion). Any attempt to deal with environmental causes of reproductive toxicity must address the couple-specific aspect.
There are other unique aspects that reflect the challenges of reproductive toxicology. Unlike renal, cardiac or pulmonary function, reproductive function occurs intermittently. This means that occupational exposures can interfere with reproduction but go unnoticed during periods when fertility is not desired. This intermittent characteristic can make the identification of a reproductive toxicant in humans more difficult. Another unique characteristic of reproduction, which follows directly from the consideration above, is that complete assessment of the functional integrity of the reproductive system requires that the couple attempt pregnancy.
Male Reproductive System and Toxicology
Spermatogenesis and spermiogenesis are the cellular processes that produce mature male sex cells. These processes take place within the seminiferous tubules of the testes of the sexually mature male, as shown in Figure 1. The human seminiferous tubules are 30 to 70 cm long and 150 to 300 mm in diameter (Zaneveld 1978). The spermatogonia (stem cells) are ppositioned along the basement membrane of the seminiferous tubules and are the basic cells for the production of sperm.
Figure 1. The male reproductive system
Sperm mature through a series of cellular divisions in which the spermatogonia proliferate and become primary spermatocytes. The resting primary spermatocytes migrate through tight junctions formed by the Sertoli cells to the luminal side of this testis barrier. By the time the spermatocytes reach the membrane barrier in the testis, the synthesis of DNA, the genetic material in the nucleus of the cell, is essentially complete. When the primary spermatocytes actually encounter the lumen of the seminiferous tubule, these undergo a special type of cell division which occurs only in germ cells and is known as meiosis. Meiotic cellular divison results in the splitting up of the chromosomes pairs in the nucleus, so that each resulting germ cell contains only a single copy of each chromosome strand rather than a matched pair.
During meiosis the chromosomes change shape by condensing and becoming filamentous. At a certain point, the nuclear membrane which surrounds them breaks down and microtubular spindles attach to the chromosomal pairs, causing them to separate. This completes the first meiotic division and two haploid secondary spermatocytes are formed. The secondary spermatocytes then undergo a second meiotic division to form equal numbers of X- and Y-chromosome bearing spermatids.
The morphological transformation of spermatids to spermatozoa is called spermiogenesis. When spermiogenesis is complete, each sperm cell is released by the Sertoli cell into the seminiferous tubule lumen by a process referred to as spermiation. The sperm migrate along the tubule to the rete testis and into the head of the epididymis. Sperm leaving the seminiferous tubules are immature: unable to fertilize an ovum and unable to swim. Spermatozoa released into the lumen of the seminiferous tubule are suspended in fluid pproduced primarily by the Sertoli cells. Concentrated sperm suspended within this fluid flow continuously from the seminiferous tubules, through slight changes in the ionic milieu within the rete testis, through the vasa efferentia, and into the epididymis. The epididymis is a single highly coiled tube (five to six metres long) in which sperm spend 12 to 21 days.
Within the epididymis, sperm progressively acquire motility and fertilizing capacity. This may be due to the changing nature of the suspension fluid in the epididymis. That is, as the cells mature the epididymis absorbs components from the fluid including secretions from the Sertoli cells (e.g., androgen binding protein), thereby increasing the concentration of spermatozoa. The epididymis also contributes its own secretions to the suspension fluid, including the chemicals glycerylphosphorylcholine (GPC) and carnitine.
Sperm morphology continues to transform in the epididymis. The cytoplasmic droplet is shed and the sperm nucleus condenses further. While the epididymis is the principal storage reservoir for sperm until ejaculation, about 30% of the sperm in an ejaculate have been stored in the vas deferens. Frequent ejaculation accelerates passage of sperm through the epididymis and may increase the number of immature (infertile) sperm in the ejaculate (Zaneveld 1978).
Ejaculation
Once within the vas deferens, the sperm are transported by the muscular contractions of ejaculation rather than by the flow of fluid. During ejaculation, fluids are forcibly expelled from the accessory sex glands giving rise to the seminal plasma. These glands do not expel their secretions at the same time. Rather, the bulbourethral (Cowper’s) gland first extrudes a clear fluid, followed by the prostatic secretions, the sperm-concentrated fluids from the epididymides and ampulla of the vas deferens, and finally the largest fraction primarily from the seminal vesicles. Thus, seminal plasma is not a homogeneous fluid.
Toxic Actions on Spermatogenesisand Spermiogenesis
Toxicants may disrupt spermatogenesis at several points. The most damaging, because of irreversibility, are toxicants that kill or genetically alter (beyond repair mechanisms) spermatogonia or Sertoli cells. Animal studies have been useful to determine the stage at which a toxicant attacks the spermatogenic process. These studies employ short term exposure to a toxicant before sampling to determine the effect. By knowing the duration for each spermatogenic stage, one can extrapolate to estimate the affected stage.
Biochemical analysis of seminal plasma pprovides insights into the function of the accessory sex glands. Chemicals that are secreted primarily by each of the accessory sex glands are typically selected to serve as a marker for each respective gland. For example, the epididymis is represented by GPC, the seminal vesicles by fructose, and the prostate gland by zinc. Note that this type of analysis pprovides only gross information on glandular function and little or no information on the other secretory constituents. Measuring semen pH and osmolality provide additional general information on the nature of seminal plasma.
Seminal plasma may be analysed for the presence of a toxicant or its metabolite. Heavy metals have been detected in seminal plasma using atomic absorption spectrophotometry, while halogenated hydrocarbons have been measured in seminal fluid by gas chromatography after extraction or protein-limiting filtration (Stachel et al. 1989; Zikarge 1986).
The viability and motility of spermatozoa in seminal plasma is typically a reflection of seminal plasma quality. Alterations in sperm viability, as measured by stain exclusion or by hypoosmotic swelling, or alterations in sperm motility parameters would suggest post-testicular toxicant effects.
Semen analyses also can indicate whether production of sperm cells has been affected by a toxicant. Sperm count and sperm morphology provide indices of the integrity of spermatogenesis and spermiogenesis. Thus, the number of sperm in the ejaculate is directly correlated with the number of germ cells per gram of testis (Zukerman et al. 1978), while abnormal morphology is probably a result of abnormal spermiogenesis. Dead sperm or immotile sperm often reflect the effects of post-testicular events. Thus, the type or timing of a toxic effect may indicate the target of the toxicant. For example, exposure of male rats to 2-methoxyethanol resulted in reduced fertility after four weeks (Chapin et al. 1985). This evidence, corroborated by histological examination, indicates that the target of toxicity is the spermatocyte (Chapin et al. 1984). While it is not ethical to intentionally expose humans to suspected reproductive toxicants, semen analyses of serial ejaculates of men inadvertently exposed for a short time to potential toxicants may provide similar useful information.
Occupational exposure to 1,2-dibromochloropropane (DBCP) reduced sperm concentration in ejaculates from a median of 79 million cells/ml in unexposed men to 46 million cells/ml in exposed workers (Whorton et al. 1979). Upon removing the workers from the exposure, those with reduced sperm counts experienced a partial recovery, while men who had been azoospermic remained sterile. Testicular biopsy revealed that the target of DBCP was the spermatogonia. This substantiates the severity of the effect when stem cells are the target of toxicants. There were no indications that DBCP exposure of men was associated with adverse pregnancy outcome (Potashnik and Abeliovich 1985). Another example of a toxicant targeting spermatogenesis/spermiogenesis was the study of workers exposed to ethylene dibromide (EDB). They had more sperm with tapered heads and fewer sperm per ejaculate than did controls (Ratcliffe et al. 1987).
Genetic damage is difficult to detect in human sperm. Several animal studies using the dominant lethal assay (Ehling et al. 1978) indicate that paternal exposure can produce an adverse pregnancy outcome. Epidemiological studies of large populations have demonstrated increased frequency of spontaneous abortions in women whose husbands were working as motor vehicle mechanics (McDonald et al. 1989). Such studies indicate a need for methods to detect genetic damage in human sperm. Such methods are being developed by several laboratories. These methods include DNA probes to discern genetic mutations (Hecht 1987), sperm chromosome karyotyping (Martin 1983), and DNA stability assessment by flow cytometry (Evenson 1986).
Figure 2. Exposures positively associated with adversely affecting semen quality
Figure 2 lists exposures known to affect sperm quality and table 1 provides a summary of the results of epidemiological studies of paternal effects on reproductive outcomes.
Table 1. Epidemiological studies of paternal effects on pregnancy outcome
| Reference | Type of exposure or occupation | Association with exposure1 | Effect |
| Record-based population studies | |||
| Lindbohm et al. 1984 | Solvents | – | Spontaneous abortion |
| Lindbohm et al. 1984 | Service station | + | Spontaneous abortion |
| Daniell and Vaughan 1988 | Organic solvents | – | Spontaneous abortion |
| McDonald et al. 1989 | Mechanics | + | Spontaneous abortion |
| McDonald et al. 1989 | Food processing | + | Developmental defects |
| Lindbohm et al. 1991a | Ethylene oxide | + | Spontaneous abortion |
| Lindbohm et al. 1991a | Petroleum refinery | + | Spontaneous abortion |
| Lindbohm et al. 1991a | Impregnates of wood | + | Spontaneous abortion |
| Lindbohm et al. 1991a | Rubber chemicals | + | Spontaneous abortion |
| Olsen et al. 1991 | Metals | + | Child cancer risk |
| Olsen et al. 1991 | Machinists | + | Child cancer risk |
| Olsen et al. 1991 | Smiths | + | Child cancer risk |
| Kristensen et al. 1993 | Solvents | + | Preterm birth |
| Kristensen et al. 1993 | Lead and solvents | + | Preterm birth |
| Kristensen et al. 1993 | Lead | + | Perinatal death |
| Kristensen et al. 1993 | Lead | + | Male child morbidity |
| Case-control studies | |||
| Kucera 1968 | Printing industry | (+) | Cleft lip |
| Kucera 1968 | Paint | (+) | Cleft palate |
| Olsen 1983 | Paint | + | Damage to central nervous system |
| Olsen 1983 | Solvents | (+) | Damage to central nervous system |
| Sever et al. 1988 | Low-level radiation | + | Neural tube defects |
| Taskinen et al. 1989 | Organic solvents | + | Spontaneous abortion |
| Taskinen et al. 1989 | Aromatic hydrocarbons | + | Spontaneous abortion |
| Taskinen et al. 1989 | Dust | + | Spontaneous abortion |
| Gardner et al. 1990 | Radiation | + | Childhood leukaemia |
| Bonde 1992 | Welding | + | Time to conception |
| Wilkins and Sinks 1990 | Agriculture | (+) | Child brain tumour |
| Wilkins and Sinks 1990 | Construction | (+) | Child brain tumour |
| Wilkins and Sinks 1990 | Food/tobacco processing | (+) | Child brain tumour |
| Wilkins and Sinks 1990 | Metal | + | Child brain tumour |
| Lindbohmn et al. 1991b | Lead | (+) | Spontaneous abortion |
| Sallmen et al. 1992 | Lead | (+) | Congenital defects |
| Veulemans et al. 1993 | Ethylene glycol ether | + | Abnormal spermiogram |
| Chia et al. 1992 | Metals | + | Cadmium in semen |
1 – no significant association; (+) marginally significant association; + significant association.
Source: Adapted from Taskinen 1993.
Neuroendocrine System
The overall functioning of the reproductive system is controlled by the nervous system and the hormones pproduced by the glands (the endocrine system). The reproductive neuroendocrine axis of the male involves principally the central nervous systems (CNS), the anterior pituitary gland and the testes. Inputs from the CNS and from the periphery are integrated by the hypothalamus, which directly regulates gonadotrophin secretion by the anterior pituitary gland. The gonadotrophins, in turn, act principally upon the Leydig cells within the interstitium and Sertoli and germ cells within the seminiferous tubules to regulate spermatogenesis and hormone production by the testes.
Hypothalamic–Pituitary Axis
The hypothalamus secretes the neurohormone gonadotrophin releasing hormone (GnRH) into the hypophysial portal vasculature for transport to the anterior pituitary gland. The pulsatile secretion of this decapeptide causes the concomitant release of luteinizing hormone (LH), and with lesser synchrony and one-fifth the potency, the release of follicle stimulating hormone (FSH) (Bardin 1986). Substantial evidence exists to support the presence of a separate FSH releasing hormone, although none has yet been isolated (Savy-Moore and Schwartz 1980; Culler and Negro-Vilar 1986). These hormones are secreted by the anterior pituitary gland. LH acts directly upon the Leydig cells to stimulate synthesis and release of testosterone, whereas FSH stimulates aromatization of testosterone to estradiol by the Sertoli cell. Gonadotropic stimulation causes the release of these steroid hormones into the spermatic vein.
Gonadotrophin secretion is, in turn, checked by testosterone and estradiol through negative feedback mechanisms. Testosterone acts principally upon the hypothalamus to regulate GnRH secretion and thereby reduces the pulse frequency, primarily, of LH release. Estradiol, on the other hand, acts upon the pituitary gland to reduce the magnitude of gonadotrophin release. Through these endocrine feedback loops, testicular function in general and testosterone secretion specifically are maintained at a relatively steady state.
Pituitary–Testicular Axis
LH and FSH are generally viewed as necessary for normal spermatogenesis. Presumably the effect of LH is secondary to inducing high intratesticular concentrations of testosterone. Therefore, FSH from the pituitary gland and testosterone from the Leydig cells act upon the Sertoli cells within the seminiferous tubule epithelium to initiate spermatogenesis. Sperm production persists, although quantitatively reduced, after removing either LH (and presumably the high intratesticular testosterone concentrations) or FSH. FSH is required for initiating spermatogenesis at puberty and, to a lesser extent, to reinitiate spermatogenesis that has been arrested (Matsumoto 1989; Sharpe 1989).
The hormonal synergism that serves to maintain spermatogenesis may entail recruitment by FSH of differentiated spermatogonia to enter meiosis, while testosterone may control specific, subsequent stages of spermatogenesis. FSH and testosterone may also act upon the Sertoli cell to stimulate production of one or more paracrine factors which may affect the number of Leydig cells and testosterone production by these cells (Sharpe 1989). FSH and testosterone stimulate protein synthesis by Sertoli cells including synthesis of androgen binding protein (ABP), while FSH alone stimulates synthesis of aromatase and inhibin. ABP is secreted primarily into the seminiferous tubular fluid and is transported to the proximal portion of the caput epididymis, possibly serving as a local carrier of androgens (Bardin 1986). Aromatase catalyses the conversion of testosterone to estradiol in the Sertoli cells and in other peripheral tissues.
Inhibin is a glycoprotein consisting of two dissimilar, disulphide-linked subunits, a and b. Although inhibin preferentially inhibits FSH release, it may also attenuate LH release in the presence of GnRH stimulation (Kotsugi et al. 1988). FSH and LH stimulate inhibin release with approximately equal potency (McLachlan et al. 1988). Interestingly, inhibin is secreted into the spermatic vein blood as pulses which are synchronous to those of testosterone (Winters 1990). This probably does not reflect direct actions of LH or testosterone on Sertoli cell activity, but rather the effects of other Leydig cell products secreted either into the interstitial spaces or the circulation.
Prolactin, which is also secreted by the anterior pituitary gland, acts synergistically with LH and testosterone to promote male reproductive function. Prolactin binds to specific receptors on the Leydig cell and increases the amount of androgen receptor complex within the nucleus of androgen responsive tissues (Baker et al. 1977). Hyperprolactinaemia is associated with reductions of testicular and prostate size, semen volume and circulating concentrations of LH and testosterone (Segal et al. 1979). Hyperprolactinaemia has also been associated with impotency, apparently independent of altering testosterone secretion (Thorner et al. 1977).
If measuring steroid hormone metabolites in urine, consideration must be given to the potential that the exposure being studied may alter the metabolism of excreted metabolites. This is especially pertinent since most metabolites are formed by the liver, a target of many toxicants. Lead, for example, reduced the amount of sulphated steroids that were excreted into the urine (Apostoli et al. 1989). Blood levels for both gonadotrophins become elevated during sleep as the male enters puberty, while testosterone levels maintain this diurnal pattern through adulthood in men (Plant 1988). Thus blood, urine or saliva samples should be collected at approximately the same time of day to avoid variations due to diurnal secretory patterns.
The overt effects of toxic exposure targeting the reproductive neuroendocrine system are most likely to be revealed through altered biological manifestations of the androgens. Manifestations significantly regulated by androgens in the adult man that may be detected during a basic physical examination include: (1) nitrogen retention and muscular development; (2) maintenance of the external genitalia and accessory sexual organs; (3) maintenance of the enlarged larynx and thickened vocal cords causing the male voice; (4) beard, axillary and pubic hair growth and temporal hair recession and balding; (5) libido and sexual performance; (6) organ specific proteins in tissues (e.g., liver, kidneys, salivary glands); and (7) aggressive behaviour (Bardin 1986). Modifications in any of these traits may indicate that androgen production has been affected.
Examples of Toxicant Effects
Lead is a classic example of a toxicant that directly affects the neuroendocrine system. Serum LH concentrations were elevated in men exposed to lead for less than one year. This effect did not progress in men exposed for more than five years. Serum FSH levels were not affected. On the other hand, serum levels of ABP were elevated and those of total testosterone were reduced in men exposed to lead for more than five years. Serum levels of free testosterone were significantly reduced after exposure to lead for three to five years (Rodamilans et al. 1988). In contrast, serum concentrations of LH, FSH, total testosterone, prolactin, and total neutral 17-ketosteroids were not altered in workers with lower circulating levels of lead, even though the distribution frequency of sperm count was altered (Assennato et al. 1986).
Exposure of shipyard painters to 2-ethoxyethanol also reduced sperm count without a concurrent change in serum LH, FSH, or testosterone concentrations (Welch et al. 1988). Thus toxicants may affect hormone production and sperm measures independently.
Male workers involved in the manufacture of the nematocide DBCP experienced elevated serum levels of LH and FSH and reduced sperm count and fertility. These effects are apparently sequelae to DBCP actions upon the Leydig cells to alter androgen production or action (Mattison et al. 1990).
Several compounds may exert toxicity by virtue of structural similarity to reproductive steroid hormones. Thus, by binding to the respective endocrine receptor, toxicants may act as agonists or antagonists to disrupt biological responses. Chlordecone (Kepone), an insecticide that binds to oestrogen receptors, reduced sperm count and motility, arrested sperm maturation and reduced libido. While it is tempting to suggest that these effects result from chlordecone interfering with oestrogen actions at the neuroendocrine or testicular level, serum levels of testosterone, LH and FSH were not shown to be altered in these studies in a manner similar to the effects of oestradiol therapy. DDT and its metabolites also exhibit steroidal properties and might be expected to alter male reproductive function by interfering with steroidal hormone functions. Xenobiotics such as polychlorinated biphenyls, polybrominated biphenyls, and organochlorine pesticides may also interfere with male reproductive functions by exerting oestrogenic agonist/antagonist activity (Mattison et al. 1990).
Sexual Function
Human sexual function refers to the integrated activities of the testes and secondary sex glands, the endocrine control systems, and the central nervous system-based behavioural and psychological components of reproduction (libido). Erection, ejaculation and orgasm are three distinct, independent, physiological and psychodynamic events which normally occur concurrently in men.
Little reliable data are available on occupational exposure effects on sexual function due to the problems described above. Drugs have been shown to affect each of the three stages xof male sexual function (Fabro 1985), indicating the potential for occupational exposures to exert similar effects. Antidepressants, testosterone antagonists and stimulants of prolactin release effectively reduce libido in men. Antihypertensive drugs which act on the sympathetic nervous system induce impotence in some men, but surprisingly, priapism in others. Phenoxybenzamine, an adrenoceptive antagonist, has been used clinically to block seminal emission but not orgasm (Shilon, Paz and Homonnai 1984). Anticholinergic antidepressant drugs permit seminal emission while blocking seminal ejection and orgasm which results in seminal plasma seeping from the urethra rather than being ejected.
Recreational drugs also affect sexual function (Fabro 1985). Ethanol may reduce impotence while enhancing libido. Cocaine, heroin and high doses of cannabinoids reduce libido. Opiates also delay or impair ejaculation.
The vast and varied array of pharmaceuticals that has been shown to affect the male reproductive system pprovides support for the notion that chemicals found in the workplace may also be reproductive toxicants. Research methods that are reliable and practical for field study conditions are needed to assess this important area of reproductive toxicology.
" DISCLAIMER: The ILO does not take responsibility for content presented on this web portal that is presented in any language other than English, which is the language used for the initial production and peer-review of original content. Certain statistics have not been updated since the production of the 4th edition of the Encyclopaedia (1998)."